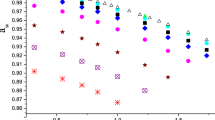
An analysis has been made of the literature data on sucrose hydration. A platform of the most reliable experimental data has been selected. In a differential evaporation microcalorimeter, the specific heat of evaporation of water was measured in the process of continuous dehydration of a solution with an initial content of sucrose of 12.5 wt.%. Curves of the reduced specific evaporation heat versus the concentration of the solution at 40, 60, and 80°C were obtained. The influence of the degree of hydration of sucrose on the specific evaporation heat was determined. It has been shown that in the interval from the initial concentration of the solution, with degree of hydration of sucrose N = 20, to a content of sucrose of ~65 wt.% (N ~ 6), the specific evaporation heat grows from a value that is similar to the evaporation heat of pure water to values 3 to 4% higher than it. The most significant growth in the specific evaporation heat (7–10%) was recorded on removing water bonded to hydroxyl groups of sucrose in the equatorial position at N ≤ 6. The concept of strongly and weakly bound hydrate water in sucrose solutions is substantiated.
Similar content being viewed by others
References
World Market of Sugar; http://www.webeconomy.ru/index.php?page=cat&newsid=2257&type=news/.
World Production of Sugar in the New Season Will Reach Record Highs; http://www.agroinvestor.ru/markets/news/27524-mirovoe-proizvodstvo-sakhara/.
P. M. Silin, The Technology of Sugar [in Russian], 2nd revised and enlarged edn., Pishchevaya Promyshlennost′, Moscow (1967).
I. L. Kanunyats (Editor-in-Chief), Chemical Encyclopedia. In Five Volumes. Vol. 2. Daffa-Media X46 [in Russian], Sovetskaya Éntsiklopediya, Moscow (1990).
L. D. Bobrovnik, I. S. Gulyi, and V. M. Klimovich, Hydration and phase transformations of sucrose, Sakharnaya Prom., No. 6, 10–11 (1992).
A. T. Allen and R. M. Wood, Molecular association in the sucrose-water system, Sugar Technol. Rev., 2, No. 12, 165–179 (1974).
N. V. Troitskii, Sucrose hydrates in aqueous solutions, Sakharnaya Prom., No. 4, 38–39 (1947).
A. G. Pasynskii, Solvation of nonelectrolytes and compressibility of their solutions, Zh. Fiz. Khim., 20, No. 9, 981–994 (1946).
S. E. Kharin and A. A. Kniga, On aqueous sugar solutions, Izv. Vyssh. Uchebn. Zaved., Pishchevaya Tekhnol., No. 5, 47–51 (1963).
E. I. Akhumov, Hydration of sucrose in solutions, Zh. Prikl. Khim., 48, No. 2, 458–460 (1975).
V. I. Buravkeva, A. V. Zubchenko, and A. Ya. Oleinikova, On the problem of hydration of sugars, Izv. Vyssh. Uchebn. Zaved., Pishchevaya Tekhnol., No. 5, 129–131 (1977).
S. E. Kharin, G. S. Sorokina, and L. L. Kniga, Degree of hydration of sucrose in aqueous sugar solutions, Izv. Vyssh. Uchebn. Zaved., Pishchevaya Tekhnol., No. 4, 75–77 (1973).
E. A. Kolosovskaya, S. R. Loskutov, and B. S. Chudinov, Physical Principles of Interaction of Wood and Water [in Russian], Nauka, Novosibirsk (1989).
J. E. Carles and A. M. Scallan, The determination of the amount of bound water within cellulosic gels by NMR spectroscopy, J. Polym. Sci., 17, No. 6, 1855–1865 (1973).
D. Simatos, M. Faure, E. Bonjour, and M. Couch, Application of differential thermal analysis and differential scanning calorimetry in studying water in foods, in: R. B. Duckworth (Ed.), Water in Foods [Russian translation], Pishchevaya Promyshlennost′, Moscow (1980), pp. 156–170.
S. Deodar and Ph. Luner, Measuring the content of bound (nonfeeezing) water by the differential-scanning-calorimetry method, in: S. Rowland (Ed.), Water in Polymers [Russian translation], Mir, Moscow (1984), pp. 273–287.
O. Parniakov, O. Bals, F. J. Barba, V. Mykhailyk, N. Lebovka, and E. Vorobiev, Application of differential scanning calorimetry to estimate quality and nutritional properties of food products, Crit. Rev. Food Sci. Nutr., 58, No. 3, 362–385 (2016).
V. A. Mykhailyk, Yu. F. Snezhkin, and N. V. Dmitrenko, Investigation of the state of water in energy trees in the process of drying by differential scanning calorimetry, J. Eng. Phys. Thermophys., 88, No. 5, 1093–1099 (2015).
Jean-Joseph Max and Camille Chapados, Sucrose hydrates in aqueous solution by IR spectroscopy, J. Phys. Chem. A, 105, No. 47, 10681–10688 (2001).
F. Franks, The properties of aqueous solutions at temperatures below 0°C, in: F. Franks (Ed.), Water and Aqueous Solutions at Temperatures below 0°C [Russian translation], Naukova Dumka, Kiev (1985), pp. 176–276.
F. Franks, Water, ice, and solutions of simple molecules, in: R. B. Duckworth (Ed.), Water in Foods [Russian translation], Pishchevaya Promyshlennost′, Moscow (1980), pp. 14–32.
V. A. Mikhailik, E. O. Davydova, and V. V. Mank, Investigation of the hydration of sucrose by the low-temperature scanning calorimetry method, in: Thermodynamics of Organic Compounds [in Russian], Gor′kii (1989), pp. 76–80.
V. A. Mykhailyk, Experimental study of the hydration of sucrose, Naukovi Prats. Odes′sk. Nats. Akad. Kharch. Tekhnol., 2, No. 28, 370–373 (2006).
V. A. Mykhailyk, Application of thermal investigation methods in solving scientific and production energy-efficiency problems, Naukovi Prats. Odes′sk. Nats. Akad. Kharch. Tekhnol., 1, No. 31, 170–177 (2007).
R. S. Burdukova, M. N. Dadenkova, L. P. Zhmyrya, A. I. Orel, and B. S. Sluchanko, On certain properties of sugars and their aqueous solutions, Izv. Vyssh. Uchebn. Zaved., Pishchevaya Tekhnol., No. 3, 37–45 (1972).
L. D. Bobrivnyk, Technological aspects of hydration of sucrose, Naukovi Prats. UDUKhT, No. 7, 44–47 (2000).
V. A. Mikhailik, Behavioral features of solutions of sugars in dehydrating, in: Proc. Third Int. Sci.-Pract. Conf. "Modern Energy-Saving Thermal Technologies (Drying and Thermal-Wet Treatment of Materials) SÉTT-2008," September 16–20, 2008, Moscow–Tambov (2008), Vol. 1, pp. 240–242.
Yu. F. Snézhkin, L. V. Dekusha, N. S. Dubovikova, T. G. Grishchenko, L. I. Vorob′iov, and L. A. Boryak, Calorimeter Device for Determining the Specific Heat of Evaporation of Moisture and Organic Substances from Materials, Ukraine Patent No. 84075, G01 N25/26, G01 N25/28. Published 10.09.08. Bull. No.17.
N. S. Dubovikova, Yu. F. Snezhkin, L. V. Dekusha, and L. I. Vorob′ev, Thermometer device of simultaneous thermal analysis to determine specific evaporation heat, Prom. Teplotekh., 35, No. 2, 87–95 (2013).
W. Hemminger and G. W. H. Höhne, Calorimetry. Theory and Practice [Russian translation], Khimiya, Moscow (1990).
M. D. Veisband and V. I. Pronenko, Technique of Performing Metrological Works [in Russian], Tekhnika, Kiev (1986).
A. A. Aleksandrov and B. A. Grigor′ev, Tables of Thermophysical Properties of Water and Steam: a Handbook [in Russian], MÉI, Moscow (1999).
P. Honig (Ed.), Principles of Sugar Technology [Russian translation], Pishchepromizdat, Moscow (1961).
R. Ts. Mishchuk, L. G. Belostotskii, and S. I. Sagan′, Activation energy of formation of crystallization sites in a sugar solution, Sakharnaya Prom., No. 10, 28–30 (1983).
I. A. Kukharenko, New theory of solutions, in: I. A. Kukharenko (Ed.), Scientific-Research Department of Farm Production Technology, Vol. 1 [in Russian], Izd. Nauchn.-Issled. Kafedry Tekhnol Sel′skokhoz. Proizvodstv, Kiev (1924), pp. 10–14.
A. J. Nanassy and R. L. Desai, NMR shows how ammoniacal primer-sealers alter the hygroscopicity of wood, Wood Sci., 10, No. 4, 204–207 (1978).
L. D. Bobrovnik, A. M. Grekhov, and I. S. Gulyi, Energy characteristics of structures in aqueous solutions of sucrose, Zh. Strukt. Khim., 39, No. 5, 860–867 (1998).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Inzhenerno-Fizicheskii Zhurnal, Vol. 92, No. 4, pp. 945–952, July–August, 2019.
Rights and permissions
About this article
Cite this article
Mikhailik, V.A., Dmitrenko, N.V. & Snezhkin, Y.F. Investigation of the Influence of Hydration on the Heat of Evaporation of Water From Sucrose Solutions. J Eng Phys Thermophy 92, 916–922 (2019). https://doi.org/10.1007/s10891-019-02003-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10891-019-02003-8