Abstract
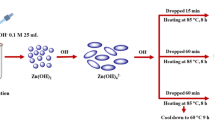
Benzamide is successfully degraded on the novel heterosystem NiMn2O4/TiO2 under visible light. The nanosized spinel is synthesized by the sol–gel method at ~850 °C. The X-ray diffraction pattern shows narrow peaks and the oxides are well crystallized. The Mott–Schottky plot (C−2−E) of NiMn2O4 is characteristic of p-type conductivity from which a flat-band potential of −0.20 VSCE is obtained. The energy-band diagram, built from the physicochemical characterizations, predicts the electron transfer from the conduction to dissolved oxygen via TiO2. The loading of TiO2 with NiMn2O4 enhances the photoactivity and NiMn2O4 islands achieve a colloidal photochemical heterosystem, tested successfully for the light-induced benzamide degradation. The spinel dose and benzamide concentration are optimized. Under the ideal conditions, the rate of the benzamide disappearance is controlled by high-performance liquid chromatography. A conversion of 85% is reported in aerated benzamide solution (15 ppm) in less than 2 h under artificial light. This conversion rate increases up to 94% under solar light and the oxidation obeys to a first-order kinetics with a half-life of 53 min.

Photodegradation of benzamide on the heterosystem NiMn2O4/TiO2 under visible light.









Similar content being viewed by others
References
Zhang D, Yin Y, Li Y, Cai Y, Liu J (2017) Critical role of natural organic matter in photodegradation of methylmercury in water: molecular weight and interactive effects with other environmental factors. Sci Total Environ 578:535–541
Mahmiani Y, Sevim AM, Gül A (2016) Photocatalytic degradation of 4-chlorophenol under visible light by using TiO2 catalysts impregnated with Co(II) and Zn(II) phthalocyanine derivatives. J Photochem Photobiol A Chem 321:24–32
Brahimi R, Bessekhouad Y, Bouguelia A, Trari M (2008) Improvement of eosin visible light degradation using PbS-sensititized TiO2. J Photochem Photobiol A Chem 194:173–180
Bessekhouada Y, Brahimi R, Hamdini F, Trari M (2012) Cu2S/TiO2 heterojunction applied to visible light Orange II degradation. J Photochem Photobiol A Chem 248:15–23
Tahiri H, Ichou YA, Herrmann JM (1998) Photocatalytic degradation of chlorobenzoic isomers in aqueous suspensions of neat and modified titania. J Photochem Photobiol A Chem 114:219–226
Belaissaa Y, Niboua D, Assadi A, Bellal B, Trari M (2016) New hetero-junction p-CuO/n-ZnO for the removal of amoxicillin by photocatalysis under solar irradiation. J Taiwan Inst Chem Eng 68:254–265
Lelarioa F, Brienzaa M, Bufoa SA, Scranob L (2016) Effectiveness of different advanced oxidation processes (AOPs) on the abatement of the model compound mepanipyrim in water. J Photochem Photobiol A Chem 321:187–201
Molinari R, Lavorato C, Argurio P (2017) Recent progress of photocatalytic membrane reactors in water treatment and in synthesis of organic compounds: a review. Catal Today 281:14–164
Ali I, Kim SR, Kim SP, Kim JO (2017) Anodization of bismuth doped TiO2 nanotubes composite for photocatalytic degradation of phenol in visible light. Catal Today 282:31–37
Bassaid S, Chaib M, Omeiri S, Bouguelia A, Trar M (2009) Photocatalytic reduction of cadmium over CuFeO2 synthesized by sol-gel. J Photochem Photobiol A Chem 201:62–68
Li H, Liu Y, Tang J, Deng Y (2016) Synthesis, characterization and photocatalytic properties of Mg1-xZnxAl2O4 spinel nanoparticles. Solid State Sci 58:14–21
Dermèche L, Rabia C, Rekhila G, Trari M (2017) Preparation and characterization of mixed caesium-tin mixed salt of Keggin-type phosphovanadomolybdate. Application to photocatalytic chromate reduction. Sol Energy Mater Sol Cells 168:45–50
Bouchaaba H, Bellal B, Maachi R, Trari M, Nasrallaha N, Mellah A (2016) Optimization of physico-chemical parameters for the photo-oxidation of neutral red on the spinel Co2SnO4. J Taiwan Inst Chem Eng 58:310–317
Chen J, Shu J, Anqi Z, Juyuan H, Yan Z, Chen J (2016) Synthesis of carbon quantum dots/TiO2 nanocomposite for photo-degradation of Rhodamine B and cefradine. Diamond Relat Mater 70:137–144
Bagtache R, Abdmeziem K, Rekhila G, Trari M (2016) Synthesis and semiconducting properties of Na2MnPO4F. Application to degradation of Rhodamine B under UV-light. Mater Sci Semicond Process 51:1–7
Fedailaine M, Berkani S, Trari M (2016) Ni2+ reduction under solar irradiation over CuFe2O4/TiO2 catalysts. J Chem Eng 33:2027–2033
Cherifi K, Allalou N, Rekhila G, Trari M, Bessekhouad Y (2015) Nitrate-processing and characterization of a cobalt-doped barium tin oxide perovskite: magnetic, transport and photoelectrochemical properties. Mater Sci Semicond Process 30:571–577
Dong H, Li Z, Xu X, Ding Z, Wu L, Wang X, Fu X (2009) Visible light-induced photocatalytic activity of delafossite AgMO2 (M=Al, Ga, In) prepared via a hydrothermal method. Appl Catal B Environ 89:551–556
Moualkia H, Rekhila G, Izerrouken M, Mahdjoub A, Trari M (2014) Influence of the film thickness on the photovoltaic properties of chemically deposited CdS thin films: application to the photodegradation of orange II. Mater Sci Semicond Process 21:186–193
Rekhila G, Bessekhouad Y, Trari M (2015) Hydrogen evolution under visible light over the solid solution NiFe2-xMnxO4 prepared by sol gel. Int J Hydrog Energy 40:12611–12618
Benreguia N, Barnabé A, Trari M (2015) Sol-gel synthesis and characterization of the delafossite CuAlO2. J Sol Gel Sci Technol 75:670–679
Brahimi R, Bessekhouad Y, Trari M (2012) Physical properties of NxTiO2 prepared by sol-gel route. Phys B 407:3897–3904
Rekhila G, Bessekhouad Y, Trari M (2016) Synthesis and characterization of the spinel ZnFe2O4, application to the chromate reduction under visible light. Environ Technol Innova 5:127–135
Gómez-Solís C, Peralta-Arriaga SL, Torres-Martínez LM, Juárez-Ramírez I, Díaz-Torres LA (2017) Photocatalytic activity of MAl2O4 (M=Mg, Sr and Ba) for hydrogen production. Fuel 188:197–204
Gurunathan K, Baeg JO, Lee SM, Subramanian E, Moon SJ, Kong KJ (2008) Visible light active pristine and Fe3+ doped CuGa2O4 spinel photocatalysts for solar hydrogen production Inter. J Hydrog Energy 33:2646–2652
Rekhila G, Bessekhouad Y, Trari M (2013) Visible light hydrogen production on the novel ferrite NiFe2O4. Int J Hydrog Energy 38:6335–6343
Pleskov YV (1994) Semiconductor photoelectrochemistry for cleaner environment: utilization of solar energy. Environ Orien Electrochem 59:417–443
Amara M, Kerdjoudj H, Bouguelia A, Trari M (2008) A combination between membrane selectivity and photoelectrochemistry to the separation of copper, zinc and nickel in aqueous solutions. J Membr Sci 312:125–131
Lahmar H, Rekhila G, Trari M, Bessekhouad Y (2015) HCrO4 - reduction on the novel heterosystem La2CuO4/SnO2 under solar light. Environ Prog Sustain Energy 34:744–750
Belaissaa Y, Niboua D, Assadi AA, Bellal B, Trari M (2016) A new hetero-junction p-CuO/n-ZnO for the removal of amoxicillin by photocatalysis under solar irradiation. J Taiwan Inst Chem Eng 68:254–265
Helaili N, Bessekhouad Y, Bouguelia A, Trari M (2010) p-Cu2O/n-ZnO heterojunction applied to visible light Orange II degradation. Sol Energy 84:1187–1192
Arlos MJ, Fraile MMH, Liang R, Bragg LM, Zhou NY, Andrews SA, Servo MR (2016) Photocatalytic decomposition of organic micropollutants using immobilized TiO2 having different isoelectric points. Water Res 101:351–361
Chhor K, Bocquet JF, Colbeau-Justi C (2004) Comparative studies of phenol and salicylic acid photocatalytic degradation: influence of adsorbed oxygen. Mater Chem Phys 86:123–131
Acknowledgements
The authors would like to express their gratitude to the Faculty of Chemistry for financial support of this research. They are grateful to N Taibi for the TEM analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Highlights
-
The benzamide was photodegraded on the hetero-system NiMn2O4/TiO2 synthesized by sol gel.
-
The direct optical transition (1.75 eV) makes the spinel NiMn2O4 attractive for the light energy conversion.
-
The benzamide elimination, controlled by HPLC, follows a first order kinetic with a rate half-life of 53 min.
-
The improved photocatalytic performance is due to the electrons transfer NiMn2O4/TiO2.
Rights and permissions
About this article
Cite this article
Rekhila, G., Gabes, Y., Brahimi, R. et al. Preparation and characterization of the system NiMn2O4/TiO2 by sol–gel: application to the photodegradation of benzamide under visible light. J Sol-Gel Sci Technol 85, 677–683 (2018). https://doi.org/10.1007/s10971-018-4598-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-018-4598-x