Abstract
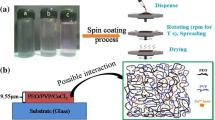
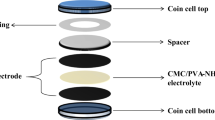
This article reports the ease and novel method to synthesise CeO2 nanoparticle in PVA polymer matrix. The optical, structural, thermal, mechanical, morphological, dielectric and electrochemical properties of the prepared films were understood by various charecterisation techniques such as UV–Visible, XRD, DSC, AFM, Impedance analyser, cyclic voltameter. The direct band gap for pure PVA is 5.426 eV and decreased to 4.278 eV. The indirect band gap values decreased from 4.86 to 2.04 eV. The degree of crystallinity for pure PVA is about 54% and increases to 79% for 15 wt% ACS doped PVA composite. Increase in Tg is observed with increase in doping concentration and is fitted to WLF–VTF model to evaluate dynamic fragility and apparent activation energy. The Non-Debye type of variation in dielectric constant with respect to frequency is observed. From the variation observed for M′ values is the indication of the presence of electrode polarization effect. The s value varied from 0.7 to 1.6, Therefore, the conductivity phenomenon explained on the NCL/SLPL type ion hopping mechanism. The prepared polymer electrolyte shows proton conductivity of the order of 10−3 Scm−1. The cyclic voltammograms shows an ideal capacitive behavior with good electrochemical stability. This article discuss about fundamentals of conducting phenomenon and its applicability in portable and flexible energy storage devices.













Similar content being viewed by others
References
H. Gao, K. Lian, RSC Adv. 4, 33091 (2014)
V. Neburchilov, J. Martin, H. Wang, J. Zhang, J. Power Sources 169, 221–238 (2007)
M. Hema, S. Selvasekerapandian, A. Sakunthala, D. Arunkuma, H. Nithya, Physica B 403, 2740–2747 (2008)
M. Rikukawa, K. Sanui, Prog. Polym. Sci. 25, 1463–1502 (2000)
M.J. Wei, J.Q. Fu, Y.D. Wang, J.Y. Gu, B.L. Liu, H.Y. Zang, E.L. Zhou, K.Z. Shao, Z.M. Su, J. Mater. Chem. A 5, 1085–1093 (2017)
G. Inzelt, M. Pineri, J.W. Schultze, M.A. Vorotyntsev, Electrochim. Acta 45, 2403–2421 (2000)
G.A. Voth, Acc. Chem. Res. 39, 143–145 (2006)
M. Sadakiyo, T. Yamada. H. Kitagawa, J. Am. Chem. Soc. 136, 13166–13169 (2014)
J. Naik, R.F. Bhajantri, T. Sheela, S.G. Rathod, Polym. Compos. (2016). https://doi.org/10.1002/pc.24063
N. Kulshrestha, B. Chatterjee, P.N. Gupta, Mater. Sci. Eng. B 184, 49–57 (2014)
P. Donoso, W. Gorecki, C. Berthier, F. Defendini, C. Poinsignon, M.B. Armand, Solid State Ion. 28, 969 (1988)
S. Sikkanthar, S. Karthikeyan, S. Selvasekarapandian, D.V. Pandi, S. Nithya, C. Sanjeeviraja, J Solid State Electrochem. 19, 987–999 (2015)
I. Albinsson, B.E. Mellander, J.R. Stevens, Solid State Ion. 72, 177–182 (1994)
K.K. Maurya, B. Bhattacharya, S. Chandr, Phys. State Solid 147, 347 (1995)
B. Haldar, R.M. Singru, K.K. Maurya, S. Chandra, Phys. Rev. B 54(10), 7143–7150 (1996)
M.E. Gouda, S.K. Badr, M.A. Hassan, E. Sheha, Ionics 17, 255–261 (2011)
M.H. Buraidah, L.P. Teo, S.R. Majid, A.K. Arof, Physica B 404, 1373–1379 (2009)
G. Hirankumar, S. Selvasekarapandian, N. Kuwata, J. Kawamura, T. Hattori, J. Power Sources 144, 262–267 (2005)
R. Bouchet, E. Siebert, Solid State Ion. 118, 287–299 (1999)
C. Ambika, G. Hirankumar, Appl. Phys. A 122, 113 (2016)
X. Meng, H.N. Wang, S.Y. Song, H.J. Zhang, Chem. Soc. Rev. (2017). https://doi.org/10.1039/C6CS00528D
F. Vaja, O. Oprea, D. Ficai, A. Ficai, C. Guran, Digest. J. Nanomater. Biostruct. 9(1), 187–195 (2014)
[V.D. Araujo, W. Avansi, H.B. de Carvalho, M.L. Moreira, E. Longo, C. Ribeiro, M.I.B. Bernardi, CrystEngComm 14, 1150 (2012)
S. Phoka, P. Laokul, E. Swatsitang, V. Promarak, S. Seraphin, S. Maensiri, Mater. Chem. Phys. 115, 423–428 (2009)
S. Gnanam, V. Rajendran, J. Sol-Gel Sci. Technol. 58, 62–69 (2011)
V. Hebbar, R.F. Bhajantri, J. Naik, S.G. Rathod, Mater. Res. Exp. 3, 075301 (2016)
H.S. Ragab, M.F.H. Abd El-Kader, Phys. Screen. 87, 025602 (2013)
C. Ho, J.C. Yu, T. Kwong, A.C. Mak, S. Lai, Chem. Mater. 17, 4514–4522 (2005)
M. Hirano, Y. Fukuda, H. Iwata, Y. Hotta, M. Inagaki, J. Am. Ceram. Soc. 83(5), 1287–1289 (2000)
M. Tanaka, Y. Takeda, T. Wakiya, Y. Wakamoto, K. Harigaya, T. Ito, T. Tarao, H. Kawakami, J. Power Sources 342, 125–134 (2017)
H. Imagawa, A. Suda, K. Yamamura, S. Sun, J. Phys. Chem. C 115, 1740–1745 (2011)
M.K. Chinnu, K.V. Anand, R.M. Kumar, T. Alagesan, R. Jayavel, Mater. Lett. 113, 170–173 (2013)
K. Selvakumar, J. Kalaiselvimary, S. Rajendran, M.R. Prabhu, Polym. Plast. Technol. Eng. 55(18), 1940–1948 (2016)
B. Ramaraj, J. Appl. Polym. Sci. 103, 1127–1132 (2007)
A.L. Agapov, A.P. Sokolov, Macromolecules 44, 4410–4414 (2011)
I. Dranca, Chem. J. Mold. 3(1)), 31–43 (2008)
B.A.P. Betancourt, J.F. Douglas, F.W. Starr, Soft Matter. 9, 241 (2013)
J.S. Karthika, B. Vishalakshi, J. Naik, Int. J. Biol. Macromol. 82, 61–67 (2016)
V. Hebbar, R.F. Bhajantri, J. Naik, J. Mater. Sci. 28, 5827–5839 (2017)
S.K. Gedam, A.P. Khandale, S.S. Bhoga, Ind. J. Pure Appl. Phys. 51, 367–371 (2013)
J. Tahalyani, K.K. Rahangdale, R. Aepuru, B. Kandasubramanian, S. Datar, RSC Adv. 6, 36588–36598 (2016)
R. Golshaei, Z. Guler, C. unsal, A.S. Sarac, Eur. Polym. J. 66, 502–512 (2015)
S. Ke, H. Huang, S. Yu, L. Zhou, J. Appl. Phys. 107, 084112 (2010)
T. Tiwari, N. Tarannum, M. Kumar, N. Srivastava, Ionics 20, 1435–1443 (2014)
H.M.E. Mallah, Acta Phys. Polym. A 1, 122 (2012)
J.J. Habasaki, K.L. Ngai, Y. Hiwatari, Chem. Phys. 120, 17 (2004)
S. Rajendran, T. Uma, Ionics 6, 288–293 (2000)
C.W. Liew, K.H. Arifin, J. Kawamura, Y. Iwai, S. Ramesh, A.K. Arof, J. Non-Cryst. Solids 458, 97–106 (2017)
S. Yang, L. Gao, J. Am. Chem. Soc. 128, 9330–9331 (2006)
T.N. Ravishankar, T. Ramakrishnappa, G. Nagaraju, H. Rajanaika, Chem. Open 4, 146–154 (2015)
Acknowledgements
The authors are thankful to Science and Engineering Research Board (SERB), Department of Science and Technology (DST), Government of India for the research projects (SR/FTP/PS-011/2010) and (SB/EMEQ-089/2013) and to UGC, New Delhi for SAP-CAS Phase-II (F.530/9/CAS-II/2015(SAP-I) research grant. The authors are also thankful to DST-PURSE Laboratory, Mangalore University for providing UV–Visible, TGA measurements. The authors are also thankful to USIC, Karnatak University, Dharwad for providing DSC, AFM, CV measurement facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Naik, J., Bhajantri, R.F. Physical and Electrochemical Studies on Ceria Filled PVA Proton Conducting Polymer Electrolyte for Energy Storage Applications. J Inorg Organomet Polym 28, 906–919 (2018). https://doi.org/10.1007/s10904-018-0801-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-018-0801-3