Abstract
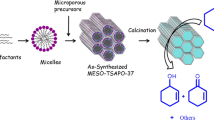
In order to prepare a highly active catalyst for the catalytic cracking of larger molecules, a novel micro-mesoporous silicoaluminophosphate composite (define as mesoporous SAPO-5) with hierarchical tri-modal pore size distributions has been firstly synthesized via post-synthetic method in acidic condition and subsequently characterized. Morphology control of the composite is attempted by adjusting pH value of the synthetic system. Three different morphologies of composite, including sphere-, rod- and net-like, are obtained in the different conditions. Possible mechanism for the formation of mesoporous SAPO-5 has been proposed. The mesoporous SAPO-5 exhibits higher cracking activity than conventional microporous SAPO-5 for cracking of 1, 3, 5-triisopropylbenzene (1, 3, 5-TIPB) under the same reaction conditions. The result indicates that the mesoporous SAPO-5 with hierarchical pore structure is favorable for catalytic cracking of large molecule. When the cumene as the reaction molecule, the microporous SAPO-5 catalyst exhibits higher conversion in catalytic cracking of cumene compared to the mesoporous SAPO-5, and the result may be attributed to that microporous SAPO-5 has much stronger acidity and specific selectivity than mesoporous SAPO-5 catalyst in catalytic cracking of cumene. Meanwhile, corresponding carbenium ion mechanism can account for the products formed during the whole reaction process.








Similar content being viewed by others
References
S.L. Qiu, M. Xue, G.S. Zhu, Chem. Soc. Rev. 43, 6116 (2014)
J. Pires, A. Carvalho, M.B. de Carvaho, Micropor. Mesopor. Mater. 43, 277 (2001)
C.S. Cundy, P.A. Cox, Chem. Rev. 103, 663 (2003)
B.M. Lok, C.A. Messina, R.L. Patton, R.T. Gajek, T.R. Cannan, E.M. Flanigen, J. Am. Chem. Soc. 106, 6092 (1984)
N. Danilina, F. Krumeich, J.A. van Bokhoven, J. Catal. 272, 37 (2010)
B. Tang, X.H. Lu, D. Zhou, P. Tian, Z.H. Niu, J.L. Zhang, X. Chen, Q.H. Xia, Catal. Commun. 31, 42 (2013)
H. Hajfarajollah, S. Askari, R. Halladj, React. Kinet. Mech. Catal. 111, 723 (2014)
I.I. Ivanova, E.E. Knyazeva, Chem. Soc. Rev. 42, 3671 (2013)
J.S. Beck, J.C. Vartuli, W.J. Roth, M.E. Leonowicz, C.T. Kresge, K.D. Schmitt, C.T.U. Chu, D.H. Olsen, E.W. Sheppard, S.B. McCullen, J.B. Higgins, J.L. Schlenker, J. Am. Chem. Soc. 114, 10834 (1992)
C.T. Kresge, M.E. Leonowicz, W.J. Roth, J.C. Vartuli, J.S. Beck, Nature 359, 710 (1992)
C. Perego, R. Millini, Chem. Soc. Rev. 42, 3956 (2013)
Y.H. Li, W. Luo, N. Qin, J.P. Dong, J. Wei, W. Li, S.S. Feng, J.C. Chen, J.Q. Xu, A.A. Elzatahry, M.H. Es-Saheb, Y.H. Deng, D.Y. Zhao, Angew. Chem. Int. Ed. 53, 9035 (2014)
L.X. Jia, X.Y. Sun, X.Q. Ye, C.L. Zou, H.F. Gu, Y. Huang, G.X. Niu, D.Y. Zhao, Micropor. Mesopor. Mater. 176, 16 (2013)
X.M. Zhang, L. Zhang, Q.H. Yang, J. Mater. Chem. A 2, 7546 (2014)
J. Qi, T.B. Zhao, F.Y. Li, G.D. Sun, X. Xu, C. Miao, H.W. Wang, X.M. Zhang, J. Porous Mater. 17, 177 (2010)
M.S. Aghakhani, A.A. Khodadadi, Sh Najafi, Y. Mortazavi, J. Ind. Eng. Chem. 20, 3037 (2014)
J. Qi, T.B. Zhao, X. Xu, F.Y. Li, G.D. Sun, C. Miao, H.W. Wang, Catal. Commun. 10, 1523 (2009)
J.S. Jin, C.Y. Peng, J.J. Wang, H.T. Liu, X.H. Gao, H.H. Liu, C.Y. Xu, Ind. Eng. Chem. Res. 53, 3406 (2014)
A.K. Singh, R. Yadav, A. Sakthivel, Micropor. Mesopor. Mater. 181, 166 (2013)
Y.L. Liu, L.Z. Wanga, J.L. Zhang, L.J. Chen, H.S. Xu, Micropor. Mesopor. Mater. 145, 150 (2011)
K. Utchariyajit, S. Wongkasemjit, Micropor. Mesopor. Mater. 135, 116 (2010)
R. Yadav, A.K. Singh, A. Sakthivel, Chem. Lett. 42, 1160 (2013)
H.Q. Yang, Z.C. Liu, H.X. Gao, Z.K. Xie, J. Mater. Chem. 20, 3227 (2010)
J.Y. Kim, J. Kim, S.T. Yang, W.S. Ahn, Fuel 108, 515 (2013)
S. Suyanta, F.I. Izul, J. Chem. Chem. Eng. 6, 531 (2012)
J. Qi, T.B. Zhao, X. Xu, F.Y. Li, G.D. Sun, J. Porous Mater. 18, 69 (2011)
W.S. Yang, B.Q. Zhang, X.F. Liu, Micropor. Mesopor. Mater. 117, 391 (2009)
K.H. Schnabel, G. Finger, J. Kornatowski, E. Löffler, C. Peuker, W. Pilz, Micropor. Mater. 11, 293 (1997)
H. Aguiar, J. Serra, P. González, B. León, J. Non-Cryst, Solids 355, 475 (2009)
K.M. Davis, M. Tomozawa, J. Non-Cryst, Solids. 201, 177 (1996)
R.M. Susarrey-Arce, R.G.P. Tiggelaar, B. Sanders, L. Geerdink, J.G.E. Lefferts, A. Gardeniers, van Houselt, J. Phys. Chem. C 117, 21936 (2013)
E.M. Flanigen, H. Khatami, H.A. Szymanski, Adv. Chem. Ser. 101, 201 (1971)
Y.F. Yeong, A.Z. Abdullah, A.L. Ahmad, S. Bhatia, Micropor. Mesopor. Mater. 123, 129 (2009)
M.T. Zhao, K. Deng, L.C. He, Y. Liu, G.D. Li, H.J. Zhao, Z.Y. Tang, J. Am. Chem. Soc. 136, 1738 (2014)
W. Xu, C. Lin, H.A. Liu, H.B. Yu, K. Tao, S.H. Zhou, RSC Adv. 5, 23981 (2015)
K. Zhao, J. Qi, S.L. Zhao, H.J. Tang, H.J. Yin, L.B. Zong, L. Chang, Y. Gao, R.B. Yu, Z.Y. Tang, Chin. J. Catal. 36, 261 (2015)
J. Qi, K. Zhao, G.D. Li, Y. Gao, H.J. Zhao, R.B. Yu, Z.Y. Tang, Nanoscale 6, 4072 (2014)
J. Qi, J. Chen, G.D. Li, S.X. Li, Y. Gao, Z.Y. Tang, Energy Environ. Sci. 5, 8937 (2012)
L. Zhu, F.S. Xiao, Z.T. Zhang, Y.Y. Sun, Y. Han, S.L. Qiu, Catal. Today 68, 209 (2001)
J. Xiao, J. Wei, Chem. Eng. Sci. 47, 1123 (1992)
R.R. Xu, W.Q. Pang, J.H. Yu, Q.S. Huo, J.S. Chen, Chemistry-Zeolites and Porous Materials, 1st edn. (Science Press, Beijing, 2004)
B. Adnadjevic, J. Vulkicevic, Z. Filipovic-Rojka, V. Markovic, Zeolites 10, 699 (1990)
M.R. Sazegar, A.A. Jalil, S. Triwahyono, R.R. Mukti, M. Aziz, M.A.A. Aziz, H.D. Setiabudi, N.H.N. Kamarudin, Chem. Eng. J. 240, 352 (2014)
L.P. Liu, F.Y. Jin, G. Xiong, H.Y. Long, X.S. Wang, J. Porous Mater. 20, 637 (2013)
S. Al-Khattaf, H. de Lasa, Appl. Catal. A Gen. 226, 139 (2002)
N.H.R. Annuara, A.A. Jalil, S. Triwahyono, N.A.A. Fatah, L.P. Teh, C.R. Mamat, Appl. Catal. A Gen. 475, 487 (2014)
H. Konno, T. Tago, Y. Nakasaka, G. Watanabe, T. Masuda, Appl. Catal. A Gen. 475, 127 (2014)
Acknowledgments
This work was supported by the 973 Program under Grant (No. 2003CB615802), the Foundation for State Key Laboratory of Multiphase Complex Systems (No. MPCS-2014-A-04 Y425016124 J. Q.), National Natural Science Foundation of China (No.51302266 Q. J.) and Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry (Q. J.).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jian Qi and Quan Jin have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qi, J., Jin, Q., Zhao, K. et al. Catalytic cracking of 1, 3, 5-triisopropylbenzene over silicoaluminophosphate with hierarchical pore structure. J Porous Mater 22, 1021–1032 (2015). https://doi.org/10.1007/s10934-015-9976-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-015-9976-y