Abstract
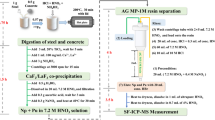
A one step Cs, lanthanides (Ln), Pu and U separation using TRU resin (Eichrom), for subsequent isotopic analyses, is presented for samples of transmutation targets containing a predominant matrix of steel elements. Cs, Ln, Pu and U are successively eluted using minor volumes (<12 mL) of 2M HNO3, 4M HCl, 0.075M oxalic acid in 1M HCl media and 0.1M ammonium hydrogen oxalate in 0.02M HNO3 media, respectively. Accurate and precise isotopic compositions obtained for these elements by thermal ionization mass spectrometry or multiple collector inductively coupled mass spectrometry show neither significant fractionation nor contamination during the complete analytical process.
Similar content being viewed by others
References
H. Isnard, M. Aubert, P. Blanchet, R. Brennetot, F. Chartier, V. Geertsen, F. Manuguerra, Spectrochim. Acta, Part 61 (2006) 150.
D. Alamelu, S. K. Aggarwal, J. Alloys and Compounds, 444–445 (2007) 640.
M. Granet, et al., High accuracy measurements of Pu isotopes by MC-ICP-MS with in situ U/Pu separation using CO2, NH3 and C2H4 gases in a collision-reaction cell, in preparation.
Z. Varga, Anal. Chim. Acta, 587 (2007) 165.
C. Pin, J. F. Santos Zalduegui, Anal. Chim. Acta, 339 (1997) 79.
S. L. Maxwell, D. J. Fauth, S. T. Nichols, J. Radioanal. Nucl. Chem., 250 (2001) 117.
U. Nygren,, I. Rodushkin, C. Nilsson, D. C. Baxter, JAAS 18 (2003) 1426.
S. -H. Lee, J. La Rosa, J. Gastaud, P. P. Povinec, J. Radioanal. Nucl. Chem., 263 (2005) 419.
S. L. Maxwell, J. Radioanal. Nucl. Chem., 267 (2006) 537.
S. L. Maxwell III, B. K. Culligan, J. Radioanal. Nucl. Chem., 270 (2006) 699.
R. Jakopic, P. Tavcar, L. Benedik, Appl. Radiat. Isotopes, 65 (2007) 504.
E. P. Horwitz, R. Chiarizia, M. L. Dietz, H. Diamond, D. Nelson, Analyt. Chim. Acta, 281 (1993) 361.
R. Chiarizia, E. P. Horwitz, Inorg. Chim. Acta, 140 (1987), 261.
M. Yamaura, H. T. Matsuda, J. Radioanal. Nucl. Chem., 241 (1999) 277.
M. Yamaura, H. T. Matsuda, J. Radioanal. Nucl. Chem., 224 (1997) 83.
J. W. Grate, O. B. Egorova, S. K. Fiskum, Analyst, 124 (1999) 1143.
O. B. Egorov, M. J. O’Hara, O. T. Farmer III, J. W. Grate, Analyst, 126 (2001) 1594.
J. M. Cleveland, The Chemistry of Plutonium, American Nuclear Society, Illinois USA, 1979, p. 653.
F. Weigel, J. J. Katz, G. T. Seaborg, in: The Chemistry of the Actinide Elements, J. J. Katz, G. T. Seaborg, L. R. Morss, (Eds.), Chapman and Hall, London, New York, ed. 2, 1986, Vol 1, p. 499.
C.-S. Kim, C.-K. Kim, P. Martin, U. Sansone, J. Anal. At. Spectrom., 22 (2007) 827.
E. P. Horwitz, M. L. Dietz, R. Chiarizia, H. Diamond, S. L. Maxwell, M. R. Nelson, Anal. Chim. Acta, 310 (1995) 63.
J. B. Truscott, L. Bromley, P. Jones, E. H. Evans, J. Turner, B. Fairman, J. Anal. At. Spectrom., 14 (1999) 627.
G. L. Silver, J. Radioanal. Nucl. Chem., 267 (2006), 53.
G. L. Silver, J. Radioanal. Nucl. Chem., 275 (2008) 225.
M. H. Lee, Y. J. Park, W. H. Kim, J. Radioanal. Nucl. Chem., 273 (2007) 375.
R. J. Lemire, J. Fuger, H. Nitsche, P. E. Potter, M. H. Rand, J. U. Rydberg, K. Spahiu, J. C. Sullivan, W. J. Ullman, P. Vitorge, H. Wanner, Chemical thermodynamics of neptunium and plutonium, Nuclear Energy Agency Data Bank, OECD, Vol 4, Chemical Thermodynamics, 2001.
J. L. Ryan, J. Phys. Chem., 64 (1960) 1375.
J. L. Ryan, J. Phys. Chem., 65, (1961) 1099.
Eichrom Industries Inc., Eichrom Analytical Products description, Eichrom Industries Inc.,1995.
R. M. Smith, A. E. Martell, R. J. Motekaitis, NIST critically selected stability constants of metal complexes database, version 8. 0 for Windows, 2004.
S. D. Tanner, C. Li, V. Vais, V. I. Baranov, D. R. Bandura, Anal. Chem., 76 (2004) 3042.
D. Alamelu, P. S., Khodade, P. M. Shah, S. K. Aggarwal, Int. J. Mass Spectrometry, 239 (2004) 51.
S. K. Aggarwal, D. Alamelu, P. S. Khodade, P. M. Shah, J. Radioanal. Nucl. Chem., 237 (2007) 775.
H. Isnard, et al., in preparation.
H. Isnard, R. Brennetot, C. Caussignac, N. Caussignac, F. Chartier, Int. J. Mass Spectrometry, 246 (2005) 66.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Quidelleur, S., Granet, M., Laszak, I. et al. One step U-Pu-Cs-Ln-steel separation using TRU preconditioned extraction resins from Eichrom for application on transmutation targets. J Radioanal Nucl Chem 280, 507–517 (2009). https://doi.org/10.1007/s10967-009-7449-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-009-7449-y