Abstract
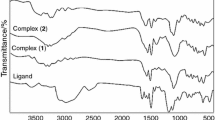
Several mononuclear Co(II), Ni(II), Cu(II), and Fe(II) complexes of tetradentate salpren-type diimine, obtained from 3,5-di-tert-butyl-2-hydroxybenzaldehyde and 1,3-diaminopropane have been prepared and characterized by analytical, spectroscopic (FT-IR, UV–VIS) techniques, magnetic susceptibility measurements and thermogravimetric analyses (TG). The thermodynamic and thermal properties of complexes have been investigated. For further characterization Direct Insertion Probe-Mass Spectrometry (DIP-MS) was used and the fragmentation pattern and also stability of the ions were evaluated. The characterization of the end products of the decomposition was achieved by X-ray diffraction. The thermal stabilities of metal complexes of N,N′-bis(3,5-di-t-butylsalicylidene)-1,3-propanediamine ligand (L) were found as Ni(II) > Cu(II) > Co(II) > Fe(II).





Similar content being viewed by others
References
Sheldon RA, Kochi JR. Metal catalyzed oxidation of organic compounds. New York: Academic Press; 1981.
Kaim W. The transition metal coordination chemistry of anion radicals. Coord Chem Rev. 1987;76:187–235.
Halfen JA, Toung VG, Tolman WB. Modeling of the chemistry of the active site of galactose oxidase. Angew Chem Int Ed. 1996;35(15):1687–90.
Zhang W, Loebach JL, Wilson SR, Jacobsen EN. Enantioselective epoxidation of unfunctionalized olefins catalyzed by salen manganese complexes. J Am Chem Soc. 1990;112(7):2801–3.
Kumar DN, Garg BS. Some new cobalt(II) complexes: synthesis, characterization and thermal studies. J Therm Anal Calorim. 2002;69:607–16.
Wang HD, Li YT, Ma PH, Zeng XC. Studies on the thermal decomposition of N,N′-ethylenebis(salicylideneiminato) diaquochromium(III) nitrate. J Therm Anal Calorim. 2002;69(2):575–81.
Doğan F, Gülcemal S, Yürekli M, Çetinkaya B. Thermal analysis study of imidazolinium and some benzimid azolium salts by TG. J Therm Anal Calorim. 2008;91(2):395–400.
Doğan F, Dayan O, Yürekli M, Çetinkaya B. Thermal study of ruthenium(II) complexes containing pyridine-2,6-diimines. J Therm Anal Calorim. 2008;91(3):943–9.
Kasumov VT, Yaman OS, Tas E. Synthesis, spectroscopy and electrochemical behaviors of nickel(II) complexes with tetradentate shiff bases derived from 3,5-But2-salicylaldehyde. Spectrochim Acta A. 2005;62(1–3):716–20.
Coats AW, Redfern JP. Kinetic parameters from thermogravimetric data. Nature. 1964;201:68–9.
van Krevelen DW, van Heerden C, Huntjons FJ. Kinetic study by thermogravimetry. Fuel. 1951;30:253–8.
MacCallum JR, Tanner J. Derivation of rate equations used in thermogravimetry. Nature. 1970;225(5238):1127–8.
Madhusudanan PM, Krishnan K, Ninan KN. New equations for kinetic analysis of non-isothermal reactions. Thermochim Acta. 1993;221:13–21.
Horowitz HH, Metzger G. New analysis of thermogravimetric traces. Anal Chem. 1963;35:1464–8.
Larrow JF, Jacobsen EN, Gao Y, Hong Y, Nie X, Zepp CM. A practical method for the large-scale preparation of [N,N′-Bis(3,5-di-tertbutylsalicylidene)-1,2-cyclohexanediaminato (2-)]manganese(III) chloride, a highly enantioselective epoxidation catalyst. J Org Chem. 1994;59(7):1939–42.
Earnshaw A. Introduction to magnetochemistry. London: Academic Press; 1968.
Tas E, Aslanoglu M, Guler M, Ulusoy M. Synthesis, spectral characterization and electrochemical studies of copper(II) and cobalt(II) complexes with novel tetradentate salicylaldimines. J Coord Chem. 2004;57(8):583–9.
Temel H, Sekerci M. Novel complexes of manganese(III), cobalt(II), copper(II), and zinc(II) with Schiff base derived from 1,2-bis(p-aminophenoxy)ethane and salicylaldehyde. Synth React Inorg Met-Org Chem. 2001;31(5):849–57.
Sacconi L, Ciampolini M, Maggio F, Cavasino FP. Studies in coordination chemistry. IX.1 Investigation of the stereochemistry of some complex compounds of cobalt(II) with N-substituted salicylaldimines. J Am Chem Soc. 1962;84(17):3246–8.
Mukherjee RN, Abrahamson AJ, Patterson GS, Stack TDP, Holm RH. A new class of (N,N′-bis(salicylideneamino)ethanato)iron(II) complexes-5-coordinate [FeII(salen)L]- preparation, properties, and mechanism of electron-transfer reactions. Inorg Chem. 1988;27(12):2137–44.
Straszko J, Humienik MO, Mozejko J. Study of the mechanism and kinetic parameters of the thermal decomposition of cobalt sulphate hexahydrate. J Therm Anal Calorim. 2000;59(3):935–42.
Sodhi GS. Correlation of thermal-stability with structures for some metal-complexes. Thermochim Acta. 1987;120:107–14.
Nagase K, Sato K, Tanaka N. Thermal dehydration and decomposition reactions of bivalent metal oxalates in the solid state. Bull Chem Soc Jpn. 1975;48(2):439–42.
Acknowledgements
The authors gratefully acknowledge Basri Gülbakan and Ömür Çelikbıçak for helpful Mass Spectrometry measurements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Doğan, F., Ulusoy, M., Öztürk, Ö.F. et al. Synthesis, characterization and thermal study of some tetradentate Schiff base transition metal complexes. J Therm Anal Calorim 98, 785–792 (2009). https://doi.org/10.1007/s10973-009-0205-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-009-0205-2