Abstract
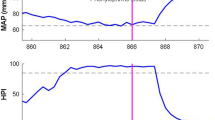
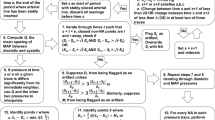
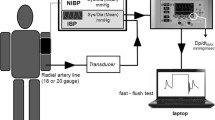
In critically ill and high-risk surgical room patients, an invasive arterial catheter is often inserted to continuously measure arterial pressure (AP). The arterial waveform pressure measurement, however, may be compromised by damping or inappropriate reference placement of the pressure transducer. Clinicians, decision support systems, or closed-loop applications that rely on such information would benefit from the ability to detect error from the waveform alone. In the present study we hypothesized that machine-learning trained algorithms could discriminate three types of transducer error from accurate monitoring with receiver operator characteristic (ROC) curve areas greater than 0.9. After obtaining written consent, patient arterial line waveform data was collected in the operating room in real-time during routine surgery requiring arterial pressure monitoring. Three deliberate error conditions were introduced during monitoring: Damping, Transducer High, and Transducer Low. The waveforms were split up into 10 s clips that were featurized. The data was also either calibrated against the patient’s own baseline or left uncalibrated. The data was then split into training and validation sets, and machine-learning algorithms were run in a Monte-Carlo fashion on the training data with variable sized training sets and hyperparameters. The algorithms with the highest balanced accuracy were pruned, then the highest performing algorithm in the training set for each error state (High, Low, Damped) for both calibrated and uncalibrated data was finally tested against the validation set and the ROC and precision-recall curve area-under the curve (AUC) calculated. 38 patients were enrolled in the study with a mean age of 52 ± 15 years. A total of 40 h of monitoring time was recorded with approximately 120,000 heart beats featurized. For all error states, ROC AUCs for algorithm performance on classification of the state were greater than 0.9; when using patient-specific calibrated data AUCs were 0.94, 0.95, and 0.99 for the transducer low, transducer high, and damped conditions respectively. Machine-learning trained algorithms were able to discriminate arterial line transducer error states from the waveform alone with a high degree of accuracy.




Similar content being viewed by others
Data availability
The datasets and code generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Slagt C, Malagon I, Groeneveld AB. Systematic review of uncalibrated arterial pressure waveform analysis to determine cardiac output and stroke volume variation. Br J Anaesth. 2014;112:626–37.
Cannesson M. Arterial pressure variation and goal-directed fluid therapy. J CardiothoracVascAnesth. 2010;24:487–97.
Cannesson M, Hofer I, Rinehart J, Lee C, Subramaniam K, Baldi P, Dubrawski A, Pinsky MR. Machine learning of physiological waveforms and electronic health record data to predict, diagnose and treat haemodynamic instability in surgical patients: protocol for a retrospective study. BMJ Open. 2019;9:e031988.
Hatib F, Jian Z, Buddi S, Lee C, Settels J, Sibert K, Rinehart J, Cannesson M. Machine-learning algorithm to predict hypotension based on high-fidelity arterial pressure waveform analysis. Anesthesiology. 2018;129:663–74.
Joosten A, Alexander B, Duranteau J, Taccone FS, Creteur J, Vincent JL, Cannesson M, Rinehart J. Feasibility of closed-loop titration of norepinephrine infusion in patients undergoing moderate- and high-risk surgery. Br J Anaesth. 2019;123:430–8.
Rinehart J, Lilot M, Lee C, Joosten A, Huynh T, Canales C, Imagawa D, Demirjian A, Cannesson M. Closed-loop assisted versus manual goal-directed fluid therapy during high-risk abdominal surgery: a case-control study with propensity matching. Crit Care. 2015;19:94.
Soltesz K, Sjoberg T, Jansson T, Johansson R, Robertsson A, Paskevicius A, Liao Q, Qin G, Steen S. Closed-loop regulation of arterial pressure after acute brain death. J ClinMonitComput. 2018;32:429–37.
NganKee WD, Tam YH, Khaw KS, Ng FF, Lee SW. Closed-loop feedback computer-controlled phenylephrine for maintenance of blood pressure during spinal anesthesia for cesarean delivery: a randomized trial comparing automated boluses versus infusion. AnesthAnalg. 2017. https://doi.org/10.1213/ANE.0000000000001974.
Marques NR, Whitehead WE, Kallu UR, Kinsky MP, Funston JS, Wassar T, Khan MN, Milosch M, Jupiter D, Grigoriadis K, Kramer GC. Physician-directed versus computerized closed-loop control of blood pressure using phenylephrine in a swine model. AnesthAnalg. 2017;125:110–6.
Cunningham S, Symon AG, McIntosh N. Changes in mean blood pressure caused by damping of the arterial pressure waveform. Early Hum Dev. 1994;36:27–30.
Kirchhoff KT, Rebenson-Piano M, Patel MK. Mean arterial pressure readings: variations with positions and transducer level. Nurs Res. 1984;33:343–5.
Smith J. Low-pass filtering by FFT convolution. In: Spectral audio signal processing. Stanford: W3K Publishing; 2011.
Pedregosa F, Varoquaux G, Gramfort A, Michel V, Thirion B, Grisel O, Blondel M, Prettenhofer P, Weiss R, Dubourg V, Vanderplas J, Passos A, Cournapeau D, Brucher M, Perrot M, Duchesnay E. Scikit-learn: machine learning in python. J Mach Learn Res. 2011;12:2825–30.
Saito T, Rehmsmeier M. The precision-recall plot is more informative than the ROC plot when evaluating binary classifiers on imbalanced datasets. PLoS ONE. 2015;10:e0118432.
Robin X, Turck N, Hainard A, Tiberti N, Lisacek F, Sanchez JC, Muller M. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011;12:77.
Funding
None.
Author information
Authors and Affiliations
Contributions
All authors read and approved the final manuscript. JR: designed the study, analyzed the data and drafted the manuscript. JT, JN, SS, HM: data collection, analysis, preparation of manuscript. PM, MDC: design of the study, review of final manuscript. MM: designed the study, assisted with data collection, reviewed the final manuscript. AJ: design of study, analysis, drafting of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
JR: Consultant for Edwards Lifesciences; Ownership in Sironis Inc & Perceptive Medical Inc. AJ: Consultant for Edwards Lifesciences. All other authors declare they have no competing interests.
Ethical approval
This study was approved by the Institutional Review Board of the University of California Irvine (protocol HS# 2019-5251).
Informed consent
All subjects in the present study provided written informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rinehart, J., Tang, J., Nam, J. et al. Detection of arterial pressure waveform error using machine learning trained algorithms. J Clin Monit Comput 36, 227–237 (2022). https://doi.org/10.1007/s10877-020-00642-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-020-00642-4