Abstract
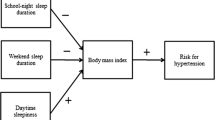
While mounting evidence suggests that sleep plays an important role in the etiology of obesity, the underlying pathogenic pathways are complex and unresolved. Experimental sleep deprivation studies demonstrate sympathovagal imbalance, indicative of diminished parasympathetic activity and/or heightened sympathetic activity, is consequent to poor sleep. Further, obese children exhibit sympathovagal imbalance, particularly during the night, compared to non-obese children. The question remains whether sympathovagal imbalance is one potential pathophysiological pathway underlying the association between sleep and obesity. The aim of the present study was to examine whether sympathovagal imbalance contributed to the association between sleep and obesity in children. Participants included 564 children aged 10 to 12 years (M = 11.67, SD = 0.95; 43.5 % girls) from the QUALITY Cohort, a longitudinal study of children at-risk for the development of obesity. While children were at-risk due to confirmed parental obesity status, 57.7 % of children were of normal body mass index (5–85th percentile). Sleep duration, sleep timing, and sleep disturbances were based on child- and parent-report. Anthropometrics were measured for central adiposity (waist circumference) and body composition (body mass index, fat mass index). Sympathovagal imbalance was derived from heart rate variability spectral analyses. Estimated path coefficients revealed that sympathovagal imbalance partially contributed to the association between poor sleep (later bedtimes, sleep-disordered breathing) and obesity. These findings highlight the importance of better understanding sympathovagal imbalance and its role in the etiology and maintenance of obesity. Future research should consider investigating nocturnal sympathovagal balance in youth.

Similar content being viewed by others
Abbreviations
- BMI:
-
Body mass index
- CSHQ:
-
Children’s Sleep Habits Questionnaire
- DEXA:
-
Dual-energy X-ray absorptiometry
- ECG:
-
Electrocardiogram
- FFT:
-
Fast Fourier Transform
- HF:
-
High frequency
- LF:
-
Low frequency
References
Alvarez, G. E., Ballard, T. P., Beske, S. D., & Davy, K. P. (2004). Subcutaneous obesity is not associated with sympathetic neural activation. American Journal of Physiology Heart and Circulatory Physiology, 287, H414–H418.
Baron, R. M., & Kenny, D. A. (1986). The moderator–mediator variable distinction in social psychological research: Conceptual, strategic and statistical considerations. Journal of Personality and Social Psychology, 51, 1171–1182.
Beebe, D. W., Lewin, D., Zeller, M., McCabe, M., MacLeod, K., Daniels, S. R., & Amin, R. (2007). Sleep in overweight adolescents: Shorter sleep, poorer sleep quality, sleepiness, and sleep-disordered breathing. Journal of Pediatric Psychology, 32, 69–79.
Berntson, G. G., Bigger, J. T., Eckberg, D. L., Grossman, P., Kaufman, P. G., Malik, M., & van der Molen, M. W. (1997). Heart rate variability: Origins, methods, and interpretive caveats. Psychophysiology, 34, 623–648.
Björntorp, P. (2001). Do stress reactions cause abdominal obesity and comorbidities? Obesity Reviews, 2, 73–86.
Bonnet, M. H., & Arand, D. L. (2010). Hyperarousal and insomnia: State of the science. Sleep Medicine Reviews, 14, 9–15.
Cacioppo, J. T., Berntson, G. G., Binkley, P. F., Quigley, K. S., Uchino, B. N., & Fieldstone, A. (1994). Autonomic cardiac control. II. Noninvasive indices and basal response as revealed by autonomic blockades. Psychophysiology, 31, 586–598.
Danese, A., & McEwen, B. S. (2012). Adverse childhood experiences, allostasis, allostatic load and age-related disease. Physiology & Behavior, 106, 29–39.
Daniels, S. R., Morrison, J. A., Sprencher, D. L., Khoury, P., & Kimball, T. R. (1999). Association of body fat distribution and cardiovascular risk factors in children and adolescents. Circulation, 99, 541–545.
Diedrich, A., Jordan, J., Tank, J., Shannon, J. R., Robertson, R., Luft, F. C., & Biaggioni, I. (2003). The sympathetic nervous system in hypertension: Assessment by blood pressure variability and ganglionic blockade. Journal of Hypertension, 21, 1677–1686.
Drapeau, V., Therrien, F., Richard, D., & Tremblay, A. (2003). Is visceral obesity a physiological adaptation to stress? Panminerva Medicine, 45, 189–195.
Ekstedt, M., Åkerstedt, T., & Söderström, M. (2004). Microarousals during sleep are associated with increased levels of lipids, cortisol, and blood pressure. Psychosomatic Medicine, 66, 925–931.
El-Sheikh, M., Buckhalt, J. A., Keller, P. S., & Granger, D. A. (2008). Children’s objective and subjective sleep disruptions: Links with afternoon cortisol levels. Health Psychology, 27, 26–33.
Fliers, E., Kreier, F., Voshol, P. J., Havekes, L. M., Saurewein, H. P., Kalsbeek, A., & Romijn, J. A. (2003). White adipose tissue: Getting nervous. Journal of Neuroendocrinology, 15, 1005–1010.
Franco, P., Seret, N., Van Hees, J. N., Lanquart, J.-P., Groswasser, J., & Kahn, A. (2003). Cardiac changes during sleep in sleep-deprived infants. Sleep, 26, 845–848.
Gaina, A., Sekine, M., Chen, X., Hamanishi, S., & Kagamimori, S. (2004). Validity of child sleep diary questionnaire among junior high school children. Journal of Epidemiology, 14, 1–4.
Graziano, P. A., Calkins, S. D., Keane, S. P., & O’Brien, M. (2011). Cardiovascular regulation profile predicts developmental trajectory of BMI and pediatric obesity. Obesity (Silver Spring), 19, 1818–1825.
Hairston, I. S., Ruby, N. F., Brooke, S., Peyron, C., Denning, D. P., Heller, H. C., & Sapolsky, R. M. (2001). Sleep deprivation elevates plasma corticosterone levels in neonatal rats. Neuroscience Letters, 15, 29–32.
Hakim, F., Gozal, D., & Kheirandish-Gozal, L. (2012). Sympathetic and catecholaminergic alterations in sleep apnea with particular emphasis on children. Frontiers in Neurology, 3, 1–13.
Haqq, A. M., DeLorey, D. S., Sharma, A. M., Freemark, M., Kreier, F., Mackenzie, M. L., & Richer, L. P. (2011). Autonomic nervous system dysfunction in obesity and Prader–Willi syndrome: Current evidence and implications for future obesity therapies. Clinical Obesity, 1, 175–183.
He, M., Harris, S., Piche, L., & Beynon, C. (2009). Understanding screen- related sedentary behavior and its contributing factors among school-aged children: A social-ecologic exploration. American Journal of Health Promotion, 23, 299–308.
Jarrin, D. C., McGrath, J. J., & Drake, C. L. (2013). Beyond sleep duration: Distinct sleep dimensions are associated with obesity in children and adolescents. International Journal of Obesity, 37, 552–558.
Jarrin, D., McGrath, J. J., Giovanniello, S., Poirier, P., & Lambert, M. (2012). Measurement fidelity of heart rate variability signal processing: The devil is in the details. International Journal of Psychophysiology, 86(1), 88–97.
Jarrin, D. C., McGrath, J. J., Poirier, P., Séguin, L., Tremblay, R. E., Montplaisir, J. Y., et al. (2014). Short-term heart rate variability in a population-based sample of 10-year-old children. Pediatric Cardiology. doi:10.1007/s00246-014-0962-y.
Kaufman, C. L., Kaiser, D. R., Steinberger, J., Kelly, A. S., & Dengel, D. R. (2007). Relationships of cardiac autonomic function with metabolic abnormalities in childhood obesity. Obesity, 15, 1164–1171.
Knutson, K. L. (2012). Does inadequate sleep play a role in vulnerability to obesity? American Journal of Human Biology, 24, 361–371.
Knutson, K. L., & van Cauter, E. (2008). Associations between sleep loss and increased risk of obesity and diabetes. Annals of New York Academy Science, 1129, 287–304.
Kwok, K.-L., Yung, T.-C., Ng, D. K., Chan, C.-H., Lau, W.-F., & Fu, Y.-M. (2011). Heart rate variability in childhood obstructive sleep apnea. Pediatric Pulmonology, 210, 205–210.
Laberge, L., Petit, D., Simard, C., Vitaro, F., Tremblay, R. E., & Montplaisir, J. (2011). Development of sleep patterns in early adolescence. Journal of Sleep Research, 10, 59–67.
Lambert, M., Van Hulst, A., O’Loughlin, J., Tremblay, A., Barnett, T. A., Charron, H., & Paradis, G. (2011). Cohort profile: The Quebec adipose and lifestyle investigation in youth cohort. International Journal of Epidemiology, 41, 1533–1544.
Lewandowski, A. S., Toliver-Sokol, M., & Palermo, T. M. (2011). Evidence-based review of subjective pediatric sleep measures. Journal of Pediatric Psychology, 36, 780–793.
Lumeng, J. C., Somashekar, D., Appugliese, D., Kaciroti, N., Corwyn, R. F., & Bradley, R. H. (2007). Shorter sleep duration is associated with increased risk for being overweight at ages 9 to 12 years. Pediatrics, 120, 1020–1029.
Marshall, W. A., & Tanner, J. M. (1969). Variations in the pattern of pubertal changes in girls. Archives of Disease in Childhood, 44, 291–303.
Marshall, W. A., & Tanner, J. M. (1970). Variations in the pattern of pubertal changes in boys. Archives of Disease in Childhood, 45, 13–23.
Martini, G., Riva, P., Rabbia, F., Molini, V., Ferrero, G. B., Cerutti, F., & Vrglio, F. (2001). Heart rate variability in childhood obesity. Clinical Autonomic Research, 11, 87–91.
Matricciani, L. (2013). Subjective reports of children’s sleep duration: Does the question matter? A literature review. Sleep Medicine, 14, 303–311.
Nagai, N., Matsumoto, T., Kita, H., & Moritani, T. (2003). Autonomic nervous system activity and the state and development of obesity in Japanese school children. Obesity Research, 11, 25–32.
Ogdon, C. L., Carroll, M. D., Kit, B. K., & Flegal, K. M. (2012). Prevalence of obesity and trends in body mass index among US children and adolescents, 1999–2010. The Journal of the American Medical Association, 307, 483–490.
Ogdon, C. L., Kuczmarski, R. J., Flegal, K. M., Mei, Z., Guo, S., Wei, R., & Johnson, C. L. (2002). Growth charts for the United States: Improvements to the 1977 National Center for Health Statistics Version. Pediatrics, 109, 45–60.
Olds, T. S., Maher, C. A., & Matricciani, L. (2011). Sleep duration or bedtime? Exploring the relationship between sleep habits and weight status and activity patterns. Sleep, 34, 1299–1307.
Owens, J. A., Spirito, A., & McGuinn, M. (2000). The children’s sleep habits questionnaire (CSHQ): Psychometric properties of a survey instrument for school-aged children. Sleep, 23, 1043–1051.
Pagani, M., Montano, N., Porta, A., Malliani, A., Abboud, F. M., Birkett, C., & Somers, V. K. (1997). Relationship between spectral components of cardiovascular variabilities and direct measures of muscle sympathetic nerve activity in humans. Circulation, 95, 1441–1448.
Peterson, H. R., Rothschild, M., Weinberg, C. R., Fell, R. D., McLeish, K. R., & Pfeifer, M. A. (1998). Body fat and the activity of the autonomic nervous system. New England of Journal Medicine, 318, 1077–1083.
Preacher, K. J., & Hayes, A. F. (2008). Asymptotic and resampling strategies for assessing and comparing indirect effects in multiple mediator models. Behavior Research Methods, 40, 879–891.
Rabbia, F., Silke, B., Conterno, A., Grosso, T., De Vito, B., Rabbone, I., & Veglio, F. (2003). Assessment of cardiac autonomic modulation during adolescent obesity. Obesity Research, 11, 541–548.
Rayner, D. V., & Trayhurn, P. (2001). Regulation of leptin production: Sympathetic nervous system interactions. Journal of Molecular Medicine, 79, 8–20.
Reyes Del Paso, G. A., Langewitz, W., Mulder, L. J., van Roon, A., & Duschek, S. (2013). The utility of low frequency heart rate variability as an index of sympathetic cardiac tone: A review with emphasis on a reanalysis of previous studies. Psychophysiology, 50, 477–487.
Riva, G., Martini, G., Milan, A., Paglieri, C., Chiandussi, L., & Veglio, F. (2001). Obesity and autonomic function in adolescence. Clinical & Experimental Hypertenion, 23, 57–67.
Rodríguez-Colón, S. M., He, F., Shaffer, M. L., Li, X., Vgontzas, A. N., Bixler, E. O., & Liao, D. (2011). Insomnia symptoms and sleep duration are associated with impaired cardiac autonomic modulation in children. Neuroscience & Medicine, 2, 288–294.
Roenneberg, T., Kuehnle, T., Pramstaller, P. P., Ricken, J., Havel, M., Guth, A., & Merrow, M. (2004). A marker for the end of adolescence. Current Biology, 14, R1038–R1039.
Sallis, J. F., Condon, S. A., Goggin, K. J., Roby, J. J., Kolody, B., & Alcaraz, J. E. (1993). The development of self-administered physical activity surveys for 4th grade students. Research Quarterly for Exercise and Sport, 64, 25–31.
Scheer, F. A., Hilton, M. F., Mantzoros, C. S., & Shea, S. A. (2009). Adverse metabolic and cardiovascular consequences of circadian misalignment. Proceedings of the National Academy of Sciences USA, 106, 4453–4458.
Spiegel, K., Leproult, R., L’Hermite-Baleriauz, M., Copinschi, G., Penev, P. D., & van Cauter, E. (2004a). Leptin levels are dependent on sleep duration: Relationships with sympathovagal balance, carbohydrate regulation, cortisol, and thyrotropin. Journal of Clinical of Endocrinology & Metabolism, 89, 5762–5771.
Spiegel, K. S., Tasali, E., Penev, P., & van Cauter, E. (2004b). Brief communication: Sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Annals of Internal Medicine, 141, 846–850.
Tasali, E., Leproult, R., Ehrmann, D. A., & van Cauter, E. (2008). Slow-wave sleep and the risk of type 2 diabetes in humans. Proceedings of the National Academy of Sciences of the United States of America, 105, 1044–1049.
Task Force of the European Society of Cardiology the North American Society of Pacing Electrophysiology. (1996). Heart rate variability: Standards of measurement, physiological interpretation and clinical use. Circulation, 93, 1043–1065.
Tochikubo, O., Ikeda, A., Miyajima, E., & Ishii, M. (1996). Effects of insufficient sleep on blood pressure monitored by a new multibiomedical recorder. Hypertension, 27, 1318–1324.
Tonetti, L., Fabbri, M., & Natale, V. (2008). Sex difference in sleep-time preference and sleep need: A cross-sectional survey among Italian pre-adolescents, adolescents, and adults. Chronobiology International: The Journal of Biological & Medical Rhythm Research, 25(5), 745–759.
van Baak, M. A. (2001). The peripheral sympathetic nervous system in human obesity. Obesity Reviews, 2, 3–14.
Walter, L. M., Foster, A. M., Patterson, R. R., Anderson, V., Davey, M. J., Nixon, G. M., et al. (2009). Cardiovascular variability during periodic leg movements in sleep in children. Sleep, 32(8), 1093–1099.
Wang, G., Xu, G., Liu, Z., Lu, N., Ma, R., & Zhang, E. (2013). Sleep patterns and sleep disturbances among Chinese school-aged children: Prevalence and associated factors. Sleep Medicine, 14, 45–52.
Wanger, J., Clausen, J. L., Coates, A., & Pedersen, O. F. (2005). Standardisation of the measurement of lung volumes. European Respiratory Journal, 26, 511–522.
Wawryk, A. M., Bates, D. J., & Couper, J. J. (1997). Power spectral analysis of heart rate variability in children and adolescents with IDDM. Diabetes Care, 20, 1416–1421.
World Health Organization. (2008). Obesity and overweight. Retrieved March 1, 2008 from http://www.who.int/dietphysicalactivity/publications/facts/obesity/en/.
Wolfson, A. R., & Carskadon, M. A. (1998). Sleep schedules and daytime functioning in adolescents. Child Development, 69, 875–887.
Yakinci, C., Mungen, B., Karabiber, H., Tayfun, M., & Evereklioglu, C. (2000). Autonomic nervous system functions in obese children. Brain & Development, 22, 151–153.
Zhong, X., Hilton, H. J., Gates, G. J., Jelic, S., Stern, Y., Bartels, M. N., & Basner, R. C. (2005). Increased sympathetic and decreased parasympathetic cardiovascular modulation in normal humans with acute sleep deprivation. Journal of Applied Physiology, 98, 2024–2032.
Acknowledgments
This work was partly supported by the Canadian Institutes of Health Research (J. McGrath OCO-79897, MOP-89886, MSH-95353; D.C. Jarrin BO512201). The QUALITY Cohort is funded by the Canadian Institutes of Health Research (M. Henderson #MOP-119512; M. Lambert #OHO-69442, #NMD-94067, #MOP-97853), the Heart and Stroke Foundation of Canada (#PG-040291), and Fonds de la recherche en santé du Québec. Dr. Marie Lambert (July 1952–February 2012), pediatric geneticist and researcher, initiated the QUALITY Cohort. Her leadership and devotion to QUALITY will always be remembered and appreciated. Sincere thanks to the dedicated QUALITY Cohort staff, especially Catherine Pelletier (Coordinator), Ginette Lagacé, Natacha Gaulin-Marion, and Hugues Charron, without whom this research would not be possible. Finally, we are grateful to all the families that participate in the QUALITY Cohort.
Conflict of interest
None to disclose.
Author contributions
The QUALITY Cohort Collaborative Group designed and coordinated the study and collected measurements at clinic visits. J.J.M. and P.P. planned the study design and analysis of the ECG recordings. D.C.J. performed the statistical analyses. D.C.J. and J.J.M. conceived the research question, interpreted the findings, and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
The QUALITY (QUebec Adipose and Lifestyle InvesTigation in Youth) Cohort Collaborative Group is an inter-university research team from Université de Montréal, Concordia University, Université Laval, McGill University, and University of Toronto including (alphabetical): Tracie A. Barnett, Arnaud Chiolero, Vicky Drapeau, Josée Dubois, Katherine Gray-Donald, Melanie Henderson (PI), Marie Lambert (posthumous), Émile Lévy, Marie-Eve Mathieu, Katerina Maximova, Jennifer J. McGrath, Belinda Nicolau, Jennifer O’Loughlin, Gilles Paradis, Paul Poirier, Catherine M. Sabiston, Angelo Tremblay, and Michael Zappitelli.
Rights and permissions
About this article
Cite this article
Jarrin, D.C., McGrath, J.J., Poirier, P. et al. Autonomic Dysfunction: A Possible Pathophysiological Pathway Underlying the Association Between Sleep and Obesity in Children At-Risk for Obesity. J Youth Adolescence 44, 285–297 (2015). https://doi.org/10.1007/s10964-014-0235-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10964-014-0235-3