Summary
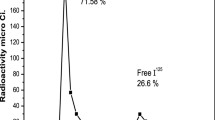
A simple, one step, inclusive immunoradiometric assay for human chorionic gonadotropin (hCG) employing monoclonal antibodies is described. Commercially available monoclonal antibodies from various commercial sources were screened. Identified “detection” antibody was radiolabeled with 125I and the selected “capture” antibody was chemically coupled to magnetizable cellulose to form a solid phase. In the procedure developed, standard/sample, radiolabeled antibody and capture antibody were incubated together for 3 hours at room temperature with shaking. After incubation, the bound complex was quantitated for the associated radioactivity. The analytical sensitivity observed was 1.0 mIU/ml with a wide concentration range up to 1000 mIU/ml of hCG. “High dose hook” of the developed assay was observed beyond 2000 mIU/ml. Results showed that the developed assay had a good precision: intra-assay CV less than 8%, inter-assay CV less than 10% and good analytical recovery of 97-109%. The clinical samples analyzed by the developed procedure showed a good correlation with that of the commercial kit (r = 0.92; y = 0.99x+0.51).
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vrinda, P., Paradkar, S., Nagvekar, U. et al. Solid phase inclusive immunoradiometric assay for human chorionic gonadotropin using monoclonal antibodies. J Radioanal Nucl Chem 266, 277–283 (2005). https://doi.org/10.1007/s10967-005-0904-5
Issue Date:
DOI: https://doi.org/10.1007/s10967-005-0904-5