Abstract
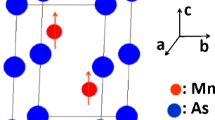
The self-consistent ab initio calculations, based on density functional theory (DFT) approach and using a full-potential linear augmented plane wave (FLAPW) method, are performed to investigate both electronic and magnetic properties of the MnHg compound. Polarized spin and spin–orbit coupling are included in calculations within the framework of the antiferromagnetic state between two adjacent Mn atoms. Magnetic moment considered to lie along (001) axis is computed. The antiferromagnetic and ferromagnetic energies of MnHg systems are obtained. Obtained data from ab initio calculations are used as input for the high-temperature series expansion (HTSE) calculations to compute other magnetic parameters. The exchange interactions between the magnetic atoms Mn–Mn in MnHg are given using the mean field theory. The HTSEs of the magnetic susceptibility of the magnetic moments in MnHg (m Mn) through the Ising model are given up to tenth order series in (x=J(Mn–Mn) / k B T). The Néel temperature (T N (K)) is obtained by HTSEs applied to the magnetic susceptibility series combined with the Padé approximant method. The critical exponent (γ) associated with the magnetic susceptibility is deduced as well.




Similar content being viewed by others
References
Verhoef, R., De Boer, F.R., Franse, J.J.M., Denissen, C.J.M., Jacobs, T.H., Buschow, K.H.J.: J. Magn. Magn. Mater. 80, 41 (1989)
Verhoef, R., Radwafiski, R.J., Franse, J.J.M.: J. Magn. Magn. Mater. 89, 176 (1992)
Ravindran, P., Kjekshus, A., Fjellvåg, H., James, P., Nordström, L., Johansson, B., Eriksson, O.: Phys. Rev. B 63, 144409 (2001)
Fujii, S., Ishida, S., Asano, S.: J. Phys. Soc. Jpn. 61, 709–713 (1992)
Masrour, R., Hlil, E.K., Hamedoun, M., Benyoussef, A., Mounkachi, O., El Moussaoui, H.: J. Magn. Magn. Mater. 361, 197 (2014)
Baker, G.A., Graves-Morris, P. (eds.): Padé approximants. Addison-Wesley, London (1981)
Blaha, P., Schwartz, K., Sorantin, P., Trikey, S.B.: Comput. Phys. Common. 59, 399 (1990)
Lihl, F.: Monaths. Chem. 86, 186 (1955)
Olés, A.: Report CEA, nr 28406, C1–787 (1964)
Holland, W.E., Brown, H.A.: Phys-Stat. Sol (a) 10, 249 (1972)
Moron, M.C., Phys, J.: Condens. Matter. 8, 11141 (1996)
Stanley, H.E., Kaplan, T.A.: Phys. Rev. Lett. 16, 981 (1966)
Yun, W.S., Odkhuu, D., Hong, S.C., Lee, J.I.: Thin Solid Films 519, 8355 (2011)
Kosterlitz, J.M., Thouless, D.J.: J. Phys. C 5, L124 (1973)
George, A., Baker, Jr., Nickel, B.G., Meiron, D.I.: Phys. Rev. B 17, 1365 (1978)
Carr, C.E.H.Y.: Phys. Rev. Lett. 39, 1558 (1977)
Ferrer, M., Moore, M.A., Wortis, M.: Phys. Rev. B 8, 5205 (1973)
Swendsen, R.H.: ibid 27, 391 (1983)
Wilson, K.G.: Phys. Rev. Lett. 28, 548 (1972)
Sardar, S., Chakraborty, K.G.: Phys. A 238, 317 (1997)
Van Dyke, J.P., Camp, W.J.: Phys. Rev. B 9, 3121 (1974)
Griffiths, R.B.: Phys. Rev. 158, 557 (1967)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Masrour, R., Hlil, E.K., Hamedoun, M. et al. Ab Initio, Mean Field and High-Temperature Series Expansion Calculation Study of Structural Stability and Magnetism of MnHg. J Supercond Nov Magn 28, 2501–2504 (2015). https://doi.org/10.1007/s10948-015-3055-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10948-015-3055-0