Abstract
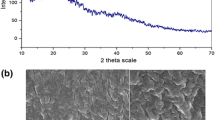
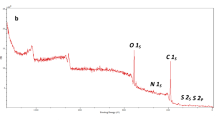
Chitosan used on dyes adsorption when in film form shows limitations due to its low regeneration capacity. In this work, chitosan films were modified using vanadium ions to improve its regeneration capacity on the dye adsorption. Films were characterized and analyzed by FT-IR, XRD and DSC. Adsorption assays were performed using chitosan-vanadate films (CVF) to remove the Reactive Black 5 dye in aqueous solution. The adsorption was favored by the pH decrease from 8.0 to 4.0. The equilibrium data were best fitted by Langmuir model, with qm of 522 mg g−1 at 298 K. The negative value of ΔH0 (−9.91 kJ mol−1) showed an exothermic operation and the value of ΔS0 was positive (0.0705 kJ mol−1 K−1), suggesting an increase in randomness at the solid/solution interface. The kinetics was represented by pseudo-first order model and around 80 min occurred a reduction in the adsorption rates, due to the mass transfer mechanism. The most appropriate eluent to remove the RB5 from CVF was NH4OH 0.01 kg mol L−1. The CVF used to the dye adsorption were regenerated and reutilized five times, and the adsorption capacity was around 80% of the initial value after the last cycle.







Similar content being viewed by others
References
Ahmad MZ, Rahman NK (2011) Chem Eng J 170:154
Ali I, Aboul-Enein HY (2006) Instrumental methods in metal ions speciation: chromatography, capillary electrophoresis and electrochemistry, Taylor & Francis Ltd., New York
Ali I, Aboul-Enein HY, Gupta VK (2009) Nano chromatography and capillary electrophoresis: pharmaceutical and environmental analyses. Wiley, Hoboken
Khan TA, Sharma S, Ali I (2011) J Toxicol Environ Health Sci 10:286
Crini G, Badot PM (2008) Prog Polym Sci 33:399
Cheng JS, Du J, Zhu W (2012) Carbohydr Polym 88:61
Wan M, Kan C, Rogel BD, Dalida MLP (2010) Carbohydr Polym 80:891
Wan Ngah WS, Teong LC, Hanafiah MAKM. (2011) Carbohydr Polym 83:1446
Reddy MCS, Sivaramakrishna L, Reddy AV (2012) J Hazard Mater 203–204:118
Ali I, Gupta VK (2006) Nat Protoc 1:2661
Ali I (2012) Chem Rev 112:5073
Ali I (2010) Sep Purif Rev 39:95
Ali I, Asim M, Khan TA (2012) J Environ Manag 113:170
Ali I (2014) Sep Purif Rev 43:175
Muzzarelli RAA, Boudrant J, Meyer D, Manno N, DeMarchis M, Paoletti MG (2012) Carbohydr Polym 87:995
Ali I, Al-Othman ZA, Alwarthan A (2017) J Mol Liq 236:205
Ali I, Al-Othman ZA, Alwarthan A (2016) J Mol Liq 224:171
Ali I, Al-Othman ZA, Alwarthan A (2016) J Mol Liq 221:1168
Ali I, Al-Othman ZA, Alwarthan A (2016) J Mol Liq 219:858
Ali I, Al-Othman ZA, Alharbi OML (2016) J Mol Liq 218:465
Vieira RS, Oliveira MLM, Guibal E, Rodríguez–Castellón E, Beppu MM (2011) Coll Surf A 374:108
Rêgo TV, Cadaval TRS Jr, Dotto GL, Pinto LAA (2013) J Coll Interface Sci 411:27
Singh V, Pandey S, Singh SK, Sanghi R (2008) Sol-Gel Sci Technol 47:58
Cadaval TRS Jr, Dotto GL, Seus ER, Mirlean N, Pinto LAA (2015) Desalin Water Treat 57:16583
Moura CM, Moura JM, Soares NM, Pinto LAA (2011) Chem Eng Process 50:351
Dotto GL, Souza VC, Moura JM, Moura CM, Pinto LAA (2011) Dry Technol 29:1784
Weska RF, Moura JM, Batista LM, Rizzi J, Pinto LAA (2007) J Food Eng 80:749
Dotto GL, Moura JM, Cadaval TRS, Pinto LAA (2013) Chem Eng J 214:8
ASTM (2000) Standard test methods for tensile properties of thin plastic sheeting. Standard D882-02, Annual book of ASTM, pp 162–170
Silverstein RM, Webster FX, Kiemle DJ (2007) Spectrometric identification of organic compounds. Wiley, New York
Rivero S, García MA, Pinotti A (2010) Innov Food Sci EmergTechnol 11:369
Cheng M, Deng J, Yang F, Gong Y, Zhao N, Zhang X (2003) Biomaterials 24:2871
Ruthven DM (1984) Principles of adsorption and adsorption process. Wiley, New York
Liu Y (2009) J Chem Eng Data 54:1981
Esquerdo VM, Cadaval TRS Jr, Dotto GL, Pinto LAA (2014) J Colloid Interface Sci 424:7
Li W, Qi L, Aiqin W (2010) Polym Bull 65:961
Dotto GL, Pinto LAA (2011) J Hazard Mater 187:164
Martinez JM (2000) J Comput Appl Math 124:97
Guibal E (2004) Sep Purif Technol 38:43
Moura JM, Farias BS, Rodrigues DAS, Moura CM, Dotto GL, Pinto LAA (2015) J Polym Environ 23:470
Maji TK, Baruah I, Dube S, Hussain MR (2007) Bioresour Technol 98:840
Cui L, Jia J, Guo Y, Liu Y, Zhu P (2014) Carbohydr Polym 99:31
Liu Z, Ge X, Lu Y, Dong S, Zhao Y, Zeng M (2012) Food Hydrocolloid 26:311
Dotto GL, Cunha JM, Calgaro CO, Tanabe EH, Bertuol DA (2015) J Hazard Mater 295:29
Kousksou T, Jamil A, El Omari K, Zeraouli Y, Le Guer Y (2011) Thermochim Acta 519:59
Homer S, Kelly M, Day L (2014) Carbohydr Polym 108:1
Wu FC, Tseng RL, Juang RS (2000) J Hazard Mater B73:63
Guzmán J, Saucedo I, Navarro R, Revilla J, Guibal E (2002) Langmuir 18:1567
Cadaval TRS Jr, Camara AS, Dotto GL, Pinto LAA (2013) Desalin WaterTreat 51:7690
Machado FM, Bergmann CP, Lima EC, Royer B, Souza FE, Jauris IM, Calvete T, Fagan SB (2012) Chem Phys 14:11139
Bakhtiari N, Azizian S (2015) J Mol Liq 206:114
Acknowledgements
The authors thank CAPES/Brazil (Coordination for the Improvement of Higher Education Personnel) and CNPq/Brazil (National Council for Scientific and Technological Development) for the financial support. The authors too thank CEME-SUL/FURG/Brazil (Electron Microscopy Center of South/Federal University of Rio Grande/RS/Brazil) due to the microscopy images.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rodrigues, D.A.S., Moura, J.M., Dotto, G.L. et al. Preparation, Characterization and Dye Adsorption/Reuse of Chitosan-Vanadate Films. J Polym Environ 26, 2917–2924 (2018). https://doi.org/10.1007/s10924-017-1171-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-017-1171-6