Abstract
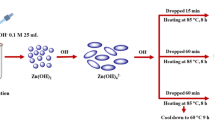
A novel plasmonic photocatalyst, i.e., acid-etched TiO2 nanobelts attached with Ag/AgI nanoparticles (NPs) was prepared by deposition–precipitation-photoreduction method. Such surface-modified nanobelts had larger area than the normal one. Ag NPs were formed from AgI by photo-reduction under Xenon lamp irradiation. X-ray diffraction, scanning electron microscopy analysis, UV–Vis diffuse reflectance spectra and fluorescence spectra were used to characterize the structure and optical properties of the sample. The obtained sample exhibited strong photodegradation of methyl orange (MO) under visible light irradiation, which were attributed to both the surface plasmon resonance of Ag NPs and the visible light actived AgI. The photodegradation was accomplished by the transfer of photoexcited electrons from the Ag NPs to the acid-etched TiO2 nanobelts. After four cycles of photodegradation the photocatalyst was still stable. This novel photocatalyst had a high potential application in wastewater-treatment and biomedical engineering.







Similar content being viewed by others
References
Cheng B, Le Y, Yu JG (2010) J Hazard Mater 177:971–977
Ksibi M, Rossignol S, Tatibouet JM, Trapalis C (2008) Mater Lett 62:4204–4206
Li C, Wang C, Li Q, Yang S, Hou L, Chen S (2009) J Mater Sci 44:3413
Asahi R, Morikawa T, Ohwaki T, Aoki K, Taga Y (2001) Science 293:269–271d
Halasi G, Kecskemeti A, Solymosi F (2010) Catal Lett 135:17
Fessenden RW, Kamat PV (1995) J Phys Chem 99:12902–12906
Armstrong G, Armstrong AR, Canales J, Bruce PG (2005) Chem Commun 24:54
Wang P, Huang BB, Qin XY, Zhang XY, Dai Y, Wei JY, Whangbo MH (2008) Angew Chem Int Ed 47:7931–7933
Schwartzberg AM, Zhang JZ (2008) J Phy Chem C 112:10323–10337
Koichi A, Makoto F, Carsten R, Junji T, Hirotaka M, Yoshimichi O, Naoya Y, Toshiya W (2008) J Am Chem Soc 130:1676–1680
Choi M, Shin KH, Jang J (2010) J Colloid Interface Sci 341:85
Zhou XF, Hu C, Hu XX, Peng TW, Qu JH (2010) J Phys Chem C 114:2746–2750
Hu C, Hu X, Wang L, Qu J, Wang A (2006) Environ Sci Technol 40:7903–7907
Rodrigues S, Uma S, Martyanov IN, Klabunde KJ (2005) J Catal 233:405–410
Zhou WJ, Liu H, Wang JY, Liu D, Du GJ, Cui JJ (2010) Appl Mater Interfaces 2:2385–2392
Plass R, Pelet S, Krueger J, Gratzel M, Bach U (2002) J Phys Chem B 106:7578–7580
Ishibashi K, Fujishima A, Watanabe T (2000) Electrochem Commun 2:207–210
Li FB, Li XZ (2002) Chemosphere 48:1103–1111
Kawahara K, Suzuki K, Ohko Y, Tatsuma T (2005) Phys Chem Chem Phys 7:3851–3855
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 50872076), the Open Research Fund Program of State Key Laboratory of Crystal materials (Grant No. KF0905), and the Ministry of Education of Shandong Province (Grant No. J09LD23). The authors also thank the Analytical Center of Shandong Institute of Light Industry for the technological support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, E., Liu, S., Lu, Q. et al. Photocatalytic property of surface-modified TiO2 nanobelts under visible light irradiation. J Sol-Gel Sci Technol 58, 705–710 (2011). https://doi.org/10.1007/s10971-011-2448-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-011-2448-1