Abstract
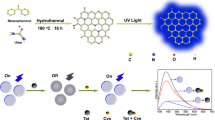
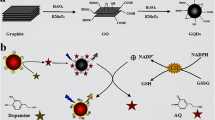
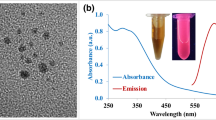
In this study, we report on the fabrication of simple and rapid graphene quantum dots (GQDs)-based fluorescence “turn-ON” nanoprobes for sensitive and selective detection of ascorbic acid (AA). Pristine GQDs and S and N co-doped-GQDs (SN-GQDs) were functionalized with 4-amino-2,2,6,6-tetramethylpiperidine-N-oxide (4-amino-TEMPO, a nitroxide free radical). The nitroxide free radicals efficiently quenched the fluorescence of the GQDs and upon interaction of the nanoconjugates with ascorbic acid, the quenched fluorescence was restored. The linear ranges recorded were 0.5–5.7 μM and 0.1–5.5 μM for GQDs-4-amino-TEMPO and SN-GQDs-4amino-TEMPO nanoprobes, respectively. Limits of detection were found to be 60 nM and 84 nM for SN-GQDS-4-amino-TEMPO and GQDs-4-amino-TEMPO for AA detection, respectively. This novel fluorescence “turn-ON” technique showed to be highly rapid and selective towards AA detection.













Similar content being viewed by others
References
Lin L, Rong M, Luo F, Chen D, Wang Y, Chen X (2014) Luminescent graphene quantum dots as new fluorescent materials for environmental and biological applications. Trends Anal Chem 54:83–102
Ge J, Minhuan L, Zhou B, Liu W, Guo L, Wang H, Jia Q, Niu G, Huang X, Zhou H, Xiangmin M, Pengfei W, Chun-Sing L, Zhang W, Han X (2014) A graphene quantum dot photodynamic therapy agent with high singlet oxygen generation. Nat Commun 5:4596
Zhu S, Zhang J, Liu X, Li B, Wang X, Tang S, Meng Q, Li Y, Shi C, Hu R, Yang B (2012) Graphene quantum dots with controllable surface oxidation, tunable fluorescence and up-conversion emission. RSC Adv 2:2717–2720
Lin F, Pei D, He W, Huang Z, Huang Y, Guo X (2012) Electron transfer quenching by nitroxide radicals of the fluorescence of carbon dots. J Mater Chem 22:11801–11807
Liu CP, TH W, Liu CY, Cheng HJ, Lin SY (2015) Interactions of nitroxide radicals with dendrimer-entrapped Au8-cluster: a fluorescent nanosensor for intracellular imaging of ascorbic acid. J Mater Chem B 3:191–197
Maurel V, Laferrière M, Billone P, Godin R, Scaiano JC (2006) Free radical sensor based on CdSe quantum dots with added 4-amino-2,2,6,6-Tetramethylpiperidine oxide functionality. J Phys Chem B 110:16353–16358
Ishii K, Kubo K, Sakurada T, Komori K, Sakai Y (2011) Phthalocyanine-based fluorescence probes for detecting ascorbic acid: phthalocyaninatosilicon covalently linked to TEMPO radicals. Chem Commun 47:4932–4934
Adegoke O, Hosten E, McCleland C, Nyokong T (2012) CdTe quantum dots functionalized with 4-amino-2,2,6,6-tetramethylpiperidine-N-oxide as luminescent nanoprobe for the sensitive recognition of bromide ion. Anal Chim Acta 721:154–161
Qu D, Zheng M, Du P, Zhou Y, Zhang L, Li D, Tan H, Zhao Z, Xied Z, Sun Z Highly luminescent S, N co-doped graphene quantum dots with broad visible absorption bands for visible light photocatalysts. Nanoscale 5:12272–12277
Connell PJ, Gormally C, Pravda M, Guilbault GG (2001) Development of an amperometric L-ascorbic acid (vitamin C) sensor based on electropolymerised aniline for pharmaceutical and food analysis. Anal Chim Acta 431:239–247
Agater IB, Jewsbury RA (1995) Determination of plasma ascorbic acid by HPLC: method and stability studies. Eur J Pharm Sci 3:231–239
Wu T, Guan Y, Ye J (2007) Determination of flavonoids and ascorbic acid in grapefruit peel and juice by capillary electrophoresis with electrochemical detection. Food Chem 100:1573–1579
Zhang YF, Li BX, Xu CL (2010) Visual detection of ascorbic acid via alkyne-azide click reaction using gold nanoparticles as a colorimetric probe. Analyst 135:1579–1584
GZ H, Guo Y, Xue QM, Shao SJ (2010) A highly selective amperometric sensor for ascorbic acid based on mesopore-rich active carbon-modified pyrolytic graphite electrode. Electrochim Acta 55:2799–2804
Park HW, Alam SM, Lee SH, Karim MM, Wabaidur SM, Kang M, Choi JH (2009) Optical ascorbic acid sensor based on the fluorescence quenching of silver nanoparticles. Luminescence 24:367–371
Liu JJ, Chen ZT, Tang DS, Wang YB, Kang LT, Yao JN (2015) Graphene quantum dots-based fluorescent probe for turn-on sensing of ascorbic acid. Sensors Actuators B Chem 212:214–219
Tshangana C, Nyokong T (2015) The photophysical properties of multi-functional quantum dots-magnetic nanoparticles – indium octacarboxy phthalocyanine nanocomposites. J Fluoresc 25:199–210
Fashina A, Antunes E, Nyokong T (2013) Characterization and photophysical behaviour of phthalocyanines when grafted onto silica nanoparticles. Polyhedron 53:278–285
Qu D, Zheng M, Zhang L, Zhao H, Xie Z, Jing X, Raid EH, Fan H, Sun Z (2014) Formation mechanism and optimization of highly luminescent N-doped graphene quantum dots. Sci Report 4:1–9
Fery-Forgues S, Lavabre D (1999) Are fluorescence quantum yields so tricky to measure? A demonstration using familiar stationery products. J Chem Ed 76:12660–11264
Fischer S, Georges J (1996) Fluorescence quantum yields of Rhodamine 6G in ethanol as a function of concentration using lens spectrometry. Chem Phys Lett 260:115–118
Idowu M, Nyokong T (2009) Interaction of water-soluble CdTe quantum dots with Octacarboxy metallophthalocyanines: a photophysical and photochemical study. J Lumin 129:356–362
Yuan F, Ding L, Li Y, Li X, Fan L, Zhou S, Fang D, Yang S (2015) Multicolor fluorescent graphene quantum dots colorimetrically responsive to all-pH and a wide temperature range. Nanoscale 7:11727–11733
Achadu OJ, Nyokong T (2016) Interaction of graphene quantum dots with 4-acetamido-2,2,6,6-tetramethylpiperidine-oxyl free radical: A spectroscopic and fluorimetric study. J Fluoresc 26:283–295
Chua CK, Sofer Z, Šimek P, Jankovský O, KlÍmová K, Bakardjieva S, Kučková SH, Pumera M (2015) Synthesis of strongly fluorescent graphene quantum dots by cage-opening buckminsterfullerene. ACS Nano 9:2548–2555
Nurunnabi M, Zehedina K, Nafiujjaman M, Dong L, Lee Y-k (2013) Surface Coating of Graphene Quantum Dots Using Mussel-Inspired Polydopamine for Biomedical Optical Imaging. ACS Appl Mater Interfaces 5:8246–8253
Zheng XT, Ananthanarayanan KQ, Luo P, Chen P (2015) Glowing graphene quantum dots and carbon dots: properties, syntheses, and biological applications. Small 11:1620–1636
Hu Y, Zhao G, Lu N, Chen Z, Zhang H, Li H, Shao L, Qu L (2013) Graphene quantum dots-carbon nanotubes hybrid arrays for supercapacitors. Nanotechnology 24:195401
Chien C, Li S, Lai W, Yeh Y, Chen H, Chen I, Chen L, Nemoto IS (2012) Tunable photoluminescence from graphene oxide. Angew Chem Int Ed 51:6662–6666
Dong Y, Lin J, Chen Y, Fu F, Chi Y, Chen G (2014) Graphene quantum dots and graphite nanocrystals in coal. Nanoscale 6:7410–7415
Hoque MN, Basu A, Das G (2014) Fluorescence turn on sensor for sulfate ion in aqueous medium using Tripodal-Cu2+ ensemble. J Fluoresc 24:411–416
Dɑnet AF, Badea M, Aboul-Enein HY (2000) Flow injection system with chemiluminometric detection for enzymatic determination of ascorbic acid. Luminescence 15:305–309
WN H, Sun DM, Ma W (2010) Silver doped poly (L-valine) modified glassy carbon electrode for the simultaneous determination of uric acid, ascorbic acid and dopamine. Electroanalysis 22:584–589
Chen YJ, Yan XP (2009) Chemical redox modulation of the surface chemistry of CdTe quantum dots for probing ascorbic acid in biological fluids. Small 5:2012–2018
Wang XX, Wu P, Hou XD, Lv Y (2013) An ascorbic acid sensor based on protein modified Au nanoclusters. Analyst 138:229–233
Sillen A, Engelborghs Y (1998) The correct use of “average” fluorescence parameters. Photochem Photobiol 67(5):475–486
Acknowledgments
This work was supported by the Department of Science and Technology (DST) and National Research Foundation (NRF), South Africa through DST/NRF South African Research Chairs Initiative for Professor of Medicinal Chemistry and Nanotechnology (UID 62620) as well as Rhodes University/DST Centre for Nanotechnology Innovation, Rhodes University, South Africa.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Achadu, O.J., Britton, J. & Nyokong, T. Graphene Quantum Dots Functionalized with 4-Amino-2, 2, 6, 6-Tetramethylpiperidine-N-Oxide as Fluorescence “Turn-ON” Nanosensors. J Fluoresc 26, 2199–2212 (2016). https://doi.org/10.1007/s10895-016-1916-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-016-1916-y