Abstract
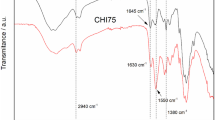
The synthesis and characterization of poly (acrylic acid) grafted pectin hydrogel followed by biosorption and desorption characteristics of cadmium, as a model heavy metal, have been studied. The grafted eco-friendly pectin based interpenetrating hydrogel was prepared in the presence of gluteraldehyde crosslinker under N2 atmosphere and characterized using 1H-NMR, FTIR, TGA and SEM techniques. Gluteraldehyde was found to form one-arm and two-arm crosslinks in the copolymer. Upon grafting, two-dimensional sheet structures bounded to tubular and vascular cylindrical rods were observed. The biosorption and desorption data, determined experimentally, were fitted to pseudo-second order reaction kinetics. At higher ionic strength values, the maximum metal uptake value (q max) was lowered and pseudo-second order rate constant (k 2) was increased. Whereas, at higher pH values the maximum metal uptake value (q max) was increased and Pseudo-second order rate constant (k 2) was decreased. 0.1 M HCl solution was a suitable eluent to regenerate the hydrogel surface and recover the adsorbed cadmium metal ions. Pectin based copolymer could be used as an efficient candidature biosorbent for the recovery of cadmium metal ions from aqueous solutions.








Similar content being viewed by others
References
Davidson MH, Dugan LL, Stocki J, Dicklin MR, Maki KC, Coletta F, Cotter R, McLeod M, Hoersten K (1998) J Nutr 128:1927
Chourasia MK, Jain SK (2004) Drug Delivery 11:129
Liu L, Fishman ML, Kost J, Hicks KB (2003) Biomaterials 24:3333
Sriamornsak P, Nunthanid J (1999) J Macroencapsul 16:303
Fares MM, Assaf SM, Abul-Haija YM (2010) J Appl Polym Sci 117(4):1945
Kartel MT, Kupchik LA, Veisov BK (1999) Chemosphere 38:2591
Khvan AM, Abduazimov KA (2001) Chem Nat Comp 37:388
Kamnev AA, Ptichkina NM, Perfiliev YD, Shkodina OG, Ignatov VV (1995) J Inorg Biochem 59:340
Harel P, Mignot L, Sauvage JP, Junter GA (1998) Ind Crop Prod 7:239
Dronnet VM, Renard CM, Axelos MA, Thibault JF (1999) Carbohydr Polym 30:253
Namasivayam C, Ranganathan K (1995) Environ Technol 16:851
Bailey SE, Olin TJ, Bricka RM, Adrian DD (1999) Water Res 33:2469
Hegazy EA, Abd SE, Taleb MF, Dessouki AM (2004) J Appl Polym Sci 92:2642
Inam R, Caykara T, Kantoglu O (2003) Nuc Inst Meth Phys Res B 208:400
Inam R, Gumu Y, Caykara T (2004) J Appl Polym Sci 94:2401
Ulusoy U, Symsek S, Ceyhan O (2003) Adsorption 9:165
Rivas BL, Hernan AP, Maturana A, Villegas S (2001) Macromol Chem Phys 202:443
Saraydin D, Karadag E, Guven O (2001) J Appl Polym Sci 79:1809
Fares MM, Othman AA (2008) J Appl Polym Sci 110(5):2815
Fares MM, Othman AA (2010) J Macromol Sci Part A: Pure Appl Chem 47(1):61
Merlin DL, Sivasankar B (2009) Euro Polym J 45:165
Culin J, Smit I, Andreis M, Veksli Z, Anzlovar A, Zigon M (2005) Polymer 46:89
Zhang XZ, Wu DQ, Chu CC (2004) Biomaterials 25:3793
Langer R (2003) N A Peppas AIChE J 49:2990
Jeong B, Gutowska A (2002) Trends Biotechnol 20:305
Renard CM, Crepeau MJ, Thibault JF (1995) Carbohydrate Res 275:155
Volesky B (2003) Sorption and biosorption. BV-Sorbex,Inc., St. Lambert, Quebec
Mehta SK, Gaur JP (2005) Crit Rev Biotechnol 25:113
Fares MM, El-faqeeh AS, Osman ME (2003) J Polym Res 10(2):119
Swinkles JJM (1985) Starch conversion technology. Marcel Dekker, New York, p 30
Acknowledgments
Jordan University of Science and Technology is acknowledged for this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fares, M.M., Tahboub, Y.R., Khatatbeh, S.T. et al. Eco-Friendly, Vascular Shape and Interpenetrating Poly (Acrylic Acid) Grafted Pectin Hydrogels; Biosorption and Desorption Investigations. J Polym Environ 19, 431–439 (2011). https://doi.org/10.1007/s10924-011-0296-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-011-0296-2