Abstract
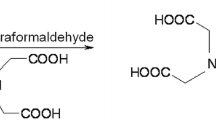
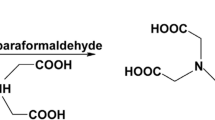
2,2′-[(8-hydroxyquinolin-7-yl)methylazanediyl]diacetic acid (HQMADA) was synthesized via reaction of 8-hydroxyquinoline with iminodiacetic acid in presence of paraformaldehyde with a yield of 27%. The obtained compound was well characterized via different analytical techniques. Labeling of the synthesized compound with technetium-99m in pertechnetate form (99mTcO4 −) in the presence of stannous chloride dihydrate was carried out via chelation reaction. The reaction parameters that affect the labeling yield such as HQMADA concentration, stannous chloride dihydrate concentration, pH of the reaction mixture, and reaction time were studied to optimize the labeling conditions. Maximum radiochemical yield of 99mTc-HQMADA complex (91.9%) was obtained by using 1.5 mg HQMADA, 50 μg SnCl2·2H2O, pH 8 and 30 min reaction time. Biodistribution studies in mice were carried out in experimentally induced infection in the left thigh using E. coli. 99mTc-HQMADA complex showed higher uptake (T/NT = 5.5 ± 0.3) in the infectious lesion than the commercially available 99mTc-ciprofloxacin (T/NT = 3.8 ± 0.8). Biodistribution studies for 99mTc-HQMADA complex in Albino mice bearing septic and aseptic inflammation models showed that 99mTc-HQMADA complex able to differentiate between septic and aseptic inflammation.






Similar content being viewed by others
References
Britton KE, Vinjamuri S, Hall AV (1997) Eur J Nucl Med 24:553–555
Seabold JE, Palestro CJ, Brown ML (1997) J Nucl Med 38:994–997
Staab EV, Mccartney H (1978) Semin Nucl Med 8:219–223
Love C, Palestro C (2004) J Nucl Med Technol 32:47–57
Chianelli M, Mather SJ, Martin-Comin J, Signore A (1997) J Nucl Med Commun 18:437–455
Corstens FHM, Van Der Meer JWM (1999) Lancet 354:765–770
Van Eerd JEM, Broekema M, Harris TD, Edwards DS, Oyen WJG, Corstens FHM, Boerman OC (2005) J Nucl Med 46:1546–1551
Laverman P, Dams ET, Oyen WJ, Storm G, Koenders EB, Prevost R, Van der Meer JW, Costens FHM, Boerman OC (1999) J Nucl Med 40:192–197
Pirmettis I, Limouris GS, Papadopoulos M (1999) Eur J Nucl Med 26:1108
Vinjamuri S, Hall AV, Solanki KK (1996) Lancet 347:233–235
Rien HS, Huub JR, Otto CB, Rudid D, Guido S (2004) J Nucl Med 45:2088–2094
Seung JO, Jin SR, Joong WS, Eun JY, Hyun JH (2002) Appl Radiat Isot 57:193–195
Welling MM, Lupetti A, Balter HS, Lanzzeri S, Souto B, Rey AM, Savio EO, Paulusma-Annema A, Pauwels EK, Nibbering PH (2001) J Nucl Med 42:788–790
Valtonen V, Karppinen L, Kariniemi AL (1989) J Infect Dis Suppl 60:79–83
El-Ghany EA, Amine AM, El-Kawy OA, Amin M (2007) J Labelled Comp Radiopharm 50:25–29
El-Ghany EA, El-Kolaly MT, Amine AM, El-Sayed AS, Abdel-Gelil F (2005) J Radioanal Nucl Chem 266:131–135
Motaleb MA (2007) J Radioanal Nucl Chem 272:95–98
Motaleb MA (2007) J Radioanal Nucl Chem 272:167–171
Yurt Lambrecht F, Durkan K, Unak P (2008) J Radioanal Nucl Chem 275:161–166
Motaleb MA (2009) J Labelled Comp Radiopharm 52:415–418
Motaleb MA (2010) J Labelled Comp Radiopharm 53:104–109
Sankha C, Sujata SD, Susmita C, Kakali D, Mridula M, Bharat RS, Samarendu S, Shantanu G (2010) Appl Radiat Isot 68:314–316
Welling MM, Paulusma-Annema A, Balter HS, Pauwels EKJ, Nibbering PH (2000) Eur J Nucl Med 27:292–296
Srivastava SC, Richards P (1983) Technetium-labled compounds. In: Rayudu GVS (ed) Radiotracers for medical applications, CRC series in radiotracers in biology and medicine. CRC Press, Boca Raton, pp 107–185
Wardell JL (1994) Tin: inorganic chemistry. In: King RB (ed) Encyclopedia of inorganic chemistry, vol 8. Wiley, New York, pp 4159–4197
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Motaleb, M.A., Alabdullah, E.S. & Zaghary, W.A. Synthesis, radiochemical and biological characteristics of 99mTc-8-hydroxy-7-substituted quinoline complex: a novel agent for infection imaging. J Radioanal Nucl Chem 287, 61–67 (2011). https://doi.org/10.1007/s10967-010-0818-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-010-0818-8