Abstract
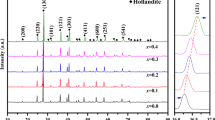
To have a clear cognition on whether clay could be used for disposal of high level radioactive waste efficiently or not, in this work, talc was irradiated by Co-60 γ ray in air at a dose rate of 54 Gy/min with doses up to 1000 kGy. Then, variations in chemical/crystalline structure and intrinsic mechanism were explored. Main results show talc occurred dehydroxylation and lattice shrink. Under 0–1000 kGy irradiation, shrink range in (002) lattice plane was near 1.5% close to 0.14 Å. Main mechanisms involve framework/Mg–OH bond break and H2O radiolysis. Framework break seems more important, Mg–OH bond break and H2O radiolysis are secondary. It seems talc exhibits poor stability to radiation. Clay matrix mightn’t ensure the effectiveness of disposal project, the effect of nuclear accident to environment can be serious.








Similar content being viewed by others
References
Hurtado-Bermudez S, Villa-Alfageme M, Mas JL, Alba MD (2018) Comparison of solvent extraction and extraction chromatography resin techniques for uranium isotopic characterization in high-level radioactive waste and barrier materials. Appl Radiat Isot 137:177–183
Kaufhold S, Dohrmann R (2016) Distinguishing between more and less suitable bentonites for storage of high-level radioactive waste. Clay Min 51(2):289–302
Fujii K, Ochi K, Ohbuchi A, Koike Y (2018) Evaluation of physicochemical properties of radioactive cesium in municipal solid waste incineration fly ash by particle size classification and leaching tests. J Environ Manag 217:157–163
Tashiro Y, Nakao A, Wagai R, Yanai J, Kosaki T (2018) Inhibition of radiocesium adsorption on 2:1 clay minerals under acidic soil environment: effect of organic matter vs. hydroxy aluminum polymer. Geoderma 319:52–60
Yin X, Horiuchi N, Utsunomiya S, Ochiai A, Takahashi H, Inaba Y, Wang X, Ohnuki T, Takeshita K (2018) Effective and efficient desorption of Cs from hydrothermal-treated clay minerals for the decontamination of fukushima radioactive soil. Chem Eng J 333:392–401
Cosenza P, Prêt D, Zamora M (2015) Effect of the local clay distribution on the effective electrical conductivity of clay rocks. J Geophys Res Solid Earth. 120:145–168
Morichon E, Beaufort D, Allard T, Quirt D (2010) Tracing past migrations of uranium in paleoproterozoic basins: new insights from radiation-induced defects in clay minerals. Geology 38:983–986
Zhang M, Hui Q, Lou X, Redfern SAT, Salje EKH, Tarantino SC (2006) Dehydroxylation, proton migration, and structural changes in heated talc: an infrared spectroscopic study. Am Miner 91(5–6):816–825
Loh E (1973) Optical vibrations in sheet silicates. J Phys-Condes Matter 6:1091–1104
Kloprogge JT, Frost RL, Rintoul L (1999) Single crystal raman microscopic study of the asbestos mineral chrysotile. Phys Chem Chem Phys 1:2559–2564
Sidorov TA (2007) Raman spectra and molecular structure of silicates. Russ J Inorg Chem 52:1586–1594
Baita C, Lottici PP, Salvioli-Mariani E, Vandenabeele P, Librenti M, Antonelli F, Bersani D (2014) An integrated raman and petrographic characterization of italian mediaeval artifacts inpietra ollare(Soapstone). J Raman Spectrosc 45:114–122
Rashchenko SV, Likhacheva AY, Goryainov SV, Krylov AS, Litasov KD (2016) In situ spectroscopic study of water intercalation into talc: new features of 10 Å phase formation. Am Miner 101:431–436
Wang A, Freeman JJ, Jolliff BL (2015) Understanding the raman spectral features of phyllosilicates. J Raman Spectrosc 46:829–845
Blaha JJ, Rosasco GJ (1978) Raman microprobe spectra of individual microcrystals and fibers of talc, tremolite, and related silicate minerals. Anal Chem 50:892–896
Sharma SK, Misra AK, Lucey PG, Lentz RC (2009) A combined remote raman and libs instrument for characterizing minerals with 532 nm laser excitation. Spectrochim Acta, Part A 73:468–476
Holtz M, Solin SA, Pinnavaia TJ (1993) Effect of pressure on the raman vibrational modes of layered aluminosilicate compounds. Phys Rev B 48:13312–13317
Rosasco GJ, Blaha JJ (1980) Raman microprobe spectra and vibrational mode assignments of talc. Appl Spectrosc 34:140–144
Chen R, Lv J, Feng J (2014) Characterization of paint by fourier-transform infrared spectroscopy, raman microscopy, and scanning electron microscopy-energy dispersive x-ray spectroscopy. Anal Lett 48:1502–1510
Reynard B, Bezacier L, Caracas R (2015) Serpentines, talc, chlorites, and their high-pressure phase transitions: a raman spectroscopic study. Phys Chem Miner 42:641–649
Wang H, Sun Y, Chu J, Wang X, Zhang M (2020) Crystalline structure variation within phlogopite, muscovite and talc under 0–1000 kGy γ ray irradiation: a clear dependence on intrinsic characteristic. Appl Clay Sci 187:105475
Wang H, Xu L, Li R, Hu J, Wang M, Wu G (2016) Improving the creep resistance and tensile property of UHMWPE sheet by radiation cross-linking and annealing. Radiat Phys Chem 125:41–49
Bosn K, Gangurly J (1994) Thermogravimetric study of the dehydration kinetics of talc. Am Miner 79:692–699
Chon C-M, Lee C-K, Song Y, Kim SA (2006) Structural changes and oxidation of ferroan phlogopite with increasing temperature: in situ neutron powder diffraction and fourier transform infrared spectroscopy. Phys Chem Miner 33:289–299
Tutti F, Lazor P (2008) Temperature-induced phase transition in phlogopite revealed by raman spectroscopy. J Phys Chem Solids 69:2535–2539
Hansen EL, Hemmen H, Fonseca DM, Coutant C, Knudsen KD, Plivelic TS, Bonn D, Fossum JO (2012) Swelling transition of a clay induced by heating. Sci Rep 2:618
Galamboš M, Daňo M, Rosskopfová O, Šeršeň F, Kufčáková J, Adamcová R, Rajec P (2012) Effect of gamma-irradiation on adsorption properties of Slovak bentonites. J Radioanal Nucl Chem 292:481–492
Ho TA, Greathouse JA, Lee AS, Criscenti LJ (2018) Enhanced ion adsorption on mineral nanoparticles. Langmuir 34:5926–5934
Acknowledgements
We are grateful to the Co-60 γ ray source operating team in Institute of Nuclear Physics and Chemistry, China Academy of Engineering Physics (Mianyang, China) for help in γ ray irradiation. This work was partially supported by China Postdoctoral Science Foundation (Grant Nos. 2018M633634XB).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, H., Sun, Y., Chu, J. et al. An intensive exploration on structure transformation of talc under γ-ray irradiation at 0–1000 kGy. J Radioanal Nucl Chem 325, 33–42 (2020). https://doi.org/10.1007/s10967-020-07226-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-020-07226-1