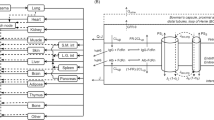
Abstract
Bortezomib is a reversible proteasome inhibitor with potent antineoplastic activity that exhibits dose- and time-dependent pharmacokinetics (PK). Proteasome-mediated bortezomib disposition is proposed as the primary source of its nonlinear and apparent nonstationary PK behavior. Single intravenous (IV) doses of bortezomib (0.25 and 1 mg/kg) were administrated to BALB/c mice, with blood and tissue samples obtained over 144 h, which were analyzed by LC/MS/MS. A physiologically based pharmacokinetic (PBPK) model incorporating tissue drug-target binding was developed to test the hypothesis of proteasome-mediated bortezomib disposition. The final model reasonably captured bortezomib plasma and tissue PK profiles, and parameters were estimated with good precision. The rank-order of model estimated tissue target density correlated well with experimentally measured proteasome concentrations reported in the literature, supporting the hypothesis that binding to proteasome influences bortezomib disposition. The PBPK model was further scaled-up to humans to assess the similarity of bortezomib disposition among species. Human plasma bortezomib PK profiles following multiple IV dosing (1.3 mg/m2) on days 1, 4, 8, and 11 were simulated by appropriately scaling estimated mouse parameters. Simulated and observed bortezomib concentrations after multiple dosing were in good agreement, suggesting target-mediated bortezomib disposition is likely for both mice and humans. Furthermore, the model predicts that renal impairment should exert minimal influence on bortezomib exposure in humans, confirming that bortezomib dose adjustment is not necessary for patients with renal impairment.





Similar content being viewed by others
References
Richardson PG, Mitsiades C, Hideshima T, Anderson KC (2006) Bortezomib: proteasome inhibition as an effective anticancer therapy. Annu Rev Med 57:33–47
Dick LR, Fleming PE (2010) Building on bortezomib: second-generation proteasome inhibitors as anti-cancer therapy. Drug Discov Today. 15:243–249
Lieu C, Chow L, Pierson AS, Eckhardt SG, O’Bryant CL, Morrow M, Tran ZV, Wright JJ, Gore L (2009) A phase I study of bortezomib, etoposide and carboplatin in patients with advanced solid tumors refractory to standard therapy. Invest New Drugs 27:53–62
Mateos MV (2012) How to maintain patients on long-term therapy: understanding the profile and kinetics of adverse events. Leuk Res 36(Suppl 1):S35–S43
Bross PF, Kane R, Farrell AT, Abraham S, Benson K, Brower ME, Bradley S, Gobburu JV, Goheer A, Lee SL, Leighton J, Liang CY, Lostritto RT, McGuinn WD, Morse DE, Rahman A, Rosario LA, Verbois SL, Williams G, Wang YC, Pazdur R (2004) Approval summary for bortezomib for injection in the treatment of multiple myeloma. Clin Cancer Res 10:3954–3964
Uttamsingh V, Lu C, Miwa G, Gan LS (2005) Relative contributions of the five major human cytochromes P450, 1A2, 2C9, 2C19, 2D6, and 3A4, to the hepatic metabolism of the proteasome inhibitor bortezomib. Drug Metab Dispos 33:1723–1728
Leveque D, Carvalho MC, Maloisel F (2007) Review. Clinical pharmacokinetics of bortezomib. In Vivo 21:273–278
Hemeryck A, Geerts R, Monbaliu J, Hassler S, Verhaeghe T, Diels L, Verluyten W, van Beijsterveldt L, Mamidi RN, Janssen C, De Coster R (2007) Tissue distribution and depletion kinetics of bortezomib and bortezomib-related radioactivity in male rats after single and repeated intravenous injection of 14 C-bortezomib. Cancer Chemother Pharmacol 60:777–787
Akhlaghi F, Monbaliu J, Kadambi V, Li Y (2009) Blood and plasma pharmacokinetics of bortezomib in relation to blood 20S proteasome activity after single and multiple dosing in cynomongolus monkeys. http://www.2009go-acoporg//acop2009 Accessed 9 Oct 2009
McConkey DJ, Zhu K (2008) Mechanisms of proteasome inhibitor action and resistance in cancer. Drug Resist Updates 11:164–179
Tanaka K, Ii K, Ichihara A, Waxman L, Goldberg AL (1986) A high molecular weight protease in the cytosol of rat liver. I. Purification, enzymological properties, and tissue distribution. J Biol Chem 261:15197–15203
Levy G (1994) Pharmacologic target-mediated drug disposition. Clin Pharmacol Ther 56:248–252
Mager DE (2006) Target-mediated drug disposition and dynamics. Biochem Pharmacol 72:1–10
Diao L, Meibohm B (2013) Pharmacokinetics and pharmacokinetic-pharmacodynamic correlations of therapeutic peptides. Clin Pharmacokinet 52:855–868
Singh J, Petter RC, Baillie TA, Whitty A (2011) The resurgence of covalent drugs. Nat Rev Drug Discov 10:307–317
Mager DE, Jusko WJ (2001) General pharmacokinetic model for drugs exhibiting target-mediated drug disposition. J Pharmacokinet Pharmacodyn 28:507–532
Gibiansky L, Gibiansky E, Kakkar T, Ma P (2008) Approximations of the target-mediated drug disposition model and identifiability of model parameters. J Pharmacokinet Pharmacodyn 35:573–591
Dua P, Hawkins E, Van der Graaf P (2015) A tutorial on target-mediated drug disposition (TMDD) models. CPT Pharmacomet Syst Pharmacol 4:324–337
Rowland M, Peck C, Tucker G (2011) Physiologically-based pharmacokinetics in drug development and regulatory science. Annu Rev Pharmacol Toxicol 51:45–73
Levy G, Mager DE, Cheung WK, Jusko WJ (2003) Comparative pharmacokinetics of coumarin anticoagulants L: physiologic modeling of S-warfarin in rats and pharmacologic target-mediated warfarin disposition in man. J Pharm Sci 92:985–994
Davies B, Morris T (1993) Physiological parameters in laboratory animals and humans. Pharm Res 10:1093–1095
Adams J, Palombella VJ, Sausville EA, Johnson J, Destree A, Lazarus DD, Maas J, Pien CS, Prakash S, Elliott PJ (1999) Proteasome inhibitors: a novel class of potent and effective antitumor agents. Cancer Res 59:2615–2622
Brown RP, Delp MD, Lindstedt SL, Rhomberg LR, Beliles RP (1997) Physiological parameter values for physiologically based pharmacokinetic models. Toxicol Ind Health 13:407–484
Kawai R, Mathew D, Tanaka C, Rowland M (1998) Physiologically based pharmacokinetics of cyclosporine A: extension to tissue distribution kinetics in rats and scale-up to human. J Pharmacol Exp Ther 287:457–468
D’Argenio DZ, Schumitzky A, Wang X (2009) ADAPT 5 user’s guide: pharmacokinetic/pharmacodynamic systems analysis software. Biomedical Simulation Resource, Los Angeles
Tsu C, Blank J, Garcia K, Liu J, Bruzzese F, Lee E, Cao Y, Bannerman B, Fitzgerald M, Fleming P, Ciavarri J, Hales P, Yu J, Yang Y, Berger A, Sintchak M, Kupperman E, Manfredi M, Dick L (2011) Beyond bortezomib: development of millennium’s next-generation proteasome inhibitors. Mol Cancer Ther. doi:10.1158/1535-7163TARG-11-C99
Reece DE, Sullivan D, Lonial S, Mohrbacher AF, Chatta G, Shustik C, Burris H 3rd, Venkatakrishnan K, Neuwirth R, Riordan WJ, Karol M, von Moltke LL, Acharya M, Zannikos P, Keith Stewart A (2011) Pharmacokinetic and pharmacodynamic study of two doses of bortezomib in patients with relapsed multiple myeloma. Cancer Chemother Pharmacol 67:57–67
Kupperman E, Lee EC, Cao Y, Bannerman B, Fitzgerald M, Berger A, Yu J, Yang Y, Hales P, Bruzzese F, Liu J, Blank J, Garcia K, Tsu C, Dick L, Fleming P, Yu L, Manfredi M, Rolfe M, Bolen J (2010) Evaluation of the proteasome inhibitor MLN9708 in preclinical models of human cancer. Cancer Res 70:1970–1980
Laplanche R, Meno-Tetang GM, Kawai R (2007) Physiologically based pharmacokinetic (PBPK) modeling of everolimus (RAD001) in rats involving non-linear tissue uptake. J Pharmacokinet Pharmacodyn 34:373–400
Chauhan D, Singh A, Brahmandam M, Podar K, Hideshima T, Richardson P, Munshi N, Palladino MA, Anderson KC (2008) Combination of proteasome inhibitors bortezomib and NPI-0052 trigger in vivo synergistic cytotoxicity in multiple myeloma. Blood 111:1654–1664
Williamson MJ, Blank JL, Bruzzese FJ, Cao Y, Daniels JS, Dick LR, Labutti J, Mazzola AM, Patil AD, Reimer CL, Solomon MS, Stirling M, Tian Y, Tsu CA, Weatherhead GS, Zhang JX, Rolfe M (2006) Comparison of biochemical and biological effects of ML858 (salinosporamide A) and bortezomib. Mol Cancer Ther 5:3052–3061
Kagan L, Abraham AK, Harrold JM, Mager DE (2010) Interspecies scaling of receptor-mediated pharmacokinetics and pharmacodynamics of type I interferons. Pharm Res 27:920–932
Nix D, Press R, Wehrman T, Aide R (2003) Tissue distribution and mass balance of bortezomib (Velcade™) in non-human primates. AAPS PharmSci 5:4
Leal TB, Remick SC, Takimoto CH, Ramanathan RK, Davies A, Egorin MJ, Hamilton A, LoRusso PA, Shibata S, Lenz HJ, Mier J, Sarantopoulos J, Mani S, Wright JJ, Ivy SP, Neuwirth R, von Moltke L, Venkatakrishnan K, Mulkerin D (2011) Dose-escalating and pharmacological study of bortezomib in adult cancer patients with impaired renal function: a National Cancer Institute Organ Dysfunction Working Group Study. Cancer Chemother Pharmacol 68:1439–1447
Lu C, Gallegos R, Li P, Xia CQ, Pusalkar S, Uttamsingh V, Nix D, Miwa GT, Gan LS (2006) Investigation of drug-drug interaction potential of bortezomib in vivo in female Sprague-Dawley rats and in vitro in human liver microsomes. Drug Metab Dispos 34:702–708
Levy G (1966) Kinetics of pharmacologic effects. Clin Pharmacol Ther 7:362–372
Lappin G, Kuhnz W, Jochemsen R, Kneer J, Chaudhary A, Oosterhuis B, Drijfhout WJ, Rowland M, Garner RC (2006) Use of microdosing to predict pharmacokinetics at the therapeutic dose: experience with 5 drugs. Clin Pharmacol Ther 80:203–215
Acknowledgments
This work was supported by National Institutes of Health [Grant GM57980]. We thank Ms. Donna Ruszaj for her valuable technical assistance in developing the LC/MS/MS assay.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest that would influence this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, L., Mager, D.E. Physiologically-based pharmacokinetic modeling of target-mediated drug disposition of bortezomib in mice. J Pharmacokinet Pharmacodyn 42, 541–552 (2015). https://doi.org/10.1007/s10928-015-9445-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-015-9445-x