Summary
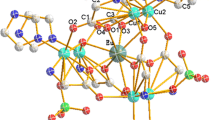
New 4f-trivalent lanthanide 2-pyrazinecarboxylate hydrates of composition Ln(pyzCOO)<Subscript>3</Subscript>·3.5H<Subscript>2</Subscript>O, where<Emphasis Type=”Italic”> Ln</Emphasis>=La, Ce, Pr, Nd, Sm and Dy have been isolated from aqueous solution containing the respective metal nitrates and 2-pyrazinecarboxylic acid (HpyzCOO), in 1:3 molar ratios. The complexes have been characterized by analytical, electronic and IR spectroscopic, thermal analysis and X-ray diffraction studies. The OCO<Superscript>-</Superscript> group of 2-pyrazinecarboxylate bridges in both mono and bidentate fashion (&ngr;<Subscript>as(COO)</Subscript>=1612 and 1578 cm<Superscript>-1</Superscript>; &ngr;<Subscript>s(COO)</Subscript>=1423 and 1388 cm<Superscript>-1</Superscript>) as evidenced by IR data. The IR spectra further indicate that coordination takes place through the carboxyl group and the nearest hetero-ring nitrogen atom. The bonding parameters &bgr;, <Emphasis Type=”Italic”>b</Emphasis><Superscript>1/2</Superscript>, %&dgr; and &eegr; have been calculated from the electronic spectral (hypersensitive) bands of Pr(III) and Nd(III) complexes. Both IR and thermal data reveal the presence of a coordinated water and two and a half lattice water molecules in the complexes. It is further substantiated by single crystal X-ray study for the corresponding lanthanum compound. Based on the results obtained, nine coordination is proposed for the lanthanide ions. Simultaneous TG-DTA of the complexes show that they are thermally stable up to 150°C and undergo endothermic (~155 and 250°C) followed by exothermic (~480°C) decompositions to give the respective metal oxides as the final product. The compounds are found to be isostructural as evidenced from X-ray powder patterns.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Govindarajan, S. Thermoanalytical and spectral properties of new rare-earth metal 2-pyrazinecarboxylate hydrates. J Therm Anal Calorim 79, 685–689 (2005). https://doi.org/10.1007/s10973-005-0596-7
Issue Date:
DOI: https://doi.org/10.1007/s10973-005-0596-7