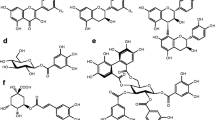
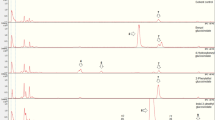
Abstract
We developed a combination of methods to estimate the alkaline oxidative conditions of the midgut of insect larvae and to reveal the alkaline and enzymatic oxidative activities for individual phenolic compounds present in the larval host plants. First, we monitored the in vitro isomerization of 5-O-caffeoylquinic acid (5-CQA) into 3-CQA, 4-CQA and 5-CQA at pH 9.0–11.0. Then we calculated the isomer ratios of 3-CQA, 4-CQA and 5-CQA from the frass of eight species of insect herbivores fed on foliage containing 5-CQA. The isomer ratios suggested that the midgut pH of these larvae ranged from 9.4 to around 10.1. Second, we developed an in situ enzymatic oxidation method that enabled oxidation of phenolics in a frozen plant sample at 30 °C by species- and tissue-specific enzymes. Then we measured the alkaline and enzymatic oxidative activities of the individual phenolics in 20 plant species by quantifying the proportion of the compound concentration lost due to the auto-oxidation of a plant extract at pH 10 and due to the enzymatic oxidation of the frozen plant sample at 30 °C. Our results showed that both of the oxidative activity types depended primarily on the type of phenolic compound, but the enzymatic oxidative activity depended also on the plant species and tissue type. This combination of methods offers an approach to characterize a wide array of phenolics that are susceptible to oxidation by the plant enzymes and/or by the alkaline conditions estimated to prevail in the insect midgut. We propose that these kinds of compound-specific results could guide future studies on specific plant-herbivore interactions to focus on the phenolics that are likely to be active rather than inactive plant phenolics.




Similar content being viewed by others
References
Aniszewski T, Lieberei R, Culewicz K (2008) Research on catecholases, laccases and cresolases in plants. Recent progress and future needs. Acta Biol Cracoviensia 50:7–18
Appel HM (1993) Phenolics in ecological interactions: the importance of oxidation. J Chem Ecol 19:1521–1552
Baert N, Karonen M, Salminen J-P (2015) Isolation, characterisation and quantification of the main oligomeric macrocyclic ellagitannins in Epilobium angustifolium by ultra-high performance chromatography with diode array detection and electrospray tandem mass spectrometry. J Chromatogr A 1419:26–36. https://doi.org/10.1016/j.chroma.2015.09.050
Barbehenn R, Dodick T, Poopat U, Spencer B (2005) Fenton-type reactions and iron concentrations in the midgut fluids of tree-feeding caterpillars. Arch Insect Biochem Physiol 60:32–43. https://doi.org/10.1002/arch.20079
Barbehenn RV, Jones CP, Hagerman AE, Karonen M, Salminen JP (2006a) Ellagitannins have greater oxidative activities than condensed tannins and galloyl glucoses at high pH: potential impact on caterpillars. J Chem Ecol 32:2253–2267. https://doi.org/10.1007/s10886-006-9143-7
Barbehenn RV, Jones CP, Karonen M, Salminen J-P (2006b) Tannin composition affects the oxidative activities of tree leaves. J Chem Ecol 32:2235–2251. https://doi.org/10.1007/s10886-006-9142-8
Clifford MN (2000) Chlorogenic acids and other cinnamates – nature, occurrence, dietary burden, absorption and metabolism. J Sci Food Agric 80:1033–1043. https://doi.org/10.1002/(SICI)1097-0010(20000515)80:7<1033::AID-JSFA595>3.0.CO;2-T
Constabel CP, Yip L, Patton JJ, Christopher ME (2000) Polyphenol oxidase from hybrid poplar. Cloning and expression in response to wounding and herbivory. Plant Physiol 124:285–295. https://doi.org/10.1104/pp.124.1.285
Engström MT, Pälijärvi M, Fryganas C, Grabber JH, Mueller-Harvey I, Salminen JP (2014) Rapid qualitative and quantitative analyses of proanthocyanidin oligomers and polymers by UPLC-MS/MS. J Agric Food Chem 62:3390–3399. https://doi.org/10.1021/jf500745y
Engström MT, Pälijärvi M, Salminen J-P (2015) Rapid fingerprint analysis of plant extracts for ellagitannins, gallic acid, and quinic acid derivatives and quercetin-, kaempferol- and myricetin-based flavonol glycosides by UPLC-QqQ-MS/MS. J Agric Food Chem 63:4068–4079. https://doi.org/10.1021/acs.jafc.5b00595
Felton GW, Donato KK, Broadway RM, Duffey SS (1992) Impact of oxidized plant phenolics on the nutritional quality of dietar protein to a noctuid herbivore, Spodoptera exigua. J Insect Physiol 38:277–285. https://doi.org/10.1016/0022-1910(92)90128-Z
Haddock EA, Gupta RK, Al-Shafi SMK et al (1982) The metabolism of gallic acid and hexahydroxydiphenic acid in plants. Part 1. Introduction. Naturally occurring galloyl esters. J Chem Soc Perkin Trans 1:2515–2524. https://doi.org/10.1039/P19820002515
Haribal M, Renwick JAA (2001) Seasonal and population variation in flavonoid and alliarinoside content of Alliaria petiolata. J Chem Ecol 27:1585–1594. https://doi.org/10.1023/A:1010406224265
Harrison J (2001) Insect acid-base physiology. Annu Rev Entomol 46:221–250
Herrmann K, Nagel CW (1989) Occurrence and content of hydroxycinnamic and hydroxybenzoic acid compounds in foods. Crit Rev Food Sci Nutr 28:315–347
Hunter MD, Hull LA (1993) Variation in concentrations of phloridzin and phloretin in apple foliage. Phytochemistry 34:1251–1254. https://doi.org/10.1016/0031-9422(91)80010-X
Jakešová H, Řepková J, Nedělník J, Hampel D, Dluhošová J, Soldánová M, Ošťádalová M (2016) Selecting plants with increased total polyphenol oxidases in the genus Trifolium. Czech J Genet Plant Breed 51:155–161. https://doi.org/10.17221/107/2015-CJGPB
Johnson KS, Barbehenn RV (2000) Oxygen levels in the gut lumens of herbivorous insects. J Insect Physiol 46:897–903. https://doi.org/10.1016/S0022-1910(99)00196-1
Karonen M, Hämäläinen M, Nieminen R, Klika KD, Loponen J, Ovcharenko VV, Moilanen E, Pihlaja K (2004) Phenolic extractives from the bark of Pinus sylvestris L. and their effects on inflammatory mediators nitric oxide and prostaglandin E2. J Agric Food Chem 52:7532–7540. https://doi.org/10.1021/jf048948q
Lahtinen M, Kapari L, Ossipov V, Salminen JP, Haukioja E, Pihlaja K (2005) Biochemical transformation of birch leaf phenolics in larvae of six species of sawflies. Chemoecology 15:153–159. https://doi.org/10.1007/s00049-005-0307-7
Lerch K (1995) Tyrosinase: molecular and active-site structure. In: Lee CY, Whitaker JR (eds) Enzymatic Browning and its prevention. American Chemical Society, Washington, DC, pp 64–80
Marsh K, Zhou W, Wigley HJ et al (2017) Oxidizable phenolic concentrations do not affect development and survival of Paropsis atomaria larvae eating Eucalyptus foliage. J Chem Ecol 43:411–421. https://doi.org/10.1007/s10886-017-0835-y
Martz F, Turunen M, Julkunen-Tiitto R, Lakkala K, Sutinen ML (2009) Effect of the temperature and the exclusion of UVB radiation on the phenolics and iridoids in Menyanthes trifoliata L. leaves in the subarctic. Environ Pollut 157:3471–3478. https://doi.org/10.1016/j.envpol.2009.06.022
Mayer AM, Harel E (1979) Polyphenol oxidases in plants. Phytochemistry 18:193–215. https://doi.org/10.1016/0031-9422(79)80057-6
Moilanen J, Salminen J-P (2008) Ecologically neglected tannins and their biologically relevant activity: chemical structures of plant ellagitannins reveal their in vitro oxidative activity at high pH. Chemoecology 18:73–83. https://doi.org/10.1007/s00049-007-0395-7
Moilanen J, Sinkkonen J, Salminen J-P (2013) Characterization of bioactive plant ellagitannins by chromatographic, spectroscopic and mass spectrometric methods. Chemoecology 23:165–179. https://doi.org/10.1007/s00049-013-0132-3
Moilanen J, Koskinen P, Salminen J-P (2015) Distribution and content of ellagitannins in Finnish plant species. Phytochemistry 116:188–197. https://doi.org/10.1016/j.phytochem.2015.03.002
van Nieukerken EJ, Kaila L, Kitching IJ et al (2011) Order Lepidoptera Linnaeus, 1758. In: Zhang, Z-Q (ed) Animal Biodiversity: An outline of higher-level classification and survey of taxonomic richness. Magnolia Press, Auckland, pp 212–221
Olszewska MA, Kwapisz A (2011) Metabolite profiling and antioxidant activity of Prunus padus L. flowers and leaves. Nat Prod Res 25:1115–1131. https://doi.org/10.1080/14786410903230359
Pinelli P, Ieri F, Vignolini P, Bacci L, Baronti S, Romani A (2008) Extraction and HPLC analysis of phenolic compounds in leaves, stalks, and textile fibers of Urtica dioica L. J Agric Food Chem 56:9127–9132. https://doi.org/10.1021/jf801552d
Polasek J, Queiroz EF, Hostettmann K (2007) On-line identification of phenolic compounds of Trifolium species using HPLC-UV-MS and post-column UV-derivatisation. Phytochem Anal 18:13–23. https://doi.org/10.1002/pca.946
Ravn H, Brimer L (1988) Structure and antibacterial activity of plantamajoside, a caffeic acid sugar ester from Plantago major subsp. major. Phytochemistry 27:3433–3437. https://doi.org/10.1016/0031-9422(88)80744-1
Ruuhola T, Yang S (2006) Wound-induced oxidative responses in mountain birch leaves. Ann Bot 97:29–37. https://doi.org/10.1093/aob/mcj005
Salminen J-P (2014) The chemistry and chemical ecology of ellagitannins in plant–insect interactions: from underestimated molecules to bioactive plant constituents. In: Romani A, Lattanzio V, Quideau S (eds) Recent advances in polyphenol research, vol 4, 1st edn. John Wiley & Sons, Ltd., pp 83–113
Salminen J-P (2018) Metabolism of 14C-labelled pentagalloylglucose by Epirrita autumnata and Agriopis aurantiaria (Lepidoptera: Geometridae) and implications for the nutrition of geometrid defoliators. Austral Entomology (available online). https://doi.org/10.1111/aen.12323
Salminen J-P, Karonen M (2011) Chemical ecology of tannins and other phenolics: we need a change in approach. Funct Ecol 25:325–338. https://doi.org/10.1111/j.1365-2435.2010.01826.x
Salminen J-P, Lempa K (2002) Effects of hydrolysable tannins on a herbivorous insect: fate of individual tannins in insect digestive tract. Chemoecology 12:203–211. https://doi.org/10.1007/PL00012670
Salminen J-P, Lahtinen M, Lempa K et al (2004) Metabolic modifications of birch leaf phenolics by an herbivorous insect: detoxification of flavonoid aglycones via glycosylation. Zeitschrift für Naturforsch 59:437–444
Schütz K, Kammerer DR, Carle R, Schieber A (2005) Characterization of phenolic acids and flavonoids in dandelion (Taraxacum officinale WEB. Ex WIGG.) root and herb by high-performance liquid chromatography/electrospray ionization mass spectrometry. Rapid Commun Mass Spectrom 19:179–186. https://doi.org/10.1002/rcm.1767
Sunnerheim K, Palo RT, Theander O, Knutsson P-G (1988) Chemical defense in birch. Platyphylloside: a phenol from Betula pendula inhibiting digestibility. J Chem Ecol 14:549–560
Teslov LS, Koretskaya LN, Tsareva GI (1983) Phenolic compounds of Campanula rotundifolia and C. persicifolia. Khimiya Prir Soedin (3):387
Thiboldeaux RL, Lindroth RL, Tracy JW (1998) Effects of juglone (5-hydroxy-1,4-naphthoquinone) on midgut morphology and glutathione status in Saturniid moth larvae. Comp Biochem Physiol C Pharmacol Toxicol Endocrinol 120:481–487. https://doi.org/10.1016/S0742-8413(98)10070-1
Tran LT, Taylor JS, Constabel CP (2012) The polyphenol oxidase gene family in land plants: lineage-specific duplication and expansion. BMC Genomics 13:395. https://doi.org/10.1186/1471-2164-13-395
Tuominen A, Sundman T (2013) Stability and oxidation products of hydrolysable tannins in basic conditions detected by HPLC/DAD-ESI/QTOF/MS. Phytochem Anal 24:424–435. https://doi.org/10.1002/pca.2456
Vihakas M, Pälijärvi M, Karonen M, Roininen H, Salminen JP (2014) Rapid estimation of the oxidative activities of individual phenolics in crude plant extracts. Phytochemistry 103:76–84. https://doi.org/10.1016/j.phytochem.2014.02.019
Vihakas M, Gómez I, Karonen M, Tähtinen P, Sääksjärvi I, Salminen JP (2015) Phenolic compounds and their fates in tropical lepidopteran larvae: modifications in alkaline conditions. J Chem Ecol 41:822–836. https://doi.org/10.1007/s10886-015-0620-8
Yoruk R, Marshall MR (2003) Physicochemical properties and function of plant polyphenol oxidase: a review. J Food Biochem 27:361–422. https://doi.org/10.1111/j.1745-4514.2003.tb00289.x
Acknowledgements
Vladimir Ossipov is acknowledged for the valuable scientific discussions about this topic. The anonymous referees and journal editors are acknowledged for their assistance with the earlier versions of the manuscript. Anne Koivuniemi and Marica Engström are acknowledged for collecting the plant samples, and Suvi Vanhakylä for species identification. The study was funded by Kone Foundation (J-P S) and the Academy of Finland (grant 258992 to J-P S). Purchase of the UHPLC-DAD-MS system was made possible by a Strategic Research Grant of the University of Turku (Ecological Interactions).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(PDF 1490 kb)
Rights and permissions
About this article
Cite this article
Kim, J., Pälijärvi, M., Karonen, M. et al. Oxidatively Active Plant Phenolics Detected by UHPLC-DAD-MS after Enzymatic and Alkaline Oxidation. J Chem Ecol 44, 483–496 (2018). https://doi.org/10.1007/s10886-018-0949-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-018-0949-x