Abstract
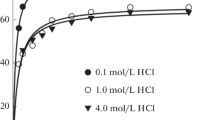
The fundamental properties and extraction capability of an ionic liquid (IL), trioctylammonium nitrate ([HTOA][NO3]), for PdII and PtIV, are investigated. At room temperature, [HTOA][NO3] is a solid (melting point: 30.7 °C), but it becomes a liquid (melting point: 16.7 °C) when saturated with water. Water-saturated [HTOA][NO3] exhibits a viscosity of 267.1 mPa·s and an aqueous solubility of 2.821 × 10−4 mol·dm−3 at 25 °C, and can be used as an extraction solvent without dilution. [HTOA][NO3] exhibits an extremely high extraction capability for PdII and PtIV in dilute hydrochloric acid (0.1–2 mol·dm−3 HCl); the distribution ratio reaches 3 × 104 for both the metals. From electrospray ionization mass spectrometry analysis, the species extracted in the IL phase are [PdCl3]− and [PdCl2(NO3)]− for PdII and [PtCl6]2− and [PtCl5]− for PtIV. A majority of the other transition metals are considerably less or marginally extracted into [HTOA][NO3] from a 0.1 mol·dm−3 hydrochloric acid solution. The extraction capacity of [HTOA][NO3] is greater than that of other hydrophobic ILs such as [HTOA]Cl and bis(trifluoromethanesulfonyl)imide-based ILs. The metals extracted into the IL phase are quantitatively back-extracted using an aqueous solution containing thiourea and nitric acid. By controlling the thiourea concentration and shaking time, PdII and PtIV are mutually separated to some extent in the back extraction process. The IL phase used for the back extraction can be reused for the forward extraction of these metals after scrubbing it with an aqueous nitric acid solution.




Similar content being viewed by others
References
PGM Market Report May 2017. Johnson Matthey Precious Metals Management, Royston (2017)
Fornalczyk, A., Saternus, M.: Removal of platinum group metals from the used auto catalytic converters. Metalurgija 48, 133–136 (2009)
Bernardis, F.L., Grant, R.A., Sherrington, D.C.: A review of methods of separation of the platinum-group metals through their chloro-complexes. React. Funct. Polym. 65, 205–217 (2005)
Cox, M.: Solvent extraction in hydrometallurgy. In: Rydberg, J., Cox, M., Musikas, C., Choppin, G.R. (eds.) Solvent Extraction Principles and Practice, 2nd edn, pp. 454–503. CRC/Taylor and Francis, Boca Raton (2004)
De Los Rios, A.P., Hernandez-Fernandez, F.J. (eds.): Ionic Liquids in Separation Technology. Elsevier, Amsterdam (2014)
Dai, S., Ju, Y.H., Barnes, C.E.: Solvent extraction of strontium nitrate by a crown ether using room-temperature ionic liquids. J. Chem. Soc. Dalton Trans. 1999, 1201–1202 (1999)
Visser, A.E., Swatloski, R.P., Griffin, S.T., Hartman, D.H., Rogers, R.D.: Liquid/liquid extraction of metal ions in room temperature ionic liquids. Sep. Sci. Technol. 36, 785–804 (2001)
Carda-Broch, S., Berthod, A., Armstrong, D.W.: Solvent properties of the 1-butyl-3-methylimidazolium hexafluorophosphate ionic liquid. Anal. Bioanal. Chem. 375, 191–199 (2003)
Vidal, S.T.M., Correia, M.J.N., Marques, M.M., Ismael, M.R., Reis, M.T.A.: Studies on the use of ionic liquids as potential extractants of phenolic compounds and metal ions. Sep. Sci. Technol. 39, 2155–2169 (2005)
Khachatryan, K.S., Smirnova, S.V., Torocheshnikova, I.I., Shvedene, N.V., Formanovsky, A.A., Pletnev, I.V.: Solvent extraction and extraction-voltammetric determination of phenols using room temperature ionic liquid. Anal. Bioanal. Chem. 381, 464–470 (2005)
Vijayaraghavan, R., Vedaraman, N., Surianarayanan, M., MacFarlane, D.R.: Extraction and recovery of azo dyes into an ionic liquid. Talanta 69, 1059–1062 (2006)
Pei, Y.C., Wang, J.J., Xuan, X.P., Fan, J., Fan, M.: Factors affecting ionic liquids based removal of anionic dyes from water. Environ. Sci. Technol. 41, 5090–5095 (2007)
de los Rios, A.P., Hernandez-Fernandez, F.J., Lozano, L.J., Sanchez, S., Moreno, J.I., Godinez, C.: Removal of metal ions from aqueous solutions by extraction with ionic liquids. J. Chem. Eng. Data 55, 605–608 (2010)
Katsuta, S., Nakamura, K., Kudo, Y., Takeda, Y., Kato, H.: Partition behavior of chlorophenols and nitrophenols between hydrophobic ionic liquids and water. J. Chem. Eng. Data 56, 4083–4089 (2011)
Katsuta, S., Nakamura, K., Kudo, Y., Takeda, Y.: Mechanisms and rules of anion partition into ionic liquids: phenolate ions in ionic liquid/water biphasic systems. J. Phys. Chem. B 116, 852–859 (2012)
Watanabe, Y., Katsuta, S.: Distribution of a monovalent anion in various ionic liquid/water biphasic systems: relationship of the distribution ratio of picrate ions with the aqueous solubility of ionic liquids. J. Chem. Eng. Data 59, 696–701 (2014)
Katsuta, S., Watanabe, Y., Araki, Y., Kudo, Y.: Extraction of gold(III) from hydrochloric acid into various ionic liquids: relationship between extraction efficiency and aqueous solubility of ionic liquids. ACS Sustain. Chem. Eng. 4, 564–571 (2016)
Hamamoto, T., Okai, M., Katsuta, S.: The laws governing ionic liquid extraction of cations: partition of 1-ethylpyridinium monocation and paraquat dication in ionic liquid/water biphasic systems. J. Phys. Chem. B 119, 6317–6325 (2015)
Seeley, F.G., Crouse, D.J.: Extraction of metals from chloride solutions with amines. J. Chem. Eng. Data 11, 424–429 (1966)
Galan, B., Urtiaga, A.M., Alonso, A.I., Irabien, J.A., Ortiz, M.I.: Extraction of anions with Aliquat 336: chemical equilibrium modeling. Ind. Eng. Chem. Res. 33, 1765–1770 (1994)
Lee, M.-S., Lee, K.-J., Oh, Y.-J.: Solvent extraction equilibria of FeCl3 from hydrochloric acid solution. Mater. Trans. 45, 2364–2368 (2004)
Jha, M.K., Gupta, D., Lee, J.-C., Kumar, V., Jeong, J.: Solvent extraction of platinum using amine based extractants in different solutions: a review. Hydrometallurgy 142, 60–69 (2014)
Cieszynska, A., Wisniewski, M.: Extraction of palladium(II) from chloride solutions with Cyphos® IL 101/toluene mixtures as novel extractant. Sep. Purif. Technol. 73, 202–207 (2010)
Wei, W., Cho, C.-W., Kim, S., Song, M.-H., Bediako, J.K., Yun, Y.-S.: Selective recovery of Au(III), Pt(IV), and Pd(II) from aqueous solutions by liquid–liquid extraction using ionic liquid Aliquat-336. J. Mol. Liq. 216, 18–24 (2016)
Fontàs, C., Salvadó, V., Hidalgo, M.: Solvent extraction of Pt(IV) by Aliquat 336 and its application to a solid supported liquid membrane system. Solvent Extr. Ion Exch. 17, 149–162 (1999)
Papaiconomou, N., Lee, J.-M., Salminen, J., von Stosch, M., Prausnitz, J.M.: Selective extraction of copper, mercury, silver, and palladium ions from water using hydrophobic ionic liquids. Ind. Eng. Chem. Res. 47, 5080–5086 (2008)
Stojanovic, A., Kogelnig, D., Fischer, L., Hann, S., Galanski, M., Groessl, M., Krachler, R., Keppler, B.K.: Phosphonium and ammonium ionic liquids with aromatic anions: synthesis, properties, and platinum extraction. Aust. J. Chem. 63, 511–524 (2010)
Katsuta, S., Yoshimoto, Y., Okai, M., Takeda, Y., Bessho, K.: Selective extraction of palladium and platinum from hydrochloric acid solutions by trioctylammonium-based mixed ionic liquids. Ind. Eng. Chem. Res. 50, 12735–12740 (2011)
Génand-Pinaz, S., Papaiconomou, N., Leveque, J.-M.: Removal of platinum from water by precipitation or liquid–liquid extraction and separation from gold using ionic liquids. Green Chem. 15, 2493–2501 (2013)
Yang, Y., Kubota, F., Baba, Y., Kamiya, N., Goto, M.: One step effective separation of platinum and palladium in an acidic chloride solution by using undiluted ionic liquids. Solvent Extr. Res. Dev. Jpn. 21, 129–135 (2014)
Tong, Y., Wang, C., Huang, Y., Yang, Y.: Extraction and stripping of platinum from hydrochloric acid medium by mixed imidazolium ionic liquids. Ind. Eng. Chem. Res. 54, 705–711 (2015)
Papaiconomou, N., Svecova, L., Bonnaud, C., Cathelin, L., Billard, I., Chainet, E.: Possibilities and limitations in separating Pt(IV) from Pd(II) combining imidazolium and phosphonium ionic liquids. Dalton Trans. 44, 20131–20138 (2015)
Kasahara, I., Kanai, M., Taniguchi, M., Kakeba, A., Hata, N., Taguchi, S., Goto, K.: Bis[2-(5-bromo-2-pyridylazo)-5-(N-propyl-N-sulphopropylamino)phenolato]cobaltate(III) as a counter ion for the extraction and spectrophotometric determination of long-chain quaternary ammonium salts and tertiary alkylamines in the presence of each other. Anal. Chim. Acta 219, 239–245 (1989)
Saito, G., Sugimoto, K., Hagino, K.: Spectrophotometric determination of nitrate and/or nitrite using brucine sulfate. Bunseki Kagaku 20, 542–549 (1971)
Nakamura, K., Kudo, Y., Takeda, Y., Katsuta, S.: Partition of substituted benzenes between hydrophobic ionic liquids and water: evaluation of interactions between substituents and ionic liquids. J. Chem. Eng. Data 56, 2160–2167 (2011)
Tokuda, H., Tsuzuki, S., Susan, M.A.B.H., Hayamizu, K., Watanabe, M.: How ionic are room-temperature ionic liquids? An indicator of the physicochemical properties. J. Phys. Chem. B 110, 19593–19600 (2006)
Kakiuchi, T., Nishi, N.: Ionic liquid|water interface: a new electrified system for electrochemistry. Electrochemistry 74, 942–948 (2006)
Riddick, J.A., Bunger, W.B., Sakano, T.K.: Organic Solvents, 4th edn. Wiley, New York (1986)
Högfeldt, E. (ed.): Stability Constants of Metal-Ion Complexes Part A: Inorganic Ligands. Pergamon Press, Oxford (1982)
Kondo, K., Ourachi, T., Kaneiwa, T., Matsumoto, M.: Solvent extraction of precious metals with quaternary ammonium salts and its application to preparation of metal particles. Solvent Extr. Res. Dev. Jpn. 7, 176–184 (2000)
Acknowledgements
The authors thank Ms. Sayaka Kado (Center for Analytical Instrumentation, Chiba University) for her technical support on mass spectrometry. The authors also thank Enago (www.enago.jp) for the English language review. This work was financially supported by JSPS KAKENHI Grant Number JP26410145.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Katsuta, S., Tamura, J. Extraction of Palladium(II) and Platinum(IV) from Hydrochloric Acid Solutions with Trioctylammonium Nitrate Ionic Liquid without Dilution. J Solution Chem 47, 1293–1308 (2018). https://doi.org/10.1007/s10953-018-0745-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-018-0745-9