Abstract
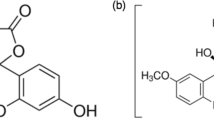
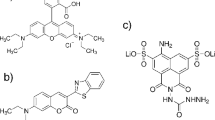
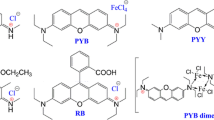
The fluorescence lifetime (τf), emission quantum yield (Φf), absorption and emission spectral data of 20 fluorescein derivatives were measured under the same conditions by using time-correlated single photon counting, steady state fluorescence and absorption methods to get comparable data. Based on the results, the factors and mechanism that control the fluorescence properties of the fluorescein dyes are discussed. Both Φf and τf are remarkably dependent on the substitution on either xanthene or phenyl rings, but their ratio (Φf/τf), i.e. rate constant of radiation process, is a constant value (0.20 × 109 s−1). The rate constant of nonradiation process, on the other hand, is varied with both the structure and the solvent used.



Similar content being viewed by others
References
Lane BC, Cohen-Gadol AA (2013) Fluorescein fluorescence use in the management of intracranial neoplastic and vascular lesions: a review and report of a new technique. Curr Drug Discov Technol 10:160–169
Rey-Dios R, Cohen-Gadol AA (2013) Technical principles and neurosurgical applications of fluorescein fluorescence using a microscope-integrated fluorescence module. Acta Neurochir 155:701–706
Patil VS, Padalkar VS, Phatangare KR, Gupta VD, Umape PG, Sekar N (2012) Synthesis of new ESIPT-fluorescein: photophysics of pH sensitivity and fluorescence. J Phys Chem A 116:536–545
Egawa T, Koide Y, Hanaoka K, Komatsu T, Terai T, Nagano T (2011) Development of a fluorescein analogue, TokyoMagenta, as a novel scaffold for fluorescence probes in red region. Chem Commun 47:4162–4164
Kobayashi H, Ogawa M, Alford R, Choyke PL, Urano Y (2010) New strategies for fluorescent probe design in medical diagnostic imaging. Chem Rev 110:2620–2640
Yao H, Jockusch RA (2013) Fluorescence and electronic action spectroscopy of mass-selected gas-phase fluorescein, 2′-7′-dichlorofluorescein, and 2′-7′-difluorofluorescein ions. J Phys Chem A 117:1351–1359
Zhang XF, Zhang I, Liu L (2010) Photophysics of halogenated fluoresceins: involvement of both intramolecular electron transfer and heavy atom effect in the deactivation of excited states. Photochem Photobiol 86:492–498
Siejak P, Fraückowiak D (2005) 64 spectral properties of fluorescein molecules in water with the addition of a colloidal suspension of silver. J Phys Chem B
Orte A, Crovetto L, Talavera EM, Boens N, Alvarez-Pez JM (2005) 73 absorption and emission study of 2′,7′-difluorofluorescein and its excited-state buffer-mediated proton exchange reactions. J Phys Chem A 109:734–737
Magde D, Wong R, Seybold PG (2002) 25 fluorescence quantum yields and their relation to lifetimes of rhodamine 6G and fluorescein in nine solvents: improved absolute standards for quantum yields. Photochem Photobiol 75:327–334
Biswas S, Bhattacharya SC, Sen PK, Moulik SP (1999) 51 absorption and emission spectroscopic studies of fluorescein dye in alkanol, micellar and microemulsion media. J Photochem Photobiol A Chem 123:121–128
Magde D, Rojas GE, Seybold PG (1999) 39 solvent dependence of the fluorescence lifetimes of xanthene dyes. Photochem Photobiol 70:737–744
Sjoback R, Nygren J, Kubista M (1995) * 31absorption and fluorescence properties of fluorescein. Spectrochim Acta A 51:L7–L21
Fleming GR, Knight AWE, Morris JM, Morrison RJS, Robinson GW (1977) 18 picosecond fluorescence studies of xanthene dyes. J Am Chem Soc 99:4306–4311
Forster LS, Dudley D (1962) 19 the luminescence of fluorescein dyes. J Phys Chem 66:838–840
Zhang X-F (2010) The effect of phenyl substitution on the fluorescence characteristics of fluorescein derivatives via intramolecular photoinduced electron transfer. Photochem Photobiol Sci 9:1261–1268
Zhang XF, Liu Q, Wang H, Zhang F, Zhao F (2008) Prototropic equilibria, tautomerization and electronic absorption properties of dibenzofluorescein in aqueous solution related to its capability as a fluorescence probe. Photochem Photobiol Sci 7:1079–1084
Zhang XF, Liu Q, Wang H, Fu Z, Zhang F (2008) 75 photophysical behavior of lipophilic xanthene dyes without the involvement of photoinduced electron transfer mechanism. J Photochem Photobiol A Chem 200:307–313
Zhang XF, Liu Q, Son A, Zhang Q, Zhao F, Zhang F (2008) Fluorescence properties of dibenzofluorescein in aqueous solution. J Fluoresc 18:1051–1057
Zhang X, Liu Q, Son A, Zhang Q, Zhang F, Zhao F (2008) 13 photophysical properties of dibenzofluorescein and the presence of its tautomers or prototropic forms in organic solvents. Photochem Photobiol Sci 7:299–302
Zhang X-F, Zhang Y, Liu L (2013) Fluorescence lifetimes and quantum yields of ten rhodamine derivatives: structural effect on emission mechanism in different solvents. J Lumin 145:448–453
Valdes-Aguilera O, Neckers DC (1989) Aggregation phenomena in xanthene dyes. Acc Chem Res 22:171–177
McHedlov-Petrossyan NO, Rubtsov MI, Lukatskaya LL (1992) Ionization and tautomerism of chloro-derivatives of fluorescein in water and aqueous acetone. Dye Pigment 18:179–198
McHedlov-Petrossyan NO, Kukhtik VI, Alekseeva VI (1994) Ionization and tautomerism of fluorescein, rhodamine B, N, N-diethylrhodol and related dyes in mixed and nonaqueous solvents. Dye Pigment 24:11–35
Tremayne M, Kariuki BM, Harris KDM (1997) Structure determination of a complex organic solid from x-ray powder diffraction data by a generalized monte carlo method: the crystal structure of red fluorescein. Angew Chem Int Ed 36:770–772
Acknowledgments
This work has been supported partially by Hebei Provincial Science Foundation (Contract B2010001518) and HBUST (Contract CXTD2012-05).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, XF., Zhang, J. & Liu, L. Fluorescence Properties of Twenty Fluorescein Derivatives: Lifetime, Quantum Yield, Absorption and Emission Spectra. J Fluoresc 24, 819–826 (2014). https://doi.org/10.1007/s10895-014-1356-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-014-1356-5