Abstract
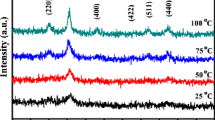
Polyethylene glycol (Mwt 400 and 10,000) stabilized Mn3O4 nanoparticles were synthesized via the thermal decomposition approach. Structural characteristics were evaluated by XRD, FT-IR, TGA, VSM, and TEM analysis. Crystallite sizes were calculated as 10±3 and 15±5 nm for PEG-400 and PEG-10,000 stabilized Mn3O4 NPs, respectively. FT-IR and TGA proved the presence of PEG on the surface of Mn3O4 NPs. Magnetization measurements carried out at room and low temperatures revealed the superparamagnetic nature of the Mn3O4 NPs. The blocking temperature was detected as 39 K and thermomagnetic irreversibility starts at 40 K. Both coercive field and saturation magnetization increases as temperature decreases below T C. Reduced magnetization compared to its bulk value has been explained by spin canting and presence of disordered spins. It was observed that the effects of different molecular weight PEG on the magnetic properties of the Mn3O4 nanoparticles are more or less the same.









Similar content being viewed by others
References
Zhou, L., Xu, J., Li, X., Wang, F.: Mater. Chem. Phys. 97, 137–141 (2006)
Durmus, Z., Kavas, H., Baykal, A., Toprak, M.S.: Cent. Eur. J. Chem. 7, 3 (2009) 555-560
Yamashita, Y., Mukai, K., Yoshinobu, J., Lippmaa, M., Kinoshita, T., Kawasaki, M.: Surf. Sci. 514, 54–60 (2002)
Zhang, Y.C., Qiao, T., Hu, X.Y.: J. Solid State Chem. 177, 4093–4097 (2004)
Niasari, M.S., Davar, F., Mazaheri, M.: Polyhedron 27, 3467–3471 (2008)
Yang, L.-X., Zhu, Y.-J., Tong, H., Wang, W.-W., Cheng, G.-F.: J. Solid State Chem. 179, 1225–1229 (2006)
Durmus, Z., Erdemi, H., Aslan, A., Toprak, M.S., Sozeri, H., Baykal, A.: Polyhedron 30, 419–426 (2011)
Karaoglu, E., Deligoz, H., Sozeri, H., Baykal, A., Toprak, M.S.: Nano-Micro Lett. 3(1), 25–33 (2011)
Durmus, Z., Tomaş, M., Baykal, A., Kavas, H., Toprak, M.S.: Synth. Reac. Inorg. Met.-Org. Nano-Met. Chem. 41, 768–773 (2011)
Kavas, H., Durmus, Z., Şenel, M., Kazan, S., Baykal, A., Toprak, M.S.: Polyhedron 29, 1375–1380 (2010)
Zhang, C., Wangler, B., Morgenstern, B., Zentgraf, H., Eisenhut, M., Untenecker, H., Kruger, R., Huss, R., Seliger, C., Semmler, W., Kiessling, F.: Langmuir 23, 1427–1434 (2007)
Durmus, Z., Kavas, H., Baykal, A., Sozeri, H., Alpsoy, L., Celik, S.Ü., Toprak, M.S.: J. Alloys Compd. 509, 2555–2561 (2011)
Kavas, H., Durmus, Z., Tanrıverdi, E., Şenel, M., Sozeri, H., Baykal, A.: J. Alloys Compd. 509, 5341–5348 (2011)
Du, J., Gao, Y.Q., Chai, L.L., Zou, G.F., Li, Y., Qian, Y.T.: Nanotechnology 17, 4923–4928 (2006)
Kavas, H., Baykal, A., Toprak, M.S., Koseoglu, Y., Sertkol, M., Aktas, B.: J. Alloys Compd. 479, 49–55 (2009)
Han, M., Ou, Y., Chen, W., Deng, L.: J. Alloys Compd. 474, 185–189 (2009)
Rahman, O., Mohapatra, S.C., Ahmad, S.: Mater. Chem. Phys. 132, 196–202 (2012)
Wejrzanowski, T., Pielaszek, R., Opaliniska, A., Matysiak, H., Lojkowski, W., Kurzydlowski, K.: J. Appl. Surf. Sci. 253, 204–208 (2006)
Ozkaya, T., Baykal, A., Kavas, H., Koseoglu, Y., Toprak, M.S.: Phys. Rev. B, Condens. Matter 403, 3760–3768 (2008)
Durmus, Z., Baykal, A., Kavas, H., Direkci, M., Toprak, M.S.: Polyhedron 28, 2119–2126 (2009)
Durmus, Z., Baykal, A., Kavas, H., Sozeri, H.: Physica B 406, 1114–1120 (2011)
Yang, H., Zhang, C., Shi, X., Hu, H., Du, X., Fang, Y., Ma, Y., Wu, H., Yang, S.: Biomaterials 31, 3667–3673 (2010)
Gozuak, F., Koseoglu, Y., Baykal, A., Kavas, H.: J. Magn. Magn. Mater. 321, 2170–2177 (2009)
Kumar, S., Singh, V., Aggarwal, S., Kumar Mandal, U., Kumar, R.K.: J. Phys. Chem. C 114, 6272–6278 (2010)
Zhao, D., Teng, P., Xu, Y., Xia, Q., Tang, J.: J. Alloys Compd. 502, 392–395 (2010)
Cai, W., Wan, J., Collo, J.: Int. Sci. 305, 366–370 (2007)
Wang, J., Zhang, C.R., Feng, J.: Int. J. Inorg. Mater. 20, 435–441 (2005)
Han, M., Ou, Y., Chen, W., Deng, L.: J. Alloys Compd. 474, 185–189 (2009)
Giri, J., Sriharsha, T., Asthana, S., Rao, T.K.G., Nigam, A.K., Bahadur, D.: J. Magn. Magn. Mater. 293, 55–61 (2005)
Unal, B., Durmus, Z., Baykal, A., Toprak, M.S., Sozeri, H., Bozkurt, A.: J. Alloys Compd. 509, 8199–8206 (2011)
Karaooğlu, E., Kavas, H., Baykal, A., Toprak, M.S., Sözeri, H.: Nano-Micro Lett. 3, 79–85 (2011)
Chang, Y.Q., Xu, X.Y., Luo, X.H., Chen, C.P., Yu, D.P.: J. Cryst. Growth 264, 232–238 (2004)
Jensen, G., Nielsen, O.: J. Phys. C, Solid State Phys. 7, 409–415 (1974)
Dwight, K., Meryuk, N.: Phys. Rev. 119, 1470–1475 (1960)
Gopalakrishnan, I.K., Bagkar, N., Ganguly, R., Kulshreshtha, S.K.: J. Cryst. Growth 280, 436–442 (2005)
Ebbe, T.R.S., Craik, D.J.: Magnetic Materials. Academic Press, New York (1969)
Kodama, R.H., Berkowitz, A.E., McNiff, E.J. Jr, Foner, S.: Phys. Rev. Lett. 77, 394–402 (1996)
Berkowitz, A.E., Lahut, J.A., Jacobs, I.S., Levinson, L.M., Forester, D.W.: Phys. Rev. Lett. 34, 594–598 (1975)
Durmus, Z., Erdemi, H., Aslan, A., Toprak, M.S., Sozeri, H., Baykal, A.: Polyhedron 30, 419–426 (2011)
Ünal, B., Durmus, Z., Baykal, A., Sözeri, H., Toprak, M.S., Alpsoy, L.: J. Alloys Compd. 505, 172–178 (2010)
Uzun, K., Çevik, E., Şenel, M., Sözeri, H., Baykal, A., Abasıyanık, M.F., Toprak, M.S.: J. Nanopart. Res. 12, 3057–3067 (2010)
Acknowledgements
The authors are thankful to the Fatih University Research Project Foundation (P50021104-B).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gunay, M., Sozeri, H. & Baykal, A. Polyol Approach for the Synthesis of Water Soluble Mn3O4 Nanoparticles Using PEG. J Supercond Nov Magn 25, 1929–1935 (2012). https://doi.org/10.1007/s10948-012-1514-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10948-012-1514-4