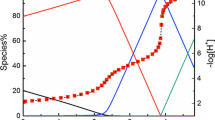
Abstract
Glufosinate, an analogue of glutamic acid (also known as phosphinothricin), is an important herbicide. The protonation constants of glufosinate were determined by means of potentiometric titrations at variable temperatures with ionic strength 0.5 mol·L−1 (NaCl solution). The heat effects of the protonation reactions of glufosinate were measured by direct calorimetry. NMR spectroscopies have demonstrated that the first protonation site occurs on the nitrogen atom in the amino group, followed by one of the oxygen atoms in the phosphono group, and finally the carboxyl oxygen atom. This trend is in good agreement with the enthalpy of protonation and crystal structure results. These data will help to predict the speciation of glufosinate in physiological systems.






Similar content being viewed by others
References
Wang, Y., Browning, M., Ruemmele, B.A., Chandlee, J.M., Kausch, A.P., Jackson, N.: Glufosinate reduces fungal diseases in transgenic glufosinate-resistant bentgrasses (Agrostis spp.). Weed Sci. 51, 130–137 (2017)
Liu, C.-A., Zhong, H., Vargas, J., Penner, D., Sticklen, M.: Prevention of fungal diseases in transgenic, bialaphos- and glufosinate-resistant creeping bentgrass (Agrostis palustris). Weed Sci. 46, 139–146 (2017)
Pline, W.A., Lacy, G.H., Stromberg, V., Hatzios, K.K.: Antibacterial activity of the herbicide glufosinate on pseudomonas syringae pathovar glycinea. Pestic. Biochem. Phys. 71, 48–55 (2001)
Christ, B., Hochstrasser, R., Guyer, L., Francisco, R., Aubry, S., Hörtensteiner, S., Weng, J.-K.: Non-specific activities of the major herbicide-resistance gene bar. Nat. Plants. 3, 937–945 (2017)
Wehrmann, A., Vliet, A.V., Opsomer, C., Botterman, J., Schulz, A.: The similarities of bar and pat gene products make them equally applicable for plant engineers. Nat. Biotechnol. 14, 1274–1278 (1996)
Duke, S.O.: Taking stock of herbicide-resistant crops ten years after introduction. Pest Manage. Sci. 61, 211–218 (2005)
Wohlleben, W., Arnold, W., Broer, I., Hillemann, D., Strauch, E., Punier, A.: Nucleotide sequence of the phosphinothricin n-acetyltransferase gene from streptomyces viridochromogenes Tü494 and its expression in nicotiana tabacum. Gene 70, 25–37 (1988)
Xu, J., Gu, X., Guo, Y., Tong, F., Chen, L.: Adsorption behavior and mechanism of glufosinate onto goethite. Sci. Total Environ. 560, 123–130 (2016)
Khenifi, A., Derriche, Z., Mousty, C., Prévot, V., Forano, C.: Adsorption of glyphosate and glufosinate by Ni2AlNO3 layered double hydroxide. Appl. Clay Sci. 47, 362–371 (2010)
Ambrose, C., Hoggard, P.E.: Metal complexes of glufosinate. J. Agric. Food Chem. 37, 1442–1444 (1989)
Liu, B., Dong, L., Yu, Q., Li, X., Wu, F., Tan, Z., Luo, S.: Thermodynamic study on the protonation reactions of glyphosate in aqueous solution: Potentiometry, calorimetry and nmr spectroscopy. J. Phys. Chem. B. 120, 2132–2137 (2016)
Li, X., Zhang, Z., Endrizzi, F., Martin, L.R., Luo, S., Rao, L.: Effect of temperature on the protonation of N-(2-hydroxyethyl)ethylenediamine-N,N′,N′-triacetic acid in aqueous solutions: Potentiometric and calorimetric studies. J. Chem. Thermodyn. 85, 35–41 (2015)
Li, Q., Liu, B., Tian, Y., Yu, Q., Mu, W., Wei, H., Wang, D., Li, X., Luo, S.: Thermodynamics of the protonation of an analogue of glyphosate, N-(phosphonomethyl)-l-proline, in aqueous solutions. J. Solution Chem. 46, 1048–1058 (2017)
Li, X., Zhang, Z., Martin, L.R., Luo, S., Rao, L.: Complexation of NpO2 + with (2-hydroxyethyl)ethylenediaminetriacetic acid (HEDTA) in aqueous solutions: thermodynamic studies and structural analysis. RSC Adv. 6, 114916–114926 (2016)
Xie, X., Tian, Y., Qin, Z., Yu, Q., Wei, H., Wang, D., Li, X., Wang, X.: Complexation of manganese with glutarimidedioxime: implication for extraction uranium from seawater. Sci. Rep. 7, 43503 (2017)
Rossotti, F.J.C., Rossotti, H.: Potentiometric titrations using Gran plots: a textbook omission. J. Chem. Educ. 42, 375 (1965)
Gran, G.: Determination of the equivalence point in potentiometric titrations: Part II. Analyst. 77, 661–671 (1952)
Gans, P., Sabatini, A., Vacca, A.: Investigation of equilibria in solution. Determination of equilibrium constants with the Hyperquad suite of programs. Talanta 43, 1739–1753 (1996)
Liu, B., Tian, Y., Yu, Q., Li, Q., Mu, W., Tan, Z., Wu, F., Wang, D., Li, X.: Determination of protonation constants of O-phospho-l-serine in aqueous solution: potentiometry, microcalorimetry, NMR spectroscopy and quantum chemical calculations. J. Solution Chem. 46, 2281–2292 (2017)
Alderighi, L., Gans, P., Ienco, A., Peters, D., Sabatini, A., Vacca, A.: Hyperquad simulation and speciation (Hyss): a utility program for the investigation of equilibria involving soluble and partially soluble species. Coord. Chem. Rev. 184, 311–318 (1999)
Endrizzi, F., Melchior, A., Tolazzi, M., Rao, L.: Complexation of uranium(VI) with glutarimidoxioxime: thermodynamic and computational studies. Dalton Trans. 44, 13835–13844 (2015)
Eyschen, J., Vitoux, B., Rahuel-Clermont, S., Marraud, M., Branlant, G., Cung, M.T.: Phosphorus-31 nuclear magnetic resonance studies on coenzyme binding and specificity in glyceraldehyde-3-phosphate dehydrogenase. Biochemistry 35, 6064–6072 (1996)
Gardiennet-Doucet, C., Assfeld, X., Henry, B., Tekely, P.: Revealing successive steps of deprotonation of l-phosphoserine through 13C and 31P chemical shielding tensor fingerprints. J. Phys. Chem. A 110, 9137–9144 (2006)
Potrzebowski, M.J., Assfeld, X., Ganicz, K., Olejniczak, S., Cartier, A., Gardiennet, C., Tekely, P.: An experimental and theoretical study of the 13C and 31P chemical shielding tensors in solid O-phosphorylated amino acids. J. Am. Chem. Soc. 125, 4223–4232 (2003)
Paulus, E.F., Grabley, S.: Molecular and crystal structure of l-phosphinothricin (2-ammonio-4-methylphosphinico-butyrate). Z. Kristallogr. Cryst. Mater. 160, 63–68 (1982)
Melchior, A., Gaillard, C., Gràcia Lanas, S., Tolazzi, M., Billard, I., Georg, S., Sarrasin, L., Boltoeva, M.: Nickel(II) complexation with nitrate in dry [C4mim][Tf2N] ionic liquid: A spectroscopic, microcalorimetric, and molecular dynamics study. Inorg. Chem. 55, 3498–3507 (2016)
Acknowledgements
The authors thank the National Natural Science Foundation of China (Grant No. 41573122) and China Academy of Engineering Physics for financial support. The authors thank Jie Shen at Tianjin Institute of Industrial Biotechnology, Chinese Academy of Sciences for the calorimetric titrations.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Li, Q., Liu, B., Mu, W. et al. Protonation States of Glufosinate in Aqueous Solution. J Solution Chem 47, 705–714 (2018). https://doi.org/10.1007/s10953-018-0751-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-018-0751-y