Abstract
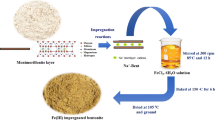
The sorption behavior of Se(IV) on Fe- and Al-modified bentonite is studied through batch experiments. In order to introduce active centers for Se(IV) sorption, bentonite was modified with iron, iron oxocations and aluminum oxocations at 400, 600 and 800 °C. It was found that calcined temperature had great effect on the sorption with above 80 % sorption at 400 °C, whereas with only 5 % sorption at 800 °C. The sorption capacities for Se(IV) were 112.5 mg/g by FeOH-B, 60.1 mg/g by AlOH-B and 71.9 mg/g by Fe-B, respectively. The pH-dependent and ionic strength-independent Se(IV) sorption on these modified bentonites demonstrated that the sorption mechanism of Se(IV) was inner-sphere surface complexation at low pH values.






Similar content being viewed by others
References
Luengo C, Puccia V, Avena M (2011) Arsenate adsorption and desorption kinetics on a Fe(III)-modified montmorillonite. J Hazard Mater 186(2–3):1713–1719
Manjanna J, Kozaki T, Sato S (2009) Fe(III)-montmorillonite: basic properties and diffusion of tracers relevant to alteration of bentonite in deep geological disposal. Appl Clay Sci 43(2):208–217
Peinemann N, Helmy AK (1992) Phosphate sorption by hydroxy-aluminium and hydroxy-iron (III) treated montmorillonites. Appl Clay Sci 6(5–6):419–428
Yan LG, Xu YY, Yu HQ, Xin XD, Wei Q, Du B (2010) Adsorption of phosphate from aqueous solution by hydroxy-aluminum, hydroxy-iron and hydroxy-iron–aluminum pillared bentonites. J Hazard Mater 179(1–3):244–250
Jörg G, Bühnemann R, Hollas S, Kivel N, Kossert K, Van Winckel S, Gostomski CLV (2010) Preparation of radiochemically pure 79Se and highly precise determination of its half-life. Appl Radiat Isot 68(12):2339–2351
Szlachta M, Chubar N (2013) The application of Fe–Mn hydrous oxides based adsorbent for removing selenium species from water. Chem Eng J 217:159–168
Kuan W-H, Lo S-L, Wang MK, Lin C-F (1998) Removal of Se(IV) and Se(VI) from water by aluminum-oxide-coated sand. Water Res 32(3):915–923
Peak D (2006) Adsorption mechanisms of selenium oxyanions at the aluminum oxide/water interface. J Colloid Interface Sci 303(2):337–345
Lo S-L, Chen T-Y (1997) Adsorption of Se(IV) and Se(VI) on an iron-coated sand from water. Chemosphere 35(5):919–930
Baur I, Johnson CA (2003) Sorption of selenite and selenate to cement minerals. Environ Sci Technol 37(15):3442–3447
Rovira M, Giménez J, Martínez M, Martínez-Lladó X, de Pablo J, Martí V, Duro L (2008) Sorption of selenium(IV) and selenium(VI) onto natural iron oxides: goethite and hematite. J Hazard Mater 150(2):279–284
Chen M-L, An M-I (2012) Selenium adsorption and speciation with Mg–FeCO3 layered double hydroxides loaded cellulose fibre. Talanta 95:31–35
You Y, Vance GF, Zhao H (2001) Selenium adsorption on Mg–Al and Zn–Al layered double hydroxides. Appl Clay Sci 20(1–2):13–25
Gonzalez CM, Hernandez J, Peralta-Videa JR, Botez CE, Parsons JG, Gardea-Torresdey JL (2012) Sorption kinetic study of selenite and selenate onto a high and low pressure aged iron oxide nanomaterial. J Hazard Mater 211–212:138–145
Zelmanov G, Semiat R (2013) Selenium removal from water and its recovery using iron (Fe3+) oxide/hydroxide-based nanoparticles sol (NanoFe) as an adsorbent. Sep Purif Technol 103:167–172
Sahin F, Volkan M, Howard AG, Ataman OY (2003) Selective pre-concentration of selenite from aqueous samples using mercapto-silica. Talanta 60(5):1003–1009
Sharrad MOM, Liu H, Fan M (2012) Evaluation of FeOOH performance on selenium reduction. Sep Purif Technol 84:29–34
Wijnja H, Schulthess CP (2000) Vibrational spectroscopy study of selenate and sulfate adsorption mechanisms on Fe and Al (Hydr)oxide surfaces. J Colloid Interface Sci 229(1):286–297
Frost RR, Griffin RA (1977) Effect of pH on adsorption of arsenic and selenium from landfill leachate by clay minerals. Soil Sci Soc Am J 41(1):53–57
Wu Z, Li C, Sun X, Xu X, Dai B, Li JE, Zhao H (2006) Characterization, acid activation and bleaching performance of bentonite from Xinjiang. Chin J Chem Eng 14(2):253–258
Mosser-Ruck R, Cathelineau M, Guillaume D, Charpentier D, Rousset D, Barres O, Michau N (2010) Effects of temperature, pH, and iron/clay and liquid/clay ratios on experimental conversion of dioctahedral smectite to berthierine, chlorite, vermiculite, or saponite. Clay Clay Miner 58(2):280–291
Sarikaya Y, Önal M, Baran B, Alemdaroğlu T (2000) The effect of thermal treatmeal on some of the physicochemical properties of a bentonite. Clay Clay Miner 48(5):557–562
Bhattacharyya KG, Sen Gupta S (2009) Calcined tetrabutylammonium kaolinite and montmorillonite and adsorption of Fe(II), Co(II) and Ni(II) from solution. Appl Clay Sci 46(2):216–221
Aytas S, Yurtlu M, Donat R (2009) Adsorption characteristic of U(VI) ion onto thermally activated bentonite. J Hazard Mater 172(2–3):667–674
Heller-Kallai L, Rozenson I (1980) Dehydroxylation of dioctahedral phyllosilicates. Clay Clay Miner. 28(5):355–368
Nguyen-Thanh D, Block K, Bandosz TJ (2005) Adsorption of hydrogen sulfide on montmorillonites modified with iron. Chemosphere 59(3):343–353
Ararem A, Bouras O, Arbaoui F (2011) Adsorption of caesium from aqueous solution on binary mixture of iron pillared layered montmorillonite and goethite. Chem Eng J 172(1):230–236
Gu L, Xu J, Lv L, Liu B, Zhang H, Yu X, Luo Z (2011) Dissolved organic nitrogen (DON) adsorption by using Al-pillared bentonite. Desalination 269(1–3):206–213
Chan YT, Kuan WH, Chen TY, Wang MK (2009) Adsorption mechanism of selenate and selenite on the binary oxide systems. Water Res 43(17):4412–4420
Ippolito JA, Scheckel KG, Barbarick KA (2009) Selenium adsorption to aluminum-based water treatment residuals. J Colloid Interface Sci 338(1):48–55
Zong P, Pan H, Wang H, He C (2013) Investigation of sequestration mechanisms of radionuclide 63Ni(II) on kaolinite in aqueous solutions. J Radioanal Nucl Chem 295(1):405–413
El-Shafey EI (2007) Sorption of Cd(II) and Se(IV) from aqueous solution using modified rice husk. J Hazard Mater 147(1–2):546–555
Acknowledgments
The authors gratefully acknowledge the financially supported by National Natural Science Foundation of China (Grant No. 11275147 and Grant No. 21207035) and Project supported by the Scientific Research Starting Foundation for Returned Overseas Chinese Scholars, Ministry of Education, China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, H., Wu, T., Chen, J. et al. Sorption of Se(IV) on Fe- and Al-modified bentonite. J Radioanal Nucl Chem 303, 107–113 (2015). https://doi.org/10.1007/s10967-014-3422-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-014-3422-5