Abstract
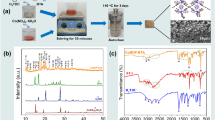
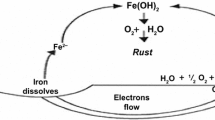
Zn pack coating formation takes place in three steps as differential scanning calorimetry shows. The initial step (at 193.9°C) is endothermic and involves the transformation of α-NH4Cl to β-NH4Cl and the NH4Cl decomposition to NH3 and HCl. During the second step (at 248.6°C), which is exothermic, Zn2+ salts are formed and most probably ZnCl2. Finally at 264.1°C (endothermic reaction) it seems that ZnCl2 is decomposed to form Zn that is deposited on the ferrous substrate. The as-cast Zn diffuses in the iron substrate forming the gamma and delta phase of the Fe–Zn phase diagram. Al2O3 is not involved in the above-mentioned mechanism and acts only as filler.
Similar content being viewed by others
References
AR Marder (2000) Prog. Mater. Sci. 45 191 Occurrence Handle10.1016/S0079-6425(98)00006-1 Occurrence Handle1:CAS:528:DC%2BD3cXisVaqsLY%3D
J. W. Cambel in ‘Hot-dip coatings’, Vol. Corrosion, ASM, 2000, pp. 436–445.
InstitutionalAuthorNameGalvanizers Association et al. (2000) The engineers and architects’ guide to hot dip galvanizing Galvanizers Association Sutton Coldfield
P Maass P Peissker et al. (1993) Handbuch Feuerverzinken Wiley-VCH Weinheim
G. W. Goward and L. L. Seigle, in ASM Handbook, Vol. Surface Treatments, ASM, 1999, pp. 611–617.
Y He D Li D Wang Z Zhang H Qi W Gao (2002) Mater. Lett. 56 554 Occurrence Handle1:CAS:528:DC%2BD38XnvVyltLg%3D
T Zmijewski B Pacewska (1997) J. Thermal Anal. 49 1187 Occurrence Handle10.1007/BF01983674 Occurrence Handle1:CAS:528:DyaK2sXkvVeksr4%3D
NRE Radwan M Mokhtar GA El-Shobaky (2003) J. Therm. Anal. Cal. 71 977 Occurrence Handle10.1023/A:1023303032138 Occurrence Handle1:CAS:528:DC%2BD3sXjvVylu7Y%3D
V Petkova Y Pelovski V Hristova (2005) J. Therm. Anal. Cal. 82 813 Occurrence Handle10.1007/s10973-005-0968-z Occurrence Handle1:CAS:528:DC%2BD28XjsFGgtQ%3D%3D
V Alexeev et al. (1975) Analyse Qualitative Editions MIR Moscou
M Olszak-Humienik (2001) Thermochim. Acta 378 107 Occurrence Handle10.1016/S0040-6031(01)00585-8 Occurrence Handle1:CAS:528:DC%2BD3MXnsV2rt7c%3D
N Pistofidis G Vourlias D Chaliampalias K Chryssafis G Stergioudis EK Polychroniadis (2006) J. Alloys Compd. 407 221 Occurrence Handle10.1016/j.jallcom.2005.06.039 Occurrence Handle1:CAS:528:DC%2BD2MXhtlCrsrfI
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pistofidis, N., Vourlias, G., Chaliampalias, D. et al. DSC study of the deposition reactions of zinc pack coatings up to 550°C. J Therm Anal Calorim 84, 191–194 (2006). https://doi.org/10.1007/s10973-005-7196-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-005-7196-4