Abstract
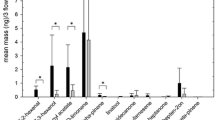
In laboratory experiments, we identified and quantified volatiles emitted by inflorescences and berries of two grape varieties (Trebbiano and Sangiovese) and examined the effects of the volatiles on oviposition by the grapevine moth Lobesia botrana. Compared to Trebbiano, Sangiovese is relatively more susceptible to L. botrana infestations under natural conditions. Chemical and electrophysiological analysis indicated only quantitative differences between the volatiles released by the two varieties. In a dual-choice oviposition bioassay based only on volatile cues, females did not show any preference between the two varieties. The six major components of the odor profiles that were GC-EAD-active to female antennae included: limonene, 4,8-dimethyl-1,(E)-3,7-nonatriene, (±)-linalool, (E)-caryophyllene, (E,E)-α-farnesene, and methyl salicylate. At the beginning of the berry touch phenological stage, their proportions were about 10:0.6:0.4:0.5:0.9:0.6 in Trebbiano and 10:1:0.4:1.5:0.4:0.3 in Sangiovese. A six-component synthetic lure (with the proportion 10:1:1:1:1:1, which approximated the ratio of components released by both varieties) was used in further laboratory oviposition bioassays. Depending on its dosage, the synthetic lure either attracted or repelled oviposition. L. botrana females laid significantly more eggs in the presence of either the grape bunches or the synthetic lure at the attractive dosage. In a release-capture experiment conducted in a field cage that covered two grapevine rows, the synthetic lure was more attractive than a grape cluster or a blank control, and it stimulated oviposition on the vegetation near the lure. The results indicate that L. botrana uses olfactory cues to select oviposition sites and that an artificial lure, containing the major volatiles released by two grape varieties, may be useful in monitoring female activity in the field.

Similar content being viewed by others
References
Anfora, G., Tasin, M., Bäckman, A. C., De Cristofaro, A., Witzgall, P., and Ioriatti, C. 2005. Attractiveness of year-old polyethylene Isonet sex pheromone dispensers for Lobesia botrana. Entomol. Exp. Appl. 117:201–207.
Anfora, G., Tasin, M., Bäckman, A. C., Leonardelli, E., De Cristofaro, A., Lucchi, A., and Ioriatti, C. 2008. Olfactory responses of Eupoecilia ambiguella (Hübner) (Lepidoptera Tortricidae) females to volatiles from grapevine. IOBC wprs Bull. 36:233–236.
Bagnoli, B. 1990. Incidenza delle infestazioni da artropodi e difesa dei vigneti in Toscana. La Difesa delle Piante 13:89–112.
Bengtsson, M., Bäckman, A. C., Liblikas, I., Ramirez, M. I., Borg-Karlson, A. K., Ansebo, L., Anderson, P., Löfqvist, J., and Witzgall, P. 2001. Plant odor analysis of apple: antennal response of codling moth females to apple volatiles during phenological development. J. Agric. Food Chem. 49:3736–3741.
Bernays, E. A., and Chapman, R. F. 1994. Host-plant selection by phytophagous insects. Chapman & Hall, London.
Bovey, P. 1966. Superfamille des Tortricoidae. L’Eudemis de la vigne, pp. 859–887, in A. S. Balachowsky (eds.). Entomologie appliquée à l’agriculture. Masson et Cie, Paris.
Bruce, J. A., Wadhams, L. J., and Woodcock, C. M. 2005. Insect host location: a volatile situation. Trends Plant. Sci. 10:269–274.
Cha, D. H., Hesler, S. P., Moser, C. L., Nojima, S., Linn, C. E., Roelofs, W. L., and Loeb, G. M. 2008a. Flight tunnel responses of female grape berry moth (Paralobesia viteana) to host plants. J. Chem. Ecol. 34:622–627.
Cha, D. H., Nojima, S., Hesler, S. P., Zhang, A., Linn, C. E., Roelofs, W. L., and Loeb, G. M. 2008b. Identification and field evaluation of grape shoot volatiles attractive to female grape berry moth (Paralobesia viteana). J. Chem. Ecol. 34:1180–1189.
Eichhorn, K. W., and Lorenz, D. H. 1977. Phänologishche Enwicklungsstadien der Rebe. Nachrichtenbl. Dtsch. Pflanzenschutzdienstes (Braunschweig) 29:119–120.
Faccoli, M., Anfora, G., and Tasin, M. 2008. Responses of the Mediterranean Pine Shoot Beetle Tomicus destruens (Wollaston) to pine shoot and bark volatiles. J. Chem. Ecol. 34:1162–1169.
Fermaud, M. 1998. Cultivar susceptibility of grape berry cluster to larvae of Lobesia botrana (Lepidoptera: Tortricidae). J. Econ. Entomol. 91:974–980.
Fermaud, M., and Le Menn, R. 1989. Association of Botrytis cinerea with grape berry moth larvae. Phytopathology 79:651–656.
Gabel, B. 1992. Tansy flowers attract European grapevine moth females, Lobesia botrana Den. & Schiff. (Lep., Tortricidae). J. Appl. Entomol. 113:153–158.
Gabel, B., Thiéry, D., Suchy, V., Marion-Poll, F., Hradsky, P., and Farkas, P. 1992. Floral volatiles of Tanacetum vulgare L. attractive to Lobesia botrana Den. et Schiff. females. J. Chem. Ecol. 18:693–701.
Grant, G. G., Guo, J., McDonald, L., and Coppens, M. D. 2007. Oviposition response of spruce budworm (Lepidoptera: Tortricidae) to host terpenes and green leaf volatiles. Can. Entomol. 139:564–575.
Heath, R. R., Teal, P. E. A., Tumlinson, J. H., and Mengelkoch, L. J. 1986. Prediction of release ratios of multicomponent pheromones from rubber septa. J. Chem. Ecol. 12:2133–2143.
Ioriatti, C., Lucchi, A., and Bagnoli, B. 2008. Grape areawide pest management in Italy, pp. 208–225, in O. Koul, G. W. Cuperus, and N. Elliott (eds.). Areawide Pest Management: Theory and Implementation. CABI.
Ioriatti, C., Anfora, G., Angeli, G., Mazzoni, V., and Trona, F. 2009. Effects of chlorantraniliprole on eggs and larvae of Lobesia botrana (Denis & Schiffermüller) (Lepidoptera: Tortricidae). Pest Manag. Sci. 65:717–722.
Kaissling, K. E. 1987. Wright Lectures on Insect Olfaction. Colbow K, Simon Fraser University, Burnaby.
Katerinopoulos, H. E., Pagona, G., Afratis, A., Stratigakis, N., and Roditakis, N. 2005. Composition and insect attracting activity of the essential oil of Rosmarinus officinalis. J. Chem. Ecol. 31:111–122.
Knudsen, G. K., Bengtsson, M., Kobro, S., Jaastad, G., Hofsvang, T., and Witzgall, P. 2008. Discrepancy in laboratory and field attraction of apple fruit moth Argyresthia conjugella to host plant volatiles. Physiol. Entomol. 33:1–6.
Maher, N., Thiery, D., and Städler, E. 2006. Oviposition by Lobesia botrana is stimulated by sugars detected by contact chemoreceptors. Physiol. Entomol. 31:14–21.
Masante-Roca, I., Anton, S., Delbac, L., Dufour, M. C., and Gadenne, C. 2007. Attraction of the grapevine moth to host and non-host plant parts in the wind tunnel: effects of plant phenology, sex, and mating status. Entomol. Exp. Appl. 122:239–245.
Moreau, J., Benrey, B., and Thiery, D. 2006. Assessing larval food quality for phytophagous insects: are the facts as simple as they appear? Funct. Ecol. 20:592–600.
Pavan, F., Stefanelli, G., Cargnus, E., and Villani, A. 2009. Assessing the influence of inflorescence traits on the susceptibility of grape to vine moths. J. Appl. Entomol. 133:394–401.
Schoonhoven, L. M., Jermy, T., and Van Loon, J. J. A. 1998. Insect-plant Biology, from Physiology to Evolution. Chapman & Hall, London
Sharon, R., Zahavi, T., Soroker, V., and Harari, A. R. 2009. The effect of grape vine cultivars on Lobesia botrana (Lepidoptera: Tortricidae) population levels. J. Pest Sci. 82:187–193.
Snjezana, H. 2004. Susceptibility of some grapevine cultivars in area of vineyards of Podgorica on the attack of European grape berry moth—Lobesia botrana Den. & Schiff. (Lepidoptera, Tortricidae). Acta Horticulturae 652:355–358.
Städler, E. 2002. Plant chemical cues important for egg deposition by herbivorous insects, pp 171–204, in M. Hilker, and T. Meiners (eds.). Chemoecology of Insect Eggs and Egg Deposition. Blackwell Publishing, Oxford, UK..
Suckling, D. M., Karg, G., Gibb, A. R., and Bradley, S. J. 1996. Electroantennogram and oviposition responses of Epiphyas postvittana (Lepidoptera: Totricidae) to plant volatiles. New Zeal. J. Crop Hort. 24:323–333.
Tasin, M., Anfora, G., Ioriatti, C., Carlin, S., De Cristofaro, A., Schmidt, S., Bengtsson, M., Versini, G., and Witzgall, P. 2005. Antennal and behavioral responses of grapevine moth Lobesia botrana females to volatiles from grapevine. J. Chem. Ecol. 31:77–87.
Tasin, M., Bäckman, A. C., Bengtsson, M., Ioriatti, C., and Witzgall, P. 2006a. Essential host plant cues in the grapevine moth. Naturwissenschaften 93:141–144.
Tasin, M., Bäckman, A. C., Bengtsson, M., Varela, N., Ioriatti, C., and Witzgall, P. 2006b. Wind tunnel attraction of grapevine moth females, Lobesia botrana, to natural and artificial grape odor. Chemoecology 16:87–92.
Tasin, M., Bäckman, A. C., Coracini, M., Casado, D., Ioriatti, C., and Witzgall, P. 2007. Synergism and redundancy in plant volatile blend attracting grapevine moth females. Phytochemistry 68:203–209.
Tasin, M., Anfora, G., Bäckman, AC., Ioriatti, C., De Cristofaro, A., Pozzolini, E., Leonardelli, E., and Lucchi, A. 2008. Volatiles from grape drive the oviposition of Lobesia botrana at short distance. IOBC wprs Bull. 36:351–353.
Tasin, M., Anfora, G., Leonardelli, E., Ioriatti, C., Lucchi, A., De Cristofaro, A., and Pertot, I. 2009. A bioassay-based approach for the evaluation of host-plant cues as oviposition stimuli in grapevine moth. IOBC wprs Bull. 41:83–86.
Thiéry, D., and Moreau, J. 2005. Relative performance of European grapevine moth (Lobesia botrana) on grapes and other hosts. Oecologia 143:548–557.
Thiéry, D., Retaud, P., and Dumas-Lattaque, L. 2006. Piégeage alimentaire de l’eudémis de la vigne: un outil intéressant et performant pour la description de la dynamique des vols zédépontes. Phytoma 592:27–30.
Visser, J. H. 1986. Host odor perception in phytophagous insects. Annu. Rev. Entomol. 31:121–144.
Wearing, C. H., and Hutchings, R. F. N. 1973. Alfa-farnesene, a naturally occurring oviposition stimulant for the codling moth Laspeyresia pomonella. J. Insect Physiol. 19:1251–1256.
Witzgall, P., Ansebo, L., Yang, Z., Angeli, G., Sauphanor, B., and Bengtsson, M. 2005. Plant volatiles affect oviposition by codling moths. Chemoecology 15:77–83.
Acknowledgements
We are grateful to Prof. Ingve Stenstrøm, Agricultural University of Norway, Department of Chemistry and Biotechnology (Aas, Norway) for the synthesis of (E)-DMNT. We thank Elisabetta Leonardelli, Elena Pozzolini, and Anna Eriksson for technical assistance. We thank the anonymous reviewers for helpful comments. This study was funded by the Government of Autonomous Province of Trento (HOST and INTERPLAY Research Projects) and by the Italian Ministry of University and Research (PRIN 2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Anfora, G., Tasin, M., De Cristofaro, A. et al. Synthetic Grape Volatiles Attract Mated Lobesia botrana Females in Laboratory and Field Bioassays. J Chem Ecol 35, 1054–1062 (2009). https://doi.org/10.1007/s10886-009-9686-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-009-9686-5