Abstract
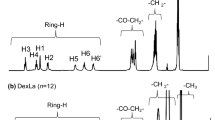
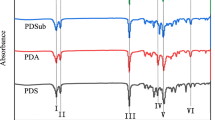
A series of fully-acylated dextrin esters (DS = 3) with varying side-chain lengths (C2–12) were synthesized by heterogeneous esterification using trifluoroacetic anhydride/carboxylic acid. The influence of side-chain lengths on structure and properties of dextrin esters were investigated by structural, thermal, mechanical and hydrophobic analysis. The thermal stability of dextrin was enhanced by esterification, presenting ca. 40–60 °C higher decomposition temperatures than that of neat-dextrin. The transition temperatures of melting and crystallization were not observed for all dextrin esters because they were amorphous polymers. The glass transition temperature (Tg) was not observed in dextrin but was observed in dextrin esters. As increasing side-chain length, Tgs of dextrin esters decreased ranged from 162.2 °C (C2) to 49.2 °C (C12). Colorless and transparent dextrin ester films were prepared to measure the film properties. Tensile strength of dextrin ester films tended to decrease with increasing side-chain lengths, whereas the elongation at break increased. And, dextrin ester films showed significantly increased hydrophobicity with a contact angle of up to 102° (C12).









Similar content being viewed by others
References
Iwata T (2015) Biodegradable and bio-based polymers: Future prospects of eco-friendly plastics. Angew Chem Int Ed 54:3210–3215
Pan Y, Farmahini-Farahani M, O’Hearn P, Xiao H, Ocampo H (2016) An overview of bio-based polymers for packaging materials. J Bioresources Bioprod 1:106–113
Ferreira AR, Alves VD, Coelhoso IM (2016) Polysaccharide-based membranes in food packaging applications. Membranes. https://doi.org/10.3390/membranes6020022
Carvalho J, Gonçalves C, Gil AM, Gama FM (2007) Production and characterization of a new dextrin based hydrogel. Eur Polym J 43:3050–3059
Zare EN, Lakouraj MM, Mohseni M (2014) Biodegradable polypyrrole/dextrin conductive nanocomposite: synthesis, characterization, antioxidant and antibacterial activity. Synth Met 187:9–16
Lin LH, Lai YC, Chen KM, Li CS (2016) Preparation and surface activities of modified soy protein–dextrin surfactants. J Surfactant Deterg 19:19–28
Das D, Patra P, Ghosh P, Rameshbabu AP, Dhara S, Pal S (2016) Dextrin and poly (lactide)-based biocompatible and biodegradable nanogel for cancer targeted delivery of doxorubicin hydrochloride. Polym Chem 7:2965–2975
Glasser WG, McCartney BK, Samaranayake G (1994) Cellulose derivatives with low degree of substitution; 3. The biodegradability of cellulose esters using a simple enzyme assay. Biotechnol Prog 10:214–219
Crépy L, Miri V, Joly N, Martin P, Lefebvre JM (2011) Effect of side chain length on structure and thermomechanical properties of fully substituted cellulose fatty esters. Carbohydr Polym 83:1812–1820
Vanmarcke A, Leroy L, Stoclet G, Duchatel-Crépy L, Lefebvre JM, Joly N, Gaucher V (2017) Influence of fatty chain length and starch composition on structure and properties of fully substituted fatty acid starch esters. Carbohydr Polym 164:249–257
Ikai T, Yun C, Kojima Y, Suzuki D, Maeda K, Kanoh S (2016) Development of amylose-and β-cyclodextrin-based chiral fluorescent sensors bearing terthienyl pendants. Molecules. https://doi.org/10.3390/molecules21111518
Skołucka-Szary K, Ramięga A, Piaskowska W, Janicki B, Grala M, Rieske P, Bartczak Z, Piaskowski S (2016) Synthesis and physicochemical characterization of chitin dihexanoate—A new biocompatible chitin derivative—In comparison to chitin dibutyrate. Mater Sci Eng C 60:489–502
Marubayashi H, Yukinaka K, Enomoto-Rogers Y, Takemura A, Iwata T (2014) Curdlan ester derivatives: Synthesis, structure, and properties. Carbohydr Polym 103:427–433
Chien CY, Enomoto-Rogers Y, Takemura A, Iwata T (2017) Synthesis and characterization of regioselectively substituted curdlan hetero esters via an unexpected acyl migration. Carbohydr Polym 155:440–447
Cumpstey I (2013) Chemical modification of polysaccharides. ISRN Org Chem 2013:417672
Hettrich K, Fischer S, Schröder N, Engelhardt J, Drechsler U, Loth F (2005) Derivatization and characterization of xylan from oat spelts. Macromol Symp 232:37–48
Hafrén J, Zou W, Córdova A (2006) Heterogeneous organoclick derivatization of polysaccharides. Macromol Rapid Commun 27:1362–1366
Yang BY, Montgomery R (2006) Acylation of starch using trifluoroacetic anhydride promoter. Starch-Stärke 58:520–526
Danjo T, Enomoto-Rogers Y, Takemura A, Iwata T (2014) Syntheses and properties of glucomannan acetate butyrate mixed esters. Polym Degrad Stab 109:373–378
Enomoto-Rogers Y, Iio N, Takemura A, Iwata T (2015) Synthesis and characterization of pullulan alkyl esters. Eur Polym J 66:470–477
Fundador NGV, Enomoto-Rogers Y, Takemura A, Iwata T (2012) Syntheses and characterization of xylan esters. Polymer 53:3885–3893
Ponder GR, Richards GN, Stevenson TT (1992) Influence of linkage position and orientation in pyrolysis of polysaccharides: A study of several glucans. J Anal Appl Pyrolysis 22:217–229
Ponder GR, Richards GN (1994) A review of some recent studies on mechanisms of pyrolysis of polysaccharides. Biomass Bioenergy 7:1–24
Aburto J, Alric I, Thiebaud S, Borredon E, Bikiaris D, Prinos J, Panayiotou C (1999) Synthesis, characterization, and biodegradability of fatty-acid esters of amylose and starch. J Appl Polym Sci 74:1440–1451
Cunha AG, Gandini A (2010) Turning polysaccharides into hydrophobic materials: a critical review. Part 1. Cellulose. Cellulose 17:875–889
Winkler H, Vorwerg W, Rihm R (2014) Thermal and mechanical properties of fatty acid starch esters. Carbohydr Polym 102:941–949
Lu X, Luo Z, Yu S, Fu X (2012) Lipase-catalyzed synthesis of starch palmitate in mixed ionic liquids. J Agric Food Chem 60:9273–9279
Crépy L, Chaveriat L, Banoub J, Martin P, Joly N (2009) Synthesis of cellulose fatty esters as plastics—influence of the degree of substitution and the fatty chain length on mechanical properties. ChemSusChem 2:165–170
Acknowledgments
This work was supported by Japanese Government (Monbukagakusho; MEXT) Scholarship for Top Global University Project 2017 and JST-ALCA Project (White Biotechnology).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Bio-Based Polymers
Rights and permissions
About this article
Cite this article
Lee, H.Y., Danjo, T. & Iwata, T. Synthesis and characterization of dextrin derivatives by heterogeneous esterification. J Polym Res 25, 183 (2018). https://doi.org/10.1007/s10965-017-1333-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-017-1333-1