Abstract
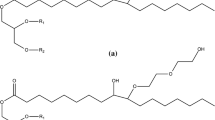
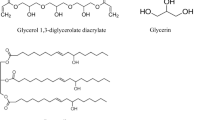
Biobased polyols were synthesized from rapeseed oil (RO) with diethanolamine (DEA), triethanolamine (TEA) and glycerol (GL) at different molar ratios. The structures of the synthesized polyols were analyzed using FTIR-ATR spectroscopy. Polyurethane (PU) networks from RO/DEA polyols and polymeric MDI showed higher tensile strength, modulus and hardness, but their elongation at break decreased, compared to the case of the PU obtained from RO/TEA and RO/GL polyols. The tensile strength and modulus of PU networks increased with increasing PU cohesion energy density (CED) and decreasing molecular weight between crosslinks M c . From the thermogravimetric analysis and its derivative thermograms, at the first stage of destruction (below 5 % weight loss) in the air and inert atmosphere, the PU obtained from RO polyols were ranked in the following order: PU RO/GL > PU RO/TEA > PU RO/DEA, and their thermostability was higher than that of the PU based on propylene oxide.









Similar content being viewed by others
References
Petrović ZS (2008) Polyurethanes from vegetable oils. Polym Rev 48:109–155
Petrović ZS, Yang L, Zlatanić A, Zhang W, Javni I (2007) Network structure and properties of polyurethanes from soybean oil. J Appl Polym Sci 105:2717–2727
Javni I, Zhang W, Petrovics Z (2004) Soybean oil based polyisocyanurate rigid foams. Polym Environ 12:123–129
Hu YH, Gao Y, Wang DN, Hu CP, Zu S, Vanoverloop L, Randal DJ (2002) Rigid polyurethane foam prepared from a rape seed oil based polyol. J Appl Polym Sci 84:591–597
Badri KH, Ahmad SH, Zakaria S (2001) Production of high-functionality RBD palm kernel oil-based polyether polyol. J Appl Polym Sci 81:384–389
Yadav S, Zafar F, Hasnat A, Ahmad S (2009) Poly(urethane fatty amide) resin from linseed oil: a renewable recourse. Prog Org Coat 64:27–32l
Petrović ZS, Cvetković I, Hong DP, Wan X, Zhang W, Abraham T, Malsam J (2008) Polyester polyols and polyurethanes from ricinoleic acid. J Appl Polym Sci 108:1184–1190
Guo A, Javni I, Petrovic Z (2000) Rigid polyurethane foams based on soybean oil. J Appl Polym Sci 77:467–473
Stirna U, Cabulis U, Beverte I (2008) Water-blown polyisocyanurate foams from vegetable oil polyols. J Cell Plast 44(2):139–160
Badri KH, Othman Z, Ahmad SH (2004) Rigid polyurethanes foams from oil palm resources. J Mater Sci 39:5541–5542
Lee CS, Ooi TL, Chuceh CH, Ahmad S (2007) Rigid polyurethane foams production from palm oil-based epoxidized diethanolamine. J Am Oil Chem Soc 84:1161–1167
Stirna U, Lazdina B, Vilsone Dz, Deme L, Yakushin V, Lopez MJ, Vargas-Garcia MC, Suarez-Estrella F, Moreno J (2011) Structure and properties of the polyurethane and polyurethane foam synthesized from castor oil polyols. In: Proceedings of the 3th international conference of Biofoams, Italy, 255–261
Feairheller SH, Bistline RG, Bilyk A Jr, Dudley RL, Kozempel MF, Haas MJ (1994) A novel technique for the preparation of secondary fatty amides. III. Alkanolamides, diamides and arylamides. J Am Oil Chem Soc 71:863–866
Javni I, Petrović ZS, Guo A, Fuller R (2000) Thermal stability of polyurethanes based on vegetable oils. J Appl Polym Sci 77:1723–1734
Wolf P, Farum Larsen H (1972) Polyurethane prepared from glycerin reaction products. USP 3(637):539
Feairheller SH, Bistline RG, Bilyk A Jr, Dudley RL, Kozempel MF, Haas MJ (1994) A novel technique for the preparation of secondary fatty amides. III. Alkanolamides, diamides and arylamides. J Am Oil Chem Soc 71:863–866
Lee CS, Lee SC (2010) Characterization of epoxidation and non–epoxidized fatty diethanolamides by high performance liquid chromatography and gas chromatography. Am J Pharm Techn 5(3):133–138
Aranguren MI, Racz I, Marcowich NE (2007) Microfoams based on castor oil polyurethanes and vegetable fibers. J Appl Polym Sci 105(5):2791–2800
Campanella A, Bonnaillie LM, Wool RP (2009) Polyurethane foams from soyoil-based polyols. J Appl Polym Sci 112(4):2567–2578
Seymour RW, Estes GM, Cooper SL (1970) Infrared studies of segmented polyurethane elastomers. I Hydrogen bonding. Macromolecules 3(5):579–583
Güner FS, Yağcı Y, Erciyes AT (2006) Polymers from triglyceride oils. Prog Polym Sci 31:633–670
David DJ, Stanley MP (1974) Analytical chemistry of polyurethane, part III: high polymers. Wiley Interscience, New York, pp 16–365
Fedors RF (1974) A method for estimating both the solubility parameters and molar volumes of liquids. Polymer Eng Sci 14(2):147–154
King JW (1995) Determination of the solubility parameter of soybean oil by inverse gas chromatography. LWT Food Sci Technol 28(2):190–195
Askadskii AA (1976) Influence of chemical structure on the properties of polymers. Pure Appl Chem 46:19–27
Kroll H, Nadeau H (1957) The chemistry of lauric acid diethanolamine condensation products. J Am Oil Chem Soc 34:323–326
Lee HS, Wang YK (1987) Spectroscopic analysis of phase separation behavior of model polyurethanes. Macromolecules 20(9):2089–2095
Chattopadhyay DK, Webster DC (2009) Thermal stability and flame retardancy of polyurethanes. Progress in Polymer Sci 34:1068–1133
Acknowledgments
The work has been financed by the European Regional Development Fund Contract No 2010/0214/2DP/2.1.1.1.0/10/APIA/VIAA/054.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stirna, U., Fridrihsone, A., Lazdiņa, B. et al. Biobased Polyurethanes from Rapeseed Oil Polyols: Structure, Mechanical and Thermal Properties. J Polym Environ 21, 952–962 (2013). https://doi.org/10.1007/s10924-012-0560-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-012-0560-0