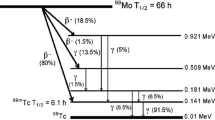
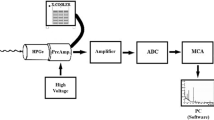
Abstract
N, N-dimethylhydroxylamine (DMHA) is a new salt-free reductant used in spent fuel reprocessing, this paper reports the effect of HNO2 on γ-ray radiolysis and radiolytic products of DMHA in nitric acid medium. DMHA concentration in irradiated samples decreases with the increasing dose and initial HNO2 concentration, and the radiolytic rate of DMHA is lower than 21% at 10 kGy supposed to be the maximum dose absorbed by the reagent during the U/Pu separation and the Pu purification. The main radiolytic products are H2, CH3NHOH, HCHO, and HCOOH, and their quantity increases with the dose and initial HNO2 concentration.





Similar content being viewed by others
References
Shi R (2018) Dream of China commercial spent fuel reprocessing plant. Energy 10:61–63
Tkac P, Mattesson B (2008) Complexation of uranium(VI) with acetohydroxamic acid. J Radioanal Nucl Chem 277:31–36
Natarajan R (2017) Reprocessing of spent nuclear fuel in India: present challenges and future programme. Prog Nucl Energy 101:118–132
Jiang ShJ, Ren FY (1995) Nuclear fuel reprocessing technology. Atomic Energy Publisher, Beijing
Schlea CS, Caverly MR, Henry HE (1963) Uranium (IV) nitrate as a reducing agent for plutonium(IV) in the Purex process. DP-808:1–20
Biddle P, Miles JH (1968) Rate of reaction of nitrous acid with hydrazine and with sulphamic acid. J Inorg Nucl Chem 30:1291–1297
Taylor RJ, Koltunov VS, Valery I (2002) Studies of U(IV) oxidation kinetics in nitric acid and TBP phases. J Nucl Sci Technol 3:355–358
Sood DD, Patil SK (1996) Chemistry of nuclear fuel reprocessing: current status. J Radioanal Nucl Chem 203:547–573
Perrott JR, Stedman G (1977) The kinetics of nitrite scavenging by hydrazine and hydrazoic acids at high acid acidities. J Inorg Nucl Chem 39:325–327
Ochsenfeld W, Petrich G (1983) Neptunium decontamination in a uranium purification cycle of a spent fuel reprocessing plant. Separ Sci Technol 18:1685–1698
Ye GA (2004) Review on the study and application of organic salt-free reagent in Purex process. At Energ Sci Technol 38:152–158
Koltunov VS, Pastushchak VG, Koltunov GV (2006) Kinetics of Pu(IV) reduction with tert-Butylhydrazine. Radiochemistry 48:348–351
Koltunov VS, Marchenko VI, Zhuravleva GI (2001) Kinetics of redox reactions of U, Pu and Np in TBP solutions VII. Kinetices of reduction of Pu(IV) and Np(VI) with butanal oxime in undiluted TBP. Radiochemistry 43:334–337
Taylor RJ, Mason Ch, Cooke R, Boxall C (2002) The reduction of Pu(IV) by formohydroxamic acid in nitric acid. J Nucl Sci Technol 39:278–281
Koltunov VS, Baranov SM, Zharova TP (1995) Organic derivatives of hydrazine and hydroxylamine in future technology of spent nuclear fuel reprocessing. In: International confenrence on evaluation of nuclear fuel cycle systems, France, pp 577–582
Koltunov VS, Baranov SM, Zharova TP (1993) Kinetics of the reactions of Np and Pu ions with hydroxylamine derivatives (II)—reaction of Np(VI) with N, N-dimethylhydroxylamine. Radiokhimiya 35:49–53
Chen YX, Tang HB, Liu JP (2011) The kinetics of the reduction reaction of plutonium(IV) with N, N-dimethylhydroxylamine. J Radioanal Nucl Chem 289:41–47
Liu JP, He H, Tang HB, Chen YX (2011) The application of N, N-dimethylhydroxylamine as reductant for the separation of plutonium from uranium. J Radioanal Nucl Chem 288:351–356
Zhang AY, Xiao GP, Hu JX, He H (2003) Hydroxylamine derivatives in the Purex Process Part VII. The redox reactive kinetics and mechanism of dimethylhydroxylamine and vanadium(V) in nitric medium. J Radioanal Nucl Chem 256:587–592
He H, Ye GA, Tang HB, Zheng WF, Li GL, Lin RSh (2014) An advanced purex process based on salt-free reductants. Radiochim Acta 102:127–133
Li GL, He H (2011) Study on mechanism for oxidation of N, N-dimethylhydroxylamine by nitrous acid. J Radioanal Nucl Chem 287:673–678
Wang JH, Li Q, Wu MH, Xu G, Li Ch, Bao BR, Zheng WF, He H, Zhang ShD (2012) Radiolysis of N, N-dimethylhydroxylamine and its radiolytic liquid organics. J Radioanal Nucl Chem 292:249–254
Wang JH, Li Ch, Wu MH, Xu G, Bao BR, Zheng WF, He H, Zhang ShD (2010) Gaseous products of aqueous N, N-dimethyl hydroxylamine degraded by radiation. Nucl Sci Techniq 21:233–236
Wang JH, Cao XJ, Li Ch, Wu MH, Bao BR, Zheng WF, He H, Zhang ShD (2015) Effect of HNO3 on the γ radiolysis and radiolytic liquid products of N, N-dimethylhydroxylamine. Acta Phys Chim Sin 31:559–565
Li HB, Su Zh, Cong HF, Song FL, Wang XR, He H, Liu ZhY, Lin CSh (2012) α and β irradiation stability of 30% TBP-kerosene-HNO3 systems. J Nucl Radiochem 34:281–285
Benedict M, Pigford TH, Levi HW (1981) Nuclear chemical engineering, 2nd edn. McGraw-Hill Book Company, New York
Choppin GR, Khankhasayev MK (1999) Chemical separation technologies and related methods of nuclear waste management. Kluwer Academic Publishers, Dordrecht
Ren FY (2002) The behavior of the nitrite in purex process. Chin J Nucl Sci Eng 22:356–360
Ren FY, Zhou ZhX (2006) Foreign nuclear fuel reprocessing. Atomic Energy Publisher, Beijing
Wang JH, Wu MH, Bao BR, Li Zh, Zhang XY, Ye GA (2007) Study on hydrogen and carbon monoxide produced by radiation degradation of N, N-dimethylhydroxylamine. J Radioanal Nucl Chem 273:371–373
Wu JL, Qi ShCh (1993) Radiation chemistry. Atomic Energy Publisher, Beijing
Spinks JWT, Woods RJ (1976) An introduction to radiation chemistry, 2nd edn. Wiley, New York
Alfassi ZB (1998) N-Centered radicals. Wiley, Hoboken
KozŁowska-Milner E, Broszkiewicz RK (1977) Pulse radiolysis study of aqueous nitric acid solutions. Formation mechanism, yield, and reactivtty of NO3 Radical. Radiat Phys Chem 11:253–260
Katsumura Y, Jiang PY, Nagaishi R, Oishi T, Ishigure K (1991) Pulse radiolysis study of aqueous nitric acid solutions: formation mechanism, yield, and reactivity of NO3 radical. Phys Chem 95:4435–4439
Wang JH, Bao BR, Wu MH, Sun XL, Zhang XY, Ye GA (2004) Qualitative and quantitative analysis of light hydrocarbons produced by radiation degradation of N, N-dimethyl hydroxylamine. Nucl Techniq 27:301–304
Ingold KU, Adamic K, Bowman DF, Gillan T (1971) Kinetic applications of electron paramagnetic resonance spectroscopy. I. Self-reactions of diethyl nitroxide radicals. J Am Chem Soc 93:902–908
Acknowledgements
The authors wish to extend warm acknowledgement for the financial assistance of National Natural Science Foundation of China (No. 20771074).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, J.H., Wang, P., Li, C. et al. Effect of nitrous acid on γ-ray radiolysis and radiolytic products of N, N-dimethylhydroxylamine. J Radioanal Nucl Chem 319, 759–765 (2019). https://doi.org/10.1007/s10967-019-06422-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-019-06422-y