Abstract
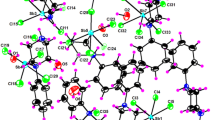
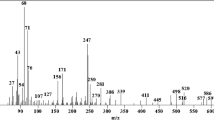
New mixed alkaline earth metal squarates, viz [Sr0.965Mg0.035(C4O4)(H2O)2]·H2O—(1) and [Sr0.88Ca0.12(C4O4)(H2O)3]—(2) have been synthesized and characterized by single-crystal X-ray diffraction, thermal analysis, and biological studies, EB-CTDNA binding, BSA binding, antioxidant and cytotoxicity activity. The complexes 1 and 2 crystallized in triclinic and monoclinic space groups with eight and nine coordination number, respectively, from the reaction mixture of squaric acid and the respective metal nitrates in aqueous medium at pH 7. The complexes on thermal analysis show that they yield mixed metal carbonates at 544 °C as residue. Their interaction with EB-CTDNA evaluated by emission method substantiates the intercalative mode of binding. The protein binding (BSA) study by the fluorescence quenching method reveals that the complexes bind strongly with BSA. Antioxidant property analysis shows that they exhibit a strong radical scavenging ability against ABTS, DPPH and NO radicals. The in vitro cytotoxicity of the complexes examined for human breast cancer (MCF-7) and lung cancer (A549) cell lines exhibit substantial cytotoxic property. AO/EB and DAPI staining methods support that they induce apoptosis and nuclear fragmentations in MCF-7 and A549 cell lines.









Similar content being viewed by others
References
C. Robl and A. Weiss (1986). Z Naturforsch B. 41, 1490–1494.
C. Robl and A. Weiss (1986). Z Naturforsch B. 41, 1485–1489.
C. Robl and A. Weiss (1987). Mater Res Bull. 22, 373–380.
A. Bouayad, J. C. Trombe, and A. Gleizes (1995). Inorganica Chim Acta. 230, 1–7.
Z. Hulvey and A. K. Cheetham (2007). Solid State Sci. 9, 137–143.
A. Bouhali, C. Trifa, S. Bouacida, C. Boudaren, and T. Bataille (2011). Acta Crystallogr Sect E Struct Rep Online. 67, m1130–m1131.
C. Trifa, A. Bouhali, S. Bouacida, C. Boudaren, and H. Merazig (2013). Acta Crystallogr Sect E Struct Rep Online 69, m366–m367.
C. Trifa, A. Bouhali, S. Bouacida, C. Boudaren, and T. Bataille (2011). Acta Crystallogr Sect E Struct Rep Online 67, m275–m276.
K. T. Priya Vadhana, S. Parveen, B. Ushadevi, R. Selvakumar, S. Sangeetha, and S. Vairam (2019). Acta Crystallogr Sect C Struct Chem. 275, 1091–1101.
Z. Liu, Y. Wang, Y. Han, L. Liu, J. Jin, H. Yi, Z. Li, J. Jiang, and D. W. Boykin (2013). Eur J Med Chem. 65, 187–194.
F. Olmo, C. Rotger, I. Ramirez-Macias, L. Martinez, C. Marin, L. Carreras, K. Urbanova, M. Vega, G. Chaves-Lemaur, A. Sampedro, M. J. Rosales, M. Sanchez-Moreno, and A. Costa (2014). J Med Chem. 57, 987–999.
P. Xu, M. Kelly, W. F. Vann, F. Qadri, E. T. Ryan, and P. Kovac (2017). ChemBioChem. 18, 799–815.
B. Palitzsch, S. Hartmann, N. Stergiou, M. Glaffig, E. Schmitt, and H. Kunz (2014). Angew Chemie Int Ed. 53, 14245–14249.
K. Morita, M. Nakamura, M. Nagamachi, T. Kishi, and Y. Miyachi (2002). J Dermatol. 29, 661–664.
P. I. A. Freyschmidt-Paul, R. Happle, S. Metz, R. Hoffmann, J. P. Sundberg, K. J. McElwee, and D. Boggess (1999). J Invest Dermatol. 113, 61–68.
C. A. Chen, V. Carlberg, and D. Kroshinsky (2017). Pediatr Dermatol. 34, e44–e46.
N. B. Silverberg, J. K. Lim, A. S. Paller, and A. J. Mancini (2001). J Am Acad Dermatol. 42, 803–808.
W. A. Kinney, N. E. Lee, D. T. Garrison, E. J. Podlesny, J. T. Simmonds, D. Bramlett, R. R. Notvest, D. M. Kowal, and R. P. Tasse (1992). J Med Chem. 35, 4720–4726.
J. A. Butera, M. M. Antane, S. A. Antane, T. M. Argentieri, C. Freeden, R. F. Graceffa, B. H. Hirth, D. Jenkins, J. R. Lennox, E. Matelan, N. Wesley Norton, D. Quagliato, J. H. Sheldon, W. Spinelli, D. Warga, A. Wojdan, and M. Woods (2000). J Med Chem. 43, 1187–1202.
A. M. Gilbert, M. M. Antane, T. M. Argentieri, J. A. Butera, G. D. Francisco, C. Freeden, E. G. Gundersen, R. F. Graceffa, D. Herbst, B. H. Hirth, J. R. Lennox, G. McFarlane, N. Wesley Norton, D. Quagliato, J. H. Sheldon, D. Warga, A. Wojdan, and M. Woods (2000). J Med Chem. 43, 1203–1214.
A. Mookerjee, M. Graciotti, and L. Kandalaft (2018). Bioimpacts. 8, 211–221.
A. Chaudhuri, Y. Venkatesh, J. Das, K. K. Behara, S. Mandal, T. K. Maiti, and N. D. P. Singh (2018). ACS Appl Nano Mater. 1, 6312–6319.
M. A. Sayeed, M. K. Bufano, P. Xu, G. Eckhoff, R. C. Charles, M. M. Alam, T. Sultana, M. R. Rashu, A. Berger, G. Gonzalez-Escobedo, A. Mandlik, T. R. Bhuiyan, D. T. Leung, R. C. LaRocque, J. B. Harris, S. B. Calderwood, F. Qadri, W. F. Vann, P. Kovac, and E. T. Ryan (2015). PLoS Negl Trop Dis. 9, 1–18.
Y. Li, C. Wen, D. Mushahary, R. Sravanthi, N. Harishankar, G. Pande, and P. Hodgson (2012). Acta Biomater. 8, 3177–3188.
H. Li, H. Yang, Y. Zheng, F. Zhou, K. Qiu, and X. Wang (2015). Mater Des. 83, 95–102.
J. Seo, E. S. Hong, H. J. Yoon, and S. K. Shin (2012). Int J Mass Spectrom. 330–332, 262–270.
J. Duguid, V. A. Bloomfield, J. Benevides, and G. J. Thomas (1993). Biophys J. 65, 1916–1928.
J. Sponer, J. V. Burda, M. Sabat, J. Leszczynski, and P. Hobza (1998). J Phys Chem A. 102, 5951–5957.
T. Marino, D. Mazzuca, N. Russo, M. Toscano, and A. Grand (2010). Int J Quantum Chem. 110, 138–147.
C. Wu, Z. Chen, D. Yi, J. Chang, and Y. Xiao (2014). ACS Appl Mater Interfaces. 6, 4264–4276.
W. Wang, J. Han, X. Yang, M. Li, P. Wan, L. Tan, Y. Zhang, and K. Yang (2016). Mater Sci Eng B. 214, 26–36.
R. Re, N. Pellegrini, A. Proteggente, A. Pannala, M. Yang, and C. Rice-Evans (1999). Free Radic Biol Med. 26, 1231–1237.
S. C. Liu, J. T. Lin, C. K. Wang, H. Y. Chen, and D. J. Yang (2009). Food Chem. 114, 577–581.
B. M. Olabinr, O. O. Odedire, M. T. Olaleye, A. S. Adekunl, L. O. Ehigie, and P. F. Olabinr (2010). Res J Biol Sci. 5, 102–105.
S. Vairam and S. Govindarajan (2006). Polish J Chem. 80, 1601–1614.
J. R. Ferraro, R. Driver, W. R. Walker, and W. Wozniak (1967). Inorg Chem. 6, 1586–1588.
J. C. Trombe, J. F. Petit, and A. Gleizes (1990). Inorganica Chim Acta. 167, 69–81.
C. C. Correa, R. Diniz, L. H. Chagas, B. L. Rodrigues, M. I. Yoshida, W. M. Teles, F. C. Machado, H. G. M. Edwards, and L. F. C. De Oliveira (2007). Vib Spectrosc. 45, 82–88.
C. C. Correa, R. Diniz, L. H. Chagas, B. L. Rodrigues, M. I. Yoshida, W. M. Teles, F. C. Machado, and L. F. C. De Oliveira (2007). Polyhedron. 26, 989–995.
E. Knaepen, J. Mullens, J. Yperman, and L. C. Van Poucke (1996). Thermochim Acta. 284, 213–227.
D. L. Boger, B. E. Fink, S. R. Brunette, W. C. Tse, and M. P. Hedrick (2001). J Am Chem Soc. 123, 5878–6589.
D. S. Raja, N. S. P. Bhuvanesh, and K. Natarajan (2012). Dalt Trans. 41, 4365–4377.
R. Prabhakaran, P. Kalaivani, R. Huang, P. Poornima, V. Vijaya Padma, F. Dallemer, and K. Natarajan (2013). J Biol Inorg Chem. 18, 233–247.
R. Manikandan, N. Chitrapriya, Y. J. Jang, and P. Viswanathamurthi (2013). RSC Adv. 3, 11647–11657.
R. Manikandan, P. Viswanathamurthi, K. Velmurugan, R. Nandhakumar, T. Hashimoto, and A. Endo (2014). J Photochem Photobiol B Biol. 130, 205–216.
O. Novakova, H. Chen, O. Vrana, A. Rodger, P. J. Sadler, and V. Brabec (2003). Biochemistry. 42, 11544–11554.
L. S. Lerman (1961). J Mol Biol. 3, 18–30.
B. C. Jonathan (1997). Biopolymers. 44, 201–215.
R. D. Snyder (2007). Mutat Res. 623, 72–82.
K. Pasternak, J. Kocot, and A. Horecka (2010). J Element. 15, 601–616.
A. Kosiha, C. Parthiban, and K. Elango (2017). J Photochem Photobiol B. 168, 165–174.
G. Kalaiarasi, S. Dharani, H. Puschmann, and R. Prabhakaran (2018). Inorg Chem Commun. 97, 34–38.
J. N. Miller (1979). Proc Anal Div Chem Soc. 16, 203–208.
J. Tang, F. Luan, and X. Chen (2006). Bioorg Med Chem. 14, 3210–3217.
D. Senthil Raja, G. Paramaguru, N. S. P. Bhuvanesh, J. H. Reibenspies, R. Renganathan, and K. Natarajan (2011). Dalt Trans. 40, 4548–4559.
J. S. Wright, E. R. Johnson, and G. A. Dilabio (2001). J Am Chem Soc. 123, 1173–1183.
K. Tsai, T. G. Hsu, K. M. Hsu, H. Cheng, T. Y. Liu, C. F. Hsu, and C. W. Kong (2001). Free Radic Biol Med. 31, 1465–1472.
R. Loganathan, S. Ramakrishnan, E. Suresh, M. Palaniandavar, A. Riyasdeen, and M. A. Akbarsha (2014). Dalt Trans. 43, 6177–6194.
M. Ganeshpandian, R. Loganathan, E. Suresh, A. Riyasdeen, M. A. Akbarsha, and M. Palaniandavar (2014). Dalt Trans. 43, 1203–1219.
S. Thakore, M. Valodkar, J. Y. Soni, K. Vyas, R. N. Jadeja, R. V. Devkar, and P. S. Rathore (2013). Bioorg Chem. 46, 26–30.
P. Naveen, F. Dallemer, R. J. Butcher, and R. Prabhakaran (2018). Inorganica Chim Acta. 471, 724–734.
C. C. Zeng, G. B. Jiang, S. H. Lai, C. Zhang, H. Yin, B. Tang, D. Wan, and Y. J. Liu (2016). J Photochem Photobiol B Biol. 161, 295–303.
T. Thirunavukkarasu, H. A. Sparkes, K. Natarajan, and V. G. Gnanasoundari (2018). Inorganica Chim Acta. 473, 255–262.
N. Nanjundan, R. Narayanasamy, R. J. Butcher, J. P. Jasinski, K. Velmurugan, R. Nandhakumar, M. D. Balakumaran, P. T. Kalaichelvan, and V. G. Gnanasoundari (2017). Inorganica Chim Acta. 455, 283–297.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authours declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Priya Vadhana, K.T., Vairam, S., Ushadevi, B. et al. New Mg(II) and Ca(II) Mixed Strontium Squarates: Structural Characterization, DNA/BSA Interaction, Antioxidant and Anticancer Activities. J Clust Sci 33, 867–885 (2022). https://doi.org/10.1007/s10876-021-01989-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-021-01989-7