Abstract
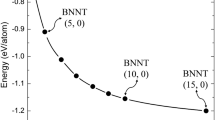
The structural stability of borophene nanosheet and nanotubes, including electronic properties was explored using the density-functional-theory technique. Moreover, the stability of isolated pristine borophene nanosheet and nanotube is substantiated with the help of formation energy. The energy band gap of pristine borophene material is found to be semi-metallic. The interaction of NH3 molecules on borophene material is analyzed using Bader charge transfer, adsorption energy, density-of-states spectrum, energy band gap and percentage of average-energy-gap variation. The interaction of hazardous NH3 gas molecules on borophene nanosheet and nanotubes is studied at an atomistic level. The interaction of nitrogen atom in ammonia molecules on borophene material is observed to be the prominent adsorption site. The findings suggest that borophene nanosheets as well as nanotubes can be used as a chemi-resistor for the detection of ammonia molecules present in the atmosphere.
Graphical Abstract








Similar content being viewed by others
References
A. Geim, Graphene: status and prospects. Science 324, 1530–1534 (2009)
A.R. Oganov, J. Chen, C. Gatti, Y.-Z. Ma, Y.-M. Ma, C.W. Glass, Z. Liu, T. Yu, O.O. Kurakevych, V.L. Solozhenko, Ionic high-pressure form of elemental boron. Nature 457 863–867 (2009)
N. Vast, S. Baroni, G. Zerah, J.M. Besson, A. Polian, M. Grimsditch, J.C. Chervin, Lattice dynamics of icosahedral α-boron under pressure. Phys. Rev. Lett. 78, 693 (1997)
H. Tang, I.B. Sohrab, First-principles study of boron sheets and nanotubes. Phys. Rev. B 82, 115412 (2010)
A.J. Mannix, X.F. Zhou, B. Kiraly, Synthesis of borophenes: anisotropic, two-dimensional boron polymorphs. Science 350, 1513–1516 (2015)
X. Yang, Y. Ding, J. Ni, Ab initio prediction of stable boron sheets and boron anotubes: structure, stability, and electronic properties. Phys. Rev. B 77, 041402 (2008)
B.J. Feng, J. Zhang, Q. Zhong, W.B. Li, S. Li, H. Li, P. Cheng, S. Meng, L. Chen, K.H. Wu, Experimental realization of two-dimensional boron sheets. Nat. Chem. 8, 563–568 (2016)
I. Boustani, A. Quandt, Nanotubes of bare boron clusters: ab initio and density functional study. Europhys. Lett. 39, 527–532 (1997)
I. Boustani, New quasi-planar surfaces of bare boron. Surf. Sci. 370, 355–363 (1997)
B. Kiran, S. Bulusu, H.-J. Zhai, S. Yoo, X.C. Zeng, L.-S. Wang, Planar-to-tubular structural transition in boron clusters: B20 as the embryo of single-walled boron nanotubes. Proc. Natl. Acad. Sci. USA 102, 961–964 (2005)
E. Oger, N.R.M. Crawford, R. Kelting, P. Weis, M.M. Kappes, R. Ahlrichs, Boron cluster cations: transition from planar to cylindrical structures. Angew. Chem. Int. Ed. 46, 8503–8506 (2007)
A.K. Singh, A. Sadrzadeh, B.I. Yakobson, Probing properties of boron R-tubes by ab initio calculations. Nano Lett. 8, 1314–1317 (2008)
H. Sun, Q. Li, X.G. Wana, First-principles study of thermal properties of borophene. Phys. Chem. Chem. Phys. 18, 14927 – 14932 (2016)
Y. Zhao, S. Zeng, N. Jun, Superconductivity in two-dimensional boron allotropes. Phys. Rev. B 93, 014502 (2016)
X. Zhang, J. Hu, Y. Cheng, H.Y. Yang, Y. Yao, A.Y. Shengyuan, Borophene as an extremely high capacity electrode material for Li-ion and Na-ion batteries. Nanoscale 8, 15340–15347 (2016)
F. Liu, C. Shen, Z. Su, X. Ding, S. Deng, J. Chen, N. Xu, H. Gao, Metal-like single crystalline boron nanotubes: synthesis and in situ study on electric transport and field emission properties. J. Mater. Chem. 20, 2197–2205 (2010)
D. Ciuparu, R.F. Klie, Y. Zhu, L. Pfefferle, Synthesis of pure boron single-wall nanotubes. J. Phys. Chem. B 108, 3967–3969 (2004)
S. Iijima, Helical microtubules of graphitic carbon. Nature 354, 56–58 (1991)
R. Chandiramouli, First-principles insights on adsorption properties of NH3 on silicane nanoribbon and nanoring. Appl. Surf. Sci. 426, 1221–1231 (2017)
V. Nagarajan, R. Chandiramouli, Investigation on electronic properties of functionalized arsenene nanoribbon and nanotubes: a first-principles study. Chem. Phys. 495, 35–41 (2017)
A. Rubio, J. Corkill, M.L. Cohen, Theory of graphitic boron nitride nanotubes. Phys. Rev. B 49, 5081–5084 (1994)
R.T. Senger, S. Dag, S. Ciraci, Chiral single-wall gold nanotubes. Phys. Rev. Lett. 93, 196807 (2004)
V. Bezugly, J. Kunstmann, B. Grundkotter-Stock, T. Frauenheim, T. Niehaus, G. Cuniberti, Highly conductive boron nanotubes: transport structural stabilities. ACS Nano 5, 4997–5005 (2011)
V. Nagarajan, R. Chandiramouli, Borophene nanosheet molecular device for detection of ethanol: a first-principles study. Comput. Theor. Chem. 1105, 52–60 (2017)
V. Nagarajan, R. Chandiramouli, Sensing properties of monolayer borophane nanosheet towards alcohol vapors: a first-principles study. J. Mol. Graph. Modell. 73, 208–216 (2017)
S. Bibi, H. Ullah, S.M. Ahmad, H.A. Shah, S. Bilal, A.A. Tahir et al., Molecular and electronic structure elucidation of polypyrrole gas sensors. J. Phys. Chem. C 119(28), 15994–16003 (2015)
H. Ullah, A.A. Shah, S. Bilal, K. Ayub, DFT study of polyaniline NH3, CO2, and CO gas sensors: comparison with recent experimental data. J. Phys. Chem. C 117, 23701–23711 (2013)
H. Ullah, K. Ayub, Z. Ullah, M. Hanif, R. Nawaz, A.A. Shah et al., Theoretical insight of polypyrrole ammonia gas sensor. Synth. Methods 172, 14–20 (2013)
H. Ullah, A.A. Shah, S. Bilal, K. Ayub, Doping and dedoping processes of polypyrrole: DFT study with hybrid functionals. J. Phys. Chem. C 118, 17819–17830 (2014)
H. Ullah, A.A. Shah, K. Ayub, S. Bilal, Density functional theory study of poly(o-phenylenediamine) oligomers. J. Phys. Chem. C 117, 4069–4078 (2013)
M. Kamran, H. Ullah, A. Anwar-ul-Haq, S. Shah, A.A. Bilal, K. Tahir, Ayub, Combined experimental and theoretical study of poly(aniline-co-pyrrole) oligomer. Polymer 72, 30–39 (2015)
Z. Fazl-i-Sattar, A. Ullah Ata-ur-Rahman, M. Rauf, A.A. Tariq, K. Tahir, H. Ayub, Ullah, Phytochemical, spectroscopic and density functional theory study of Diospyrin, and non-bonding interactions of Diospyrin with atmospheric gases. Spectrochim. Acta A 141, 71–79 (2015)
H. Ullah, A.A. Tahir, T.K. Mallick, Polypyrrole/TiO2 composites for the application of photocatalysis. Sens. Actuators B 241, 1161–1169 (2017)
H. Ullah, Inter-molecular interaction in polypyrrole/TiO2: a DFT study. J.Alloys Compd. 692, 140–148 (2017)
A.A. Peyghan, H. Soleymanabadi, Z. Bagheri, Hydrogen release from NH3 in the presence of BN graphene: DFT studies. J. Mex. Chem. Soc. 59(1), 67–73 (2015)
A.A. Peyghan, S.F. Rastegar, N.L. Hadipour, DFT study of NH3 adsorption on pristine, Ni- and Si-doped graphynes. Phys. Lett. A 378, 2184–2190 (2014)
A.A. Peyghan, M.B. Tabar, J. Kakemam, NH3 on a BC3 nanotube: effect of doping and decoration of aluminum. J. Mol. Model. 19, 3793–3798 (2013)
J. Beheshtian, A.A. Peyghan, Z. Bagheri, Ab initio study of NH3 and H2O adsorption on pristine and Na-doped MgO nanotubes. Struct. Chem. 24, 165–170 (2013)
J.M. Soler, E. Artacho, J.D. Gale, A. Garcia, J. Junquera, P. Ordejon, D. Sanchez-Portal, The SIESTA method for ab initio order-N materials simulation. J. Phys. Condens. Matter. 14, 2745 (2002)
G. Roman-Perez, J.M. Soler, Efficient implementation of a van der waals density functional: application to double-wall carbon nanotubes. Phys. Rev. Lett. 103, 096102 (2009)
J.P. Perdew, K. Burke, Y. Wang, Generalized gradient approximation for the exchange-correlation hole of a many-electron system. Phys. Rev. B 54, 16533 (1996)
J.P. Perdew, J.A. Chevary, S.H. Vosko, K.A. Jackson, M.R. Pederson, D.J. Singh, C. Fiolhais, Atoms,molecules, solids, and surfaces: applications of the generalized gradient approximation for exchange and correlation. Phys. Rev. B 46, 6671 (1992)
L. Li, P. Jin, D.-E. Jiang, L. Wang, S.B. Zhang, J. Zhao, Z.J. Chen, B80 and B101-103 clusters: remarkable stability of the core-shell structures established by validated density functionals. Chem. Phys. 136(7), 074302 (2012)
R. Bader, Atoms in Molecules: A Quantum Theory. (Oxford University Press, New York, 1990)
V. Nagarajan, R. Chandiramouli, Interaction of alcohols on monolayer stanane nanosheet: a first-principles investigation. Appl. Surf. Sci. 419, 9–15 (2017)
J. Yuan, L.W. Zhang, K.M. Liew, Effect of grafted amine groups on in-plane tensile properties and high temperature structural stability of borophene nanoribbons. RSC Adv. 5, 74399–74407 (2015)
F. Ma, Y. Jiao, G. Gao, Y. Gu, A. Bilic, Z. Chen, A. Du, Graphene-like two dimensional ionic boron with double Dirac cones at ambient condition. Nano Lett. 16, 3022–3028 (2016)
J. Beheshtian, M. Noei, H. Soleymanabadi, A.A. Peyghan, Ammonia monitoring by carbon nitride nanotubes: a density functional study. Thin Solid Films 534, 650–654 (2013)
A. Ullah Ata-ur-Rahman, Z. Fazl-i-Sattar, A. Rauf, M. Yaseen, W. Hassan, M. Tariq, K. Ayub, A.A. Tahir, H. Ullah, Density functional theory and phytochemical study of 8-hydroxyisodiospyrin. J. Mol. Struct. 1095, 69–78 (2015)
A.A. Javad Beheshtian, N.L. Hadipour, Interaction of NH3 with aluminum nitride nanotube: electrostatic vs. covalent. Physica E 43, 1717–1719 (2011)
J. Beheshtian, I. Ravaei, Toxic CO detection by Li-encapsulated fullerene-like BeO. Struct. Chem. (2017). 10.1007/s11224-017-1022-z
M. Eslami, V. Vahabi, A.A. Peyghan, Sensing properties of BN nanotube toward carcinogenic 4-chloroaniline: a computational study. Physica E 76, 6–11 (2016)
V. Nagarajan, R. Chandiramouli, Interaction of NH3 gas on α-MoO3 nanostructures: a DFT investigation. Condens. Matter Phys. 20, 23705 (2017)
M. Noei, A.A. Peyghan, A DFT study on the sensing behavior of a BC2N nanotube toward formaldehyde. J. Mol. Model. 19, 3843–3850 (2013)
H. Ullah, A.-H.A. Shah, S. Bilal, K. Ayub, DFT study of polyaniline NH3, CO2, and CO gas sensors: comparison with recent experimental data. J. Phys. Chem. C 117, 23701–23711 (2013)
R. Chandiramouli, B.G. Jeyaprakash, Operating temperature dependent ethanol and formaldehyde detection of spray deposited mixed CdO and MnO2 thin films. RSC Adv. 5, 43930–43940 (2015)
A.S. Kootenaei, G. Ansari, B36 borophene as an electronic sensor for formaldehyde: quantum chemical analysis. Phys. Lett. A 380, 2664–2668 (2016)
B. Mortazavi, A. Dianat, O. Rahaman, G. Cuniberti, T. Rabczuk, Borophene as an anode material for Ca, Mg, Na or Li ion storage: a first-principle study. J. Power Sources 329, 456–461 (2016)
V. Nagarajan, R. Chandiramouli, Adsorption of NO2 molecules on armchair phosphorene nanosheet for nano sensor applications: a first-principles study. J. Mol. Graph. Modell. 75, 365–374 (2017)
J. Beheshtian, A.A. Peyghan, Z. Bagheri, Arsenic interactions with a fullerene-like BN cage in the vacuum and aqueous phase. J. Mol. Model. 19, 833–837 (2013)
R. Chandiramouli, V. Nagarajan, Borospherene nanostructure as CO and NO sensor: a first-principles study. Vacuum 142, 13–20 (2017)
S. Bibi, H. Ullah, S.M. Ahmad, A.-H.A. Shah, S. Bilal, A.A. Tahir, K. Ayub, Molecular and electronic structure elucidation of polypyrrole gas sensors. J. Phys. Chem. C 119, 15994–16003 (2015)
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nagarajan, V., Chandiramouli, R. Interaction Studies of Ammonia Gas Molecules on Borophene Nanosheet and Nanotubes: A Density Functional Study. J Inorg Organomet Polym 28, 920–931 (2018). https://doi.org/10.1007/s10904-017-0761-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-017-0761-z