Abstract
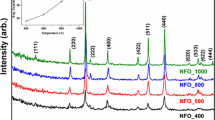
Magnetic nanoparticles were prepared by a wet chemical method. Precursors of MFe2O4 (M = Co, Mn, Zn) were prepared from a mixture of metal chloride and metasilicate nonahydrate aqueous solutions. The precipitates obtained in the wet chemical method were calcined to obtain MFe2O4 nanoparticles encapsulated by amorphous SiO2. The blocking temperatures T B’s were between 20 and 320 K, in this temperature range, the anisotropy energy of the particles decreased below their thermal energy. T B increased with the particle size. In order to clarify the nanoparticle formation process, differential thermal analysis and thermogravimetric (TG-DTA) measurements were performed for the as-prepared samples.





Similar content being viewed by others
References
Gorter EW. Saturation magnetization and crystal chemistry of ferromagnetic oxides (1. The spinel structure). Philips Res Rep. 1954;9:295–320.
Ichiyanagi Y, Uehashi T, Yamada S. Magnetic properties of Ni-Zn ferrite nanoparticles. Phys Stat Sol (c). 2004;1:3485–8.
Ichiyanagi Y, Yamada S. The size-dependent magnetic properties of Co3O4 nanoparticles. Polyhedron. 2005;24:2813–6.
Ichiyanagi Y, Kubota M. Magnetic properties of Mg-ferrite nanoparticles. J Magn Magn Mater. 2007;310:2378–80.
Ichiyanagi Y, Kimishima Y. Structural, magnetic and thermal characterization of Fe2O3 nanoparticles. J Therm Anal Calorim. 2002;69:919–23.
Ichiyanagi Y, Uehashi T. Thermal fluctuation and magnetization of Ni-Zn ferrite nanoparticles by particle size. J Therm Anal Calorim. 2005;81:541–4.
Kubota M, Kanazawa Y, Nasu K, Moritake S, Kawaji H, Atake T, et al. Effect of heat treatment on magnetic MgFe2O4 nanoparticles. J Therm Anal Calorim. 2008;92:461–3.
Ichiyanagi Y, Moritake S, Taira S, Setou M. Functional magnetic nanoparticles for medical application. J Magn Magn Mater. 2007;310:2877–9.
Moritake S, Taira S, Ichiyanagi Y, Morone N, Song SY, Hatanaka T, et al. Functionalized ultranano magnetic particles for an in vivo delivery system. J Nanosci Nanotech. 2007;7:937–44.
Satya Murthy NS, Natera MG, Begam RJ, Youssef SI. Ferrites, Porc Int Conf. 1971;60.
Pekeris CL. Ground state of two-electron atoms. Phys Rev. 1958;112:1649–58.
Pekeris CL. 1 1S, 2 1S, and 2 3S States of H- and of He. Phys Rev. 1962;126:1470–6.
Smit J, Wijn HPJ. Ferrite, Philips Tech Lib. 1959;278–83.
Jeyadevan B, Tohji K, Nakatuska K, Narayanasamy A. Irregular distribution of metal ions in ferrites prepared by co-precipitation technique structure analysis of Mn–Zn ferrite using extended X-ray absorption fine structure. J Magn Magn Mater. 2000;217:99–105.
Kittel C. Theory of the structure of ferromagnetic domains in films and small particles. Phys Rev. 1946;70:965–71.
Néel C. Compt Rend. 1947;224:1488.
Stoner EC, Wohlfarth EP. Interpretation of high coercivity in ferromagnetic materials. Nature. 1947;160:650.
Dunlop DJ. Philos Mag Ser. 1969;19:329.
Congiu F, Concas G, Ennas G, Falqui A, Fiorani D, Marongiu G, et al. Magnetic properties of nanocrystalline CoFe2O4 dispersed in amorphous silica. J Magn Magn Mater. 2004;272–276:1561–2.
Acknowledgements
This study was partially supported by Precursory Research for Embryonic Science and Technology of the Japan Science and Technology Agency, and a Promotion of Science Grant-in Aid for Scientific Research (No. 1851009100). I would like to thank Prof. T. Atake and Prof. H. Kawaji at the Tokyo Institute of Technology for the TG-DTA measurement and useful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ichiyanagi, Y., Moro, Y., Katayanagi, H. et al. Magnetic and thermal analysis of MFe2O4 (M = Co, Mn, Zn) nanoparticles. J Therm Anal Calorim 99, 83–86 (2010). https://doi.org/10.1007/s10973-009-0564-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-009-0564-8