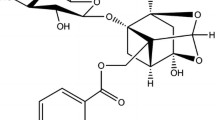
Abstract
The interaction of a flavonoid molecule (puerarin) with bovine serum albumin (BSA) was characterized by isothermal titration calorimetry (ITC), optical spectroscopic technique, and molecular modeling method under physiological conditions. The binding parameters for the reaction were calculated according to ITC experiments at different temperatures. The thermodynamic parameters, negative enthalpy changes (ΔH), and positive entropy (ΔS) indicated that the binding processes were entropically driven. The alterations of protein secondary structure in the presence of puerarin in aqueous solution were estimated by the evidences from FT-IR and CD spectroscopy with reductions of α-helices. On the basis of fluorescence resonance energy transfer (FRET) between excited tryptophan in BSA and BSA bound puerarin, the critical transfer distance and mean distance between tryptophan in BSA and puerarin were estimated.






Similar content being viewed by others
References
Guo Z, Jin Q, Fan G, Duan Y, Qin C, Wen M. Microwave assisted extraction of effective constituents from a Chinese herbal medicine radix puerariae. Anal Chim Acta. 2001;436:41–7.
Yeung DKY, Leung SWS, Xu YC, Vanhoutte PM, Man RYK. Puerarin an isoflavonoid derived from Radix puerariae, potentiates endothelium-independent relaxation via the cyclic AMP pathway in porcine coronary artery. Eur J Pharmacol. 2006;552:105–11.
Zhu JH, Wang XX, Chen JZ. Effects of puerarin on number and activity of endothelial progenitor cells from peripheral blood. Acta Pharmacol Sin. 2004;25:1045–51.
Cervellati R, Renzulli C, Guerra MC, Speroni E. Evaluation of antioxidant activity of some natural polyphenolic compounds using the Briggs-Rauscher reaction method. J Agric Food Chem. 2002;50:7504–9.
Benlhabib E, Baker JI, Keyler DE, Singh AK. Effects of purified puerarin on voluntary alcohol intake and alcohol withdrawal symptoms in p rats receiving free access to water and alcohol. J Med Food. 2004;7:180–6.
Yong PH, Hye GJ. Mechanism of phytoestrogen puerarin-mediated cytoprotection following oxidative injury: estrogen receptor-dependent up-regulation of PI3K/Akt and HO-1. Toxicol Appl Pharmacol. 2008;233:371–81.
Eishun T, Keitaro S, Akito N, Hiromi S, Teruyuki K, Koichi K. Artificial oxygen carriers, hemoglobin vesicles and albumin–hemes, based on bioconjugate chemistry. Bioconjug Chem. 2009;20:1419–40.
Katrahalli U, Jaldappagari S, Kalanur SS. Study of the interaction between fluoxetine hydrochloride and bovine serum albumin in the imitated physiological conditions by multi-spectroscopic methods. J Lumin. 2010;130:211–6.
Bojko B, Sułkowska A, Maciążek-Jurczyk M, Równicka J, Sułkowski WW. The influence of dietary habits and pathological conditions on the binding of theophylline to serum albumin. J Pharm Biomed Anal. 2010;52:384–90.
Zhang HM, Chen TT, Zhou QH, Wang YQ. Binding of caffeine, theophylline, and theobromine with human serum albumin: a spectroscopic study. J Mol Struct. 2009;938:221–2.
Li X, Wang C, Li J, Wang Z. Microcalorimetric studies on the interactions of lanthanide ions with bovine serum albumin. J Therm Anal Calorim. 2007;89:899–900.
Banerjee T, Singh SK, Kishore NJ. Binding of naproxen and amitriptyline to bovine serum albumin: biophysical aspects. J Phys Chem B. 2006;110:24147–56.
Kun R, Szekeres M, Dékány I. Isothermal titration calorimetric studies of the pH induced conformational changes of bovine serum albumin. J Therm Anal Calorim. 2009;96:1009–17.
Mithu B, Asim P, Gopa M, Avadhesha S, Takashi BB. Sulfonamide drugs binding to the colchicine site of tubulin: thermodynamic analysis of the drug-tubulin interactions by isothermal titration calorimetry. J Med Chem. 2005;48:547–55.
Goldberg RN, Kishore N, Lennen RM. CRC handbook of chemistry and physics. 83rd ed., Lide DR, editor. Boca Raton: CRC Press; 2002.
Ross PD, Subramanian S. Thermodynamics of protein association reactions: forces contributing to stability. Biochemistry. 1981;20:3096–102.
He WY, Li Y, Xue CX, Hu ZD, Chen XG, Sheng FL. Bioorg Med Chem. 2005;13:1837–45.
Xi JQ, Guo R. Acid-base equbilium of puerarin in CTAB micelles. J Pharm Biomed Anal. 2007;43:111–8.
Wang YL, Wang HF. Interaction of bovine albumin with benzoate. Universitatis Pekinensia Scientiarum Naturalium. 2002;38:159–63.
Witold KS, Henry HM, Dennis C. Determination of protein secondary structure by Fourier transform infrared spectroscopy: a critical assessment. Biochemistry. 1993;32:389–94.
Stryer L. Fluorescence energy transfer as a spectroscopic ruler. Annu Rev Biochem. 1978;47:819–46.
Cui FL, Fan J, Li JP, Hu Z. Interactions between 1-benzoyl-4-p-chlorophenyl thiosemicarbazide and serum albumin: investigation by fluorescence spectroscopy. Bioorg Med Chem. 2004;12:151–7.
Hu YJ, Liu Y, Pi ZB, Qu SS. Interaction of cromolyn sodium with human serum albumin: a fluorescence quenching study. Bioorg Med Chem. 2005;13:6609–14.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xi, J., Fan, L. Study on the binding of puerarin to bovine serum albumin by isothermal titration calorimetry and spectroscopic approaches. J Therm Anal Calorim 102, 217–223 (2010). https://doi.org/10.1007/s10973-010-0792-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-010-0792-y