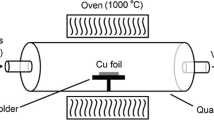
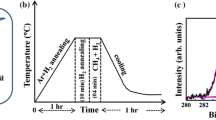
Abstract
We present a comprehensive study of the parameter for graphene growth by low pressure chemical vapor deposition on Cu foil. The growth of graphene was investigated in various conditions, changing the gas pressures, gas ratio, growth temperature and growth time. The synthesized graphene were characterized using Raman spectroscopy and Field Emission Scanning Electron Microscopy (FESEM). By varying the growth time it can be concluded that the domain size increases when the growth time increases. The absence of 2D band in this section suggest that the produced graphene-like films are not monolayer and graphite can be formed in this growth conditions. The FESEM images demonstrate that increasing growth temperature while holding other parameters constant yields in larger domains which may be due to a faster growth at the higher temperature. The Raman spectra also showed the recovery of the 2D peak by increasing the growth temperature, indicating the crystallization of the damaged graphene layer. Hence the quality of grown graphene-like films is highly correlated to the process temperature. We show that the methane-to-hydrogen ratio is critical parameters that affect the structural perfection of graphene like domains. The Raman studies denote graphene like carbon nature due to the appearance of significant G and 2D peaks. Also the 2D peak intensity increases by increasing the H2 concentration in gas mixture, indicating a low graphene layers. The resulting domain size and the nucleation density are maximum at maximum value of H2 concentration. We can conclude that by controlling the growth parameters, the morphology and structure of graphene can be changed. We suggest that graphene can be used for fusion reactor first wall due to its unique physical and chemical properties. Our results provide important guidance toward the synthesis of high quality and uniform graphene films.









Similar content being viewed by others
References
A. Litnovsky, P. Wienhold, V. Philipps et al., J. Nucl. Mater. 363–365, 1395 (2007)
A. Litnovsky, D.L. Rudakov, G. De Temmerman et al., Fusion Eng. Des. 83, 79 (2008)
C. Li, Q. Huang, Y. Feng et al., Plasma Sci. Technol. 9, 484–487 (2007)
O.I. Buzhinskij, V.A. Barsuk, V.G. Otroshchenko, J. Nucl. Mater. 390–391, 996 (2009)
V.K. Gusev et al., J. Nucl. Mater. 386–388, 708 (2009)
C.P.C. Wong, J. Nucl. Mater. 390–391, 1026 (2009)
M. Shimada, R.A. Pitts, J. Nucl. Mater. 415, S1013 (2011)
T. Yamashina, T. Hino, J. Nucl. Sci. Technol. 27(7), 589–600 (1990)
A. Matsumoto, Y. Yamauchi, T. Hino, Y. Ueda, K. Nishimura, Plasma Fusion Res. Regul. Artic. 7, 2402090 (2012)
A.A. Balandin, Nat. Mater. 10, 569–581 (2011)
R. Faccio, P.A. Denis, H. Pardo, C. Goyenola, Á.W. Mombrú, J. Phys.: Condens. Matter 21, 285304 (2009)
A.K. Geim, K.S. Novoselov, The rise of graphene. Nat. Mater. 6, 183–191 (2007)
J.C. Meyer, A.K. Geim, M.I. Katsnelson, K.S. Novoselov, T.J. Booth, S. Roth, The structure of suspended graphene sheets. Nature 446, 60–63 (2007)
A.K. Geim, Graphene: status and prospects. Science 324, 1530–1534 (2009)
S. Gilje, S. Han, M.S. Wang, K.L. Wang, R.B. Kaner, A chemical route to graphene for device applications. Nano Lett. 7, 3394–3398 (2007)
K.S. Novoselov, A.K. Geim, S.V. Morozov, D. Jiang, Y. Zhang, S.V. Dubonos et al., Electric field effect in atomically thin carbon films. Science 306, 666–669 (2004)
C. Riedl, C. Coletti, T. Iwasaki, A.A. Zakharov, U. Starke, Quasi-free-standing epitaxial graphene on SiC obtained by hydrogen intercalation. Phys. Rev. Lett. 103(24), 246804 (2009)
J. Kraus, S. Bocklein, R. Reichelt, S. Gunther, B. Santos, T.O. Mentes, A. Locatelli, Towards the perfect graphene membrane-improvement and limits during formation of high quality graphene grown on Cu-foils. Carbon 64, 377–390 (2013)
J. Zhang, P. Hu, X. Wang, Z. Wang, Structural evolution and growth mechanism of graphene domains on copper foil by ambient pressure chemical vapor deposition. Chem. Phys. Lett. 536, 123–128 (2012)
A. Kumar, A.A. Voevodin, D. Zemlyanov, D.N. Zakharov, T.S. Fisher, Rapid synthesis of few layer graphene over Cu foil. Carbon 50, 1546–1553 (2012)
M. Sarno, C. Cirillo, R. Piscitelli, P. Ciambelli, A study of the key parameters, including the crucial role of H2 for uniform graphene growth on Ni foil. J. Mol. Catal. A Chem. 366, 303–314 (2013)
A. Ferrari, J. Meyer, V. Scardaci, C. Casiraghi, M. Lazzeri, F. Mauri, S. Piscanec, D. Jiang, K. Novoselov, S. Roth, A. Geim, Raman spectrum of graphene and graphene layers. Phys. Rev. Lett. 97(18), 187401 (2006)
S.P. Park, J.H. An, R.D. Piner, I. Jung, D.X. Yang, A. Velamakanni et al., Aqueous suspension and characterization of chemically modified graphene sheets. Chem. Mater. 20, 6592–6594 (2008)
W. Bao, F. Miao, Z. Chen, H. Zhang, W. Jang, C. Dames et al., Controlled ripple texturing of suspended graphene and ultrathin graphite membranes. Nat. Nanotechnol. 4, 562–566 (2009)
J.A. Venables, G.D.T. Spiller, M. Hanbucken, Nucleation and growth of thin films. Rep. Prog. Phys. 47, 399 (1984)
V.N.E. Robinson, J.L. Robins, Observation of nucleation processes. Thin Solid Films 20, 155 (1974)
P.F. Williams, S.P.S. Porto, Symmetry-forbidden resonant Raman scattering in Cu2O. Phys. Rev. B 8, 1782–1785 (1973)
C. Thomsen, S. Reich, Double resonant Raman scattering in graphite. Phys. Rev. Lett. 85, 5214–5217 (2000)
A.C. Ferrari, J. Robertson, Interpretation of Raman spectra of disordered and amorphous carbon. Phys. Rev. B 61, 14095–14107 (2000)
A.C. Ferrari, J. Robertson, Resonant Raman spectroscopy of disordered, amorphous, and diamondlike carbon. Phys. Rev. B 64, 075414 (2001)
J. Maultzsch, S. Reich, C. Thomsen, Double-resonant Raman scattering in graphite: interference effects, selection rules, and phonon dispersion. Phys Rev B 70, 155403 (2004)
G.V. Saparin, Microcharacterization of CVD diamond films by scanning electronmicroscopy: morphology, structure and microdefects. Diam. Relat. Mater. 3, 1337–1351 (1994)
M. Losurdo, M.M. Giangregorio, P. Capezzuto, G. Bruno, Graphene CVD growth on copper and nickel: role of hydrogen in kinetics and structure. Phys. Chem. Chem. Phys. 13(46), 20836–20843 (2011)
E. Vaghri, Z. Khalaj, M. Ghoranneviss, Characterization of diamond: like carbon films synthesized by DC-plasma enhanced chemical vapor deposition. J Fusion Energ 30, 447–452 (2011)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jafari, A., Ghoranneviss, M. & Hantehzadeh, M.R. Morphology Control of Graphene by LPCVD. J Fusion Energ 34, 532–539 (2015). https://doi.org/10.1007/s10894-014-9836-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10894-014-9836-9