Abstract
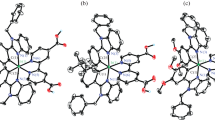
A series of new cyclometalated btp-based iridium(III) complexes with three different ancillary ligands, Ir(btp)2(bozp) (3a), Ir(btp)2(btzp) (3b) and Ir(btp)2(izp) (3c) (btp = 2-(benzo[b]thiophen-2-yl)pyridine, bozp =2-(benzo[d]oxazol-2-yl)phenol, btzp =2-(benzo[d]thiazol-2-yl)phenol, izp = 2-(2 H-indazol-2-yl)phenol), have been synthesized and fully characterized. The crystal structure of 3b has been determined by single crystal X-ray diffraction analysis. A comparative study has been carried out for complexes 3a − 3c by UV-vis absorption spectroscopy, photoluminescence spectroscopy, cyclic voltammetry and DFT calculations. This observation illustrates that the substitution of N or S in ancillary ligand can lead to a marked bathochromic shift of absorption and emission wavelengths. The spectroscopic characterisation of these complexes has been complemented by DFT and TD-DFT calculations, supporting the assignment of 3MLCT/3LC to the lowest energy excited state.






Similar content being viewed by others
References
Baldo MA, Adachi C, Forrest SR (2000) Transient analysis of organic electrophosphorescence. II. Transientanalysis of triplet-triplet annihilation. Phys Rev B 62:10967–10977
Wu C, Chen HF, Wong KT, Thompson ME (2010) Study of ion-paired iridium complexes (soft salts) and their application in organic light emitting diodes. J Am Chem Soc 132:3133–3139
Lupton JM, Samuel IDW, Frampton MJ, Beavington R, Burn PL (2001) Control of electrophosphorescence in conjugated dendrimer light-emitting diodes. Adv Funct Mater 11:287–294
Niu ZG, Liu D, Zuo J, Zou Y, Yang JM, Su YH, Yang YD, Li GN (2014) Four new cyclometalated phenylisoquinoline-based Ir(III) complexes: syntheses, structures, properties and DFT calculations. Inorg Chem Commun 43:146–150
Lamansky S, Djurovich P, Murphy D, Abdel-Razzaq F, Lee HE, Adachi C, Burrows PE, Forrest SR, Thompson ME (2001) Highly phosphorescent bis-cyclometalated iridium complexes: synthesis, photophysical characterization, and use in organic light emitting diodes. J Am Chem Soc 123:4304–4312
Chen LQ, Yang CL, Qin JG, Gao J, Ma DG (2006) Tuning of emission: synthesis, structure and photophysical properties of imidazole, oxazole and thiazole-based iridium (III) complexes. Inorg Chim Acta 359:4207–4214
Zhao Q, Jiang CY, Shi M, Li FY, Yi T, Cao Y, Huang CH (2006) Synthesis and photophysical, electrochemical, and electrophosphorescent properties of a series of iridium(III) complexes based on quinoline derivatives and different β-diketonate ligands. Organometallics 25:3631–3638
Li YF, Liu Y, Zhou M (2012) Acid induced acetylacetonato replacement in biscyclometalated iridium(III) complexes. Dalton Trans 41:3807–3816
Hay PJ (2002) Theoretical studies of the ground and excited electronic states in cyclometalated phenylpyridine Ir(III) complexes using density functional theory. J Phys Chem A 106:1634–1641
Tamayo AB, Alleyne BD, Djurovich PI, Lamansky S, Tsyba I, Ho NN, Bau R, Thompson ME (2003) Synthesis and characterization of facial and meridional tris-cyclometalated iridium(III) complexes. J Am Chem Soc 125:7377–7387
Sharma S, Kim H, Lee YH, Kim T, Lee YS, Lee MH (2014) Heteroleptic cyclometalated iridium(III) complexes supported by triarylborylpicolinate ligand: ratiometric turn-on phosphorescence response upon fluoride binding. Inorg Chem 53:8672–8680
Li GN, Zou Y, Yang YD, Liang J, Cui F, Zheng T, Xie H, Niu ZG (2014) Deep-red phosphorescent iridium(III) complexes containing 1-(benzo[b] thiophen-2-yl) isoquinoline ligand: synthesis, photophysical and electrochemical properties and DFT calculations. J Fluoresc 24:1545–1552
Juris A, Balzani V, Barigelletti F, Campagna S, Belser P, Vonzelewsky A (1988) Ru(II) polypyridine complexes: photophysics, photochemistry, eletrochemistry, and chemiluminescence. Coord Chem Rev 84:85–277
Frank M, Nieger M, Vögtle F, Belser P, Vonzelewsky A, Cola LD, Balzani V, Barigelletti F, Flamigni L (1996) Dinuclear RuII and/or OsII complexes of bis-bipyridine bridging ligands containing adamantane spacers: synthesis, luminescence properties, intercomponent energy and electron transfer processes. Inorg Chim Acta 242:281–291
Oh H, Park KM, Hwang H, Oh S, Lee JH, Lu JS, Wang S, Kang Y (2013) Effective alkoxylation of phosphorescent heteroleptic iridium(III) compounds bearing fluorinated bipyridine ligands. Organometallics 32:6427–6436
CrysAlisPro Version 1.171.36.21. (2012) Agilent technologies Inc. Santa Clara, CA
Sheldrick GM (2008) A short history of SHELX. Acta Cryst Sect A 64:112–122
Dolomanov OV, Bourhis LJ, Gildea RJ, Howard JK, Puschmann H (2009) OLEX2: a complete structure solution, refinement and analysis program. J Appl Crystallogr 42:339–341
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2009) Gaussian 09, Revision A.01. Wallingford, Gaussian, Inc.
Lee C, Yang W, Parr RG (1988) Development of the colle-salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789
Miehlich B, Savin A, Stoll H, Preuss H (1989) Results obtained with the correlation energy density functionals of becke and Lee, Yang and Parr. Chem Phys Lett 157:200–206
Becke AD (1993) Density-functional thermochemistry.III. The role of exact exchange. J Chem Phys 98:5648–5652
Cossi M, Rega N, Scalmani G, Barone V (2003) Energies, structures, and electronic properties of molecules in solution with the C-PCM solvation model. J Comput Chem 24:669–681
Tomasi J, Mennucci B, Cammi R (2005) Quantum mechanical continuum solvation models. Chem Rev 105:2999–3094
Jiang N, Yang CL, Dong XW, Sun XL, Dan Z, Liu CL (2014) An ESIPT fluorescent probe sensitive to protein α-helix structures. Org Biomol Chem 12:5250–5259
Khan KM, Rahim F, Halim SA, Taha M, Khan M, Perveen S, Haq Z, Mesaik MA, Choudhary MI (2011) Synthesis of novel inhibitors of β-glucuronidase based on benzothiazole skeleton and study of their binding affinity by molecular docking. Bioorg Med Chem 19:4286–4294
Varughese DJ, Manhas MS, Bose AK (2006) Microwave enhanced greener synthesis of indazoles via nitrenes. Tetrahedron Lett 47:6795–6797
Neve F, La Deda M, Crispini A, Bellusci A, Puntoriero F, Campagna S (2004) Cationic cyclometalated iridium luminophores: photophysical, redox, and structural characterization. Organometallics 23:5856–5863
Wang RJ, Deng LJ, Zhang T, Li JY (2012) Substituent effect on the photophysical properties, electrochemical properties and electroluminescence performance of orange-emitting iridium complexes. Dalton Trans 41:6833–6841
Okada S, Okinaka K, Iwawaki H, Furugori M, Hashimoto M, Mukaide T, Kamatani J, Igawa S, Tsuboyama A, Takiguchi T, Ueno K (2005) Substituent effects of iridium complexes for highly efficient red OLEDs. Dalton Trans 9:1583–1590
Zhang LY, Liu GF, Zheng SL, Ye BH, Zhang XM, Chen XM (2003) Helical ribbons of cadmium(II) and zinc(II) dicarboxylates with bipyridyl-like chelates − syntheses, crystal structures and photoluminescence. Eur J Inorg Chem 2003:2965–2971
Wu FI, Su HJ, Shu CF, Luo LY, Diau WG, Cheng CH, Duan JP, Lee GH (2005) Tuning the emission and morphology of cyclometalated iridium complexes and their applications to organic light-emitting diodes. J Mater Chem 15:1035–1042
Thomas KRJ, Velusamy M, Lin JT, Chien CH, Tao YT, Wen YS, Hu YH, Chou PT (2005) Efficient red-emitting cyclometalated iridium(III) complexes containing lepidine-based ligands. Inorg Chem 44:5677–5685
Lee W, Kwon TH, Kwon J, Kim JY, Lee C, Hong JI (2011) Effect of main ligands on organic photovoltaic performance of Ir(III) complexes. New J Chem 35:2557–2563
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 21501037), the Natural Science Foundation of Hainan Province (No. 20152017), the Science and Research Project of Education Department of Hainan Province (Nos. Hjkj2013-25 and Hnky2015-27) and Hainan Normal University’s Innovation Experiment Program for University Students (No. cxcyxj2015005).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
10895_2015_1718_MOESM1_ESM.pdf
Crystallographic data (excluding structure factors) for the structural analysis have been deposited with the Cambridge Crystallographic Data Center as supplementary publication No. CCDC 1412058 (3b). Copies of the data can be obtained free of charge via www.ccdc.ac.uk/conts/retrieving.html (or from The Director, CCDC, 12 Union Road, Cambridge CB2 1EZ, UK, Fax: +44-1223-336-033. E-mail: deposit@ccdc.cam.ac.uk.). (PDF 170 kb)
Rights and permissions
About this article
Cite this article
Li, GN., Zeng, YP., Li, KX. et al. Synthesis, Characterization, Properties and DFT Calculations of 2-(Benzo[b]thiophen-2-yl)pyridine-based Iridium(III) Complexes with Different Ancillary Ligands. J Fluoresc 26, 323–331 (2016). https://doi.org/10.1007/s10895-015-1718-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-015-1718-7