Abstract
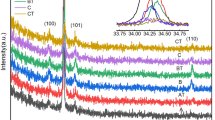
Vanadium dioxide (VO2) thin films were fabricated on single crystal Si (100) substrates by sol–gel method, including a process of annealing a vanadium pentoxide (V2O5) gel precursor at different temperatures. The crystalline structure and morphology of the films were investigated by XRD, FE-SEM and AFM, indicating that the films underwent the grain growth, agglomeration and grain refinement process with increased annealing temperatures. The film annealed at 500 °C exhibits the formation of VO2 phase with a strong (011) preferred orientation and high crystallinity, the surface of the film is uniform and compact with a grain size of about 120 nm. Meanwhile, the film exhibits excellent phase transition properties, with a decrease of transmittance from 35.5 to 2.5% at λ = 25 μm and more than 3 orders of resistivity magnitude variation bellow and above the phase transition temperature. The phase transition temperature is evaluated at 60.4 °C in the heating transition and 55.8 °C in the cooling transition. Furthermore, the phase transition property of the VO2 film appears to be able to remain stable over repetitive cycles 100 times.










Similar content being viewed by others
References
Manning TD, Parkin IP, Pemble ME, Sheel D, Vernardou D (2006) Intelligent window coatings: atmospheric pressure chemical vapor deposition of tungsten-doped vanadium dioxide. Chem Mater 16:744–749
Muraoka Y, Ueda Y, Hiroi Z (2002) Large modification of the metal-insulator transition temperature in strained VO2 films grown on TiO2 substrates. J Phys Chem Solids 63:965–967
Lappalainen J, Heinilehto S, Jantunen H, Lantto V (2008) Electrical and optical properties of metal-insulator-transition VO2 thin films. J Electroceram 22(1–3):73–77
Morin FJ (1959) Oxides which show a metal-insulator transition at the neel temperature. Phys Rev Lett 3:34–36
Kim HT, Chae BG, Youn DH, Kim G, Kang KY, Lee SJ, Kim K, Lim YS (2005) Raman study of electric-field-induced first-order metal-insulator transition in VO2-based devices. Appl Phys Lett 86:242101
Cavalleri A, Tóth Cs, Siders CW, Squier JA et al (2001) Femtosecond structure dynamics in VO2 during an ultrafast solid–solid phase transition. Phys Rev Lett 87:237401
Cao J, Ertekin E, Srinivasan V, Fan W, Huang S et al (2009) Strain engineering and one-dimensional organization of metal-insulator domains in single-crystal vanadium dioxide beams. Nat Nanotechnol 4:732–737
Manning TD, Parkin IP (2004) Atmospheric pressure chemical vapour deposition of tungsten doped vanadium (IV) oxide from VOCl3, water and WCl6. J Mater Chem 14:2554–2559
Jerominek H, Picard F, Vincent D (1993) Vanadium oxide films for optical switching and detection. Opt Eng 32:2092–2099
Huang WX, Yin XG, Huang CP, Wang QJ, Miao TF, Zhu YY (2010) Optical switching of a metamaterial by temperature controlling. Appl Phys Lett 96:261908
Messaoud TB, Landry G, Gariépy JP, Ramamoorthy B, Ashrit PV, Haché A (2008) High contrast optical switching in vanadium dioxide thin films. Opt Commun 281:6024–6027
Driscoll T, Palit S, Qazilbash MM, Brehm M, Keilmann F et al (2008) Dynamic tuning of an infrared hybrid-metamaterial resonance using vanadium dioxide. Appl phys lett 93:024101
Driscoll T, Kim HT, Chae BG, Kim BJ, Lee YW et al (2009) Memory metamaterials. Science 325:1518–1521
Dai JM, Zhang JQ, Zhang WL, Grischkowsky D (2004) Terahertz time-domain spectroscopy characterization of the far-infrared absorption and index of refraction of high-resistivity, float-zone silicon. J Opt Soc Am B 21:1379–1386
Jeon TI, Grischkowsky D (1997) Nature of conduction in doped silicon. Phys Rev Lett 78:1106–1109
Partlow DP, Gurkovich SR, Radford KC, Denes LJ (1991) Switchable vanadium oxide films by a sol-gel process. J Appl Phys 70:443–452
Ozer N (1997) Electrochemical properties of sol-gel deposited vanadium pentoxide films. Thin Solid Films 305:80–87
Yuan NY, Li JH, Lin CL (2002) Valence reduction process from sol-gel V2O5 to VO2 thin films. Appl Surf Sci 191:176–180
Guhathakurta S, Subramanian A (2007) Effect of hydrofluoric acid in oxidizing acid mixtures on the hydroxylation of silicon surface. J Electrochem Soc 154:136–147
Yan JZ, Zhang Y, Huang WX, Tu MJ (2008) Effect of Mo-W Co-doping on semiconductor-metal phase transition temperature of vanadium dioxide film. Thin Solid Films 516:8554–8558
Nag J, Hanlund RF Jr (2008) Synthesis of vanadium dioxide thin films and nanoparticles. J Phys-Condens Mat 20:264016
Livage J, Guzman G, Beteille F, Dacadson P (1997) Optical properties of sol-gel derived vanadium oxide films. J Sol-Gel Sci Technol 8:857–865
Bhushan B, Tokachichu DR, Keener MT, Lee SC (2005) Morphology and adhesion of biomolecules on silicon based surfaces. Acta Biomater 1:327–341
Chan BG, Kim HT, Yun SJ, Kim BJ et al (2007) Comparative analysis of VO2 thin films prepared on sapphire and SiO2 substrates by the sol-gel process. Jpn J Appl Phys 46:738–743
Yan JZ, Huang WX, Zhang Y, Liu XJ, Tu MJ (2008) Characterization of preferred orientated vanadium dioxide film on muscovite (001) substrate. Phys Stat Sol 205:2409–2412
Jepsen PU, Fischer BM, Thoman A et al (2006) Metal-insulator phase transition in a VO2 thin film observed with terahertz spectroscopy. Phys Rev B 74:205103
Yang TH, Aggarwal R, Gupta A et al (2010) Semiconductor-metal transition characteristics of VO2 thin films grown on c- and r-sapphire substrates. J Appl Phys 107:053514
Youn DH, Kim HT, Chae BG, Hwang YJ, Lee JW, Maeng SL, Kang KY (2004) Phase and structural characterization of vanadium oxide films grown on amorphous SiO2/Si substrates. J Vac Sci Technol A 22:719–724
Acknowledgments
This work was financially supported by the National Science Foundation of China (Grant Nos. 61072036) and the Science and Technology Supporting Programs Fund Project of Sichuan province (2009SZ0199). We would also thank Analytical and Testing center of Sichuan University for their XRD analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shi, Q., Huang, W., Yan, J. et al. Preparation and phase transition characterization of VO2 thin film on single crystal Si (100) substrate by sol–gel process. J Sol-Gel Sci Technol 59, 591–597 (2011). https://doi.org/10.1007/s10971-011-2533-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-011-2533-5