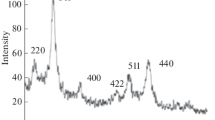
Abstract
Fe3O4 particles have been synthesized using co-precipitation method employing HCl and NH4OH as dissolving and precipitating agents, respectively. The particles synthesized without and with polyethylene-glycol (PEG)-400 as template having the particle size <50 nm were dissolved in tetra-methyl-ammonium-hydroxide (TMAH) surfactant to form a magnetic fluid, which was then spin-coated onto the aluminum substrate. The results of coating were characterized for studies covering the particles distribution on the surface, their morphologies, and magnetic properties.






Similar content being viewed by others
References
Vijayakumar, R., Koltypin, Y., Felner, I., Gedanken, A.: Sonochemical synthesis and characterization of pure nanometer-sized Fe3O4 particles. Mater. Sci. Eng. A 286(1), 101–105 (2000). doi:10.1016/S0921-5093(00)00647-X
Fan, R., Chen, X.H., Gui, Z., Liu, L., Chen, Z.Y.: A new simple hydrothermal preparation of nanocrystalline magnetite Fe3O4. Mater. Res. Bulletin 36, 497–502 (2001). doi:10.1016/S0025-5408(01)00527-X
Xu, J., Yang, H., Fu, W., Du, K., Sui, Y., Chen, J., Zeng, Y., Li, M., Zou, G.: Preparation and magnetic properties of magnetite nanoparticles by sol–gel method. J. Magn. Magn. Mater. 309(2), 307–311 (2007). doi:10.1016/j.jmmm.2006.07.037
Hong, R., Li, J., Wang, J., Li, H.: Comparison of schemes for preparing magnetic Fe3O4 nanoparticles. China Particuol. 5(1–2), 186–191 (2007). doi:10.1016/j.cpart.2007.01.011
Wei, Y., Han, B., Hu, X., Lin, Y., Wang, X., Deng, X.: 2011 Chinese materials conference synthesis of Fe3O4 nanoparticles and their magnetic properties. Proced. Eng. 27, 632–637 (2012). doi:10.1016/j.proeng.2011.12.498
Wu, J.H., Ko, S.P., Liu, H.L., Jung, M.-H., Lee, J.H., Ju, J.-S., Kim, Y.K.: Sub 5 nm Fe3O4 nanocrystals via coprecipitation method. Colloids Surf. A Physicochem. Eng. Asp. 313–314, 268–272 (2008). doi:10.1016/j.colsurfa.2007.04.108
Darminto, Cholishoh, M.N., Perdana, F.A., Baqiya, M.A., Mashuri, Cahyono, Y.: Triwikantoro: Preparing Fe3O4 nanoparticles from Fe2+ ions source by co-precipitation process in various pH. AIP Conf. Proc. 1415 (1), 234–237 (2011). doi:10.1063/1.3667264
Gnanaprakash, G., Mahadevan, S., Jayakumar, T., Kalyanasundaram, P., Philip, J., Raj, B.: Effect of initial pH and temperature of iron salt solutions on formation of magnetite nanoparticles. Mater. Chem. Phys. 103(1), 168–175 (2007). doi:10.1016/j.matchemphys.2007.02.011
Bica, D., Vékás, L., Avdeev, M.V., Marinică, O., Socoliuc, V., Bălăsoiu, M., Garamus, V.M.: Sterically stabilized water based magnetic fluids: synthesis, structure and properties. J. Magn. Magn. Mater. 311(1), 17–21 (2007). doi:10.1016/j.jmmm.2006.11.158
Hong, R.Y., Pan, T.T., Han, Y.P., Li, H.Z., Ding, J., Han, S.: Magnetic field synthesis of Fe3O4 nanoparticles used as a precursor of ferrofluids. J. Magn. Magn. Mater. 310(1), 37–47 (2007). doi:10.1016/j.jmmm.2006.07.026
Hong, R.Y., Zhang, S.Z., Han, Y.P., Li, H.Z., Ding, J., Zheng, Y.: Preparation, characterization and application of bilayer surfactant-stabilized ferrofluids. Powder Technol. 170(1), 1–11 (2006). doi:10.1016/j.powtec.2006.08.017
Răcuciu, M.: Synthesis protocol influence on aqueous magnetic fluid properties. Curr. Appl. Phys. 9(5), 1062–1066 (2009). doi:10.1016/j.cap.2008.12.003
Wang, Y.M., Cao, X., Liu, G.H., Hong, R.Y., Chen, Y.M., Chen, X.F., Li, H.Z., Xu, B., Wei, D.G.: Synthesis of Fe3O4 magnetic fluid used for magnetic resonance imaging and hyperthermia. J. Magn. Magn. Mater. 323(23), 2953–2959 (2011). doi:10.1016/j.jmmm.2011.05.060
Yangde, Z., Zhaowu, Z., Weihua, Z., Xingyan, L., Zhenfa, L., Jun, L., Jianfeng, X., Yulin, L., Tiehui, H., Yifeng, P.: The roles of hydrochloric acid and polyethylene glycol in magnetic fluids. J. Magn. Magn. Mater. 320(7), 1328–1334 (2008). doi:10.1016/j.jmmm.2007.10.014
Lian, S., Kang, Z., Wang, E., Jiang, M., Hu, C., Xu, L.: Convenient synthesis of single crystalline magnetic Fe3O4 nanorods. Solid State Commun. 127(9–10), 605–608 (2003). doi:10.1016/S0038-1098(03)00580-5
Andrade, Â.L., Fabris, J.D., Ardisson, J.D., Valente, M.A., Ferreira, J.M.F.: Effect of tetramethylammonium hydroxide on nucleation, surface modification and growth of magnetic nanoparticles. J. Nanomater. 2012, 10 (2012). doi:10.1155/2012/454759
Odenbach, S.: Ferrofluids—magnetically controlled suspensions. Colloids Surf. A Physicochem. Eng. Asp. 217(1–3), 171–178 (2003). doi:10.1016/S0927-7757(02)00573-3
Berger, P., Adelman, N.B., Beckman, K.J., Campbell, D.J., Ellis, A.B., Lisensky, G.C.: Preparation and properties of an aqueous ferrofluid. J. Chem. Educ. 76(7), 943 (1999). doi:10.1021/ed076p943
Taufiq, A., Sunaryono, Rachman Putra, E.G., Okazawa, A., Watanabe, I., Kojima, N., Pratapa, S.: Darminto: Nanoscale clustering and magnetic properties of MnxFe3−xO4 particles prepared from natural magnetite. J. Supercond. Novel Magn. 28(9), 2855–2863 (2015). doi:10.1007/s10948-015-3111-9
Köseoğlu, Y., Baykal, A., Toprak, M.S., Gözüak, F., Başaran, A.C., Aktaş, B.: Synthesis and characterization of ZnFe2O4 magnetic nanoparticles via a PEG-assisted route. J. Alloys Compd. 462(1–2), 209–213 (2008). doi:10.1016/j.jallcom.2007.07.121
Szklarska-Smialowska, Z.: Pitting corrosion of aluminum. Corros. Sci. 41(9), 1743–1767 (1999). doi:10.1016/S0010-938X(99)00012-8
Acknowledgments
This research was partially supported by “Hibah Kompetensi” research grant, 2013–2015, provided by DP2M, Ditjen Dikti, Ministry of Education and Culture, Indonesia. The use of A/MFM of Ambios Tech, Kuala Lumpur, was appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Triwikantoro, Baqiya, M.A., Heriyanto, T. et al. Nano-coating of Aluminum Surface Using Fe3O4-based Magnetic Fluids. J Supercond Nov Magn 30, 555–560 (2017). https://doi.org/10.1007/s10948-016-3813-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10948-016-3813-7