Abstract
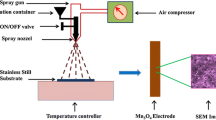
Nanostructures of manganese oxides are a promising pseudocapacitive electrode material due to its eco-friendly, low cost, and intrinsically high capacity. In this work, we prepared monometallic manganese oxide Mn3O4, and MnO2 using a facile, one-step hydrothermal method at low processing temperature. The structural, morphological, elemental analysis of manganese oxide (Mn3O4 nanoparticles (NPs) and one-dimensional MnO2 nanowires (1-D NWs)) powders confirmed from XRD, TEM, SEM, and EDAX techniques. The prepared materials have the same crystal structure with different phase and morphology. The morphology information of prepared Mn3O4, MnO2 powders visualized from an electron microscope of TEM and SEM techniques as particle and wire type morphology. The electrochemical performance of fabricated individual Mn3O4 and MnO2 @ Ni foam electrodes exhibited specific capacitance around 182 and 243 F/g at a current density of 0.5 A/g from galvanostatic charge–discharge cycles measurement in presence of 1 M KOH compared with 0.5 M KOH electrolyte solution. Also, from these results, the morphology of MnO2 (NWs) has better specific capacitance than Mn3O4 (NPs) in 1 M KOH electrolyte solution.







Similar content being viewed by others
References
M. Huang, F. Li, F. Dong, Y. X. Zhang, and L. L. Zhang (2015). J. Mater. Chem. A 3, 21380.
X. Shi, S. Zhang, X. Chen, P. K. Chu, T. Tang, and E. Mijowska (2019). Int. J. Hydrogen Energy 44, 13675.
M. A. A. Mohd Abdah, N. H. N. Azman, S. Kulandaivalu, and Y. Sulaiman (2020). Mater. Des. 186, 108199.
A. R. Dehghani-Sanij, E. Tharumalingam, M. B. Dusseault, and R. Fraser (2019). Renew. Sustain. Energy Rev. 104, 192.
A. Mayyas, D. Steward, and M. Mann (2019). Sustain. Mater. Technol. 19, e00087.
Z. Li, L. Cao, W. Chen, Z. Huang, and H. Liu (2019). Small 15, 1.
Q. Abbas, R. Raza, I. Shabbir, and A. G. Olabi (2019). J. Sci. Adv. Mater. Devices 4, 341.
A. Afif, S. M. Rahman, A. Tasfiah Azad, J. Zaini, M. A. Islan, and A. K. Azad (2019). J. Energy Storage 25, 100852.
R. Thangavel, A. G. Kannan, R. Ponraj, V. Thangavel, D. W. Kim, and Y. S. Lee (2018). J. Power Sources 383, 102.
P. Nagaraju, A. Alsalme, A. Alswieleh, and R. Jayavel (2018). J. Electroanal. Chem. 808, 90.
C. Xiang, M. Li, M. Zhi, A. Manivannan, and N. Wu (2012). J. Mater. Chem. 22, 19161.
H. Xiao, W. Guo, B. Sun, M. Pei, and G. Zhou (2016). Electrochim. Acta 190, 104.
V. Sharavath, S. Sarkar, and S. Ghosh (2018). J. Electroanal. Chem. 829, 208.
Z. Bi, Q. Kong, Y. Cao, G. Sun, F. Su, X. Wei, X. Li, A. Ahmad, L. Xie, and C. M. Chen (2019). J. Mater. Chem. A 7, 16028.
C. F. Liu, Y. C. Liu, T. Y. Yi, and C. C. Hu (2019). Carbon 145, 529.
S. Bashir, P. Hanumandla, H. Y. Huang, and J. L. Liu (2018). Nanostructured Mater. Next-Gen Energy Storage Convers. Fuel Cells 4, 517.
B. E. Conway, V. Birss, and J. Wojtowicz (1997). J. Power. Sources 66, 1.
R. B. Rakhi, W. Chen, and H. N. Alshareef (2012). J. Mater. Chem. 22, 5177.
Q. Yang, Z. Li, R. Zhang, L. Zhou, M. Shao, and M. Wei (2017). Nano. Energy. 41, 408.
Y. Han and L. Dai (2019). Macromol. Chem. Phys. 220, 1.
G. A. Snook, P. Kao, and A. S. Best (2011). J. Power. Sources. 196, 1.
A. Rudge, J. Davey, I. Raistrick, and S. Gottesfeld (1994). J. Power Sources 47, 89.
X. Lang, A. Hirata, T. Fujita, and M. Chen (2011). Nat. Nanotechnol. 6, 232.
S. N. Pusawale, P. R. Deshmukh, P. S. Jadhav, and C. D. Lokhande (2019). Mater. Renew. SustainEnergy 8, 1.
S. Mothkuri, S. Chakrabarti, H. Gupta, B. Padya, T. N. Rao, and P. K. Jain (2019). Mater. Today. Proc.. https://doi.org/10.1016/j.matpr.2019.03.236.
S. Qiu, R. Li, Z. Huang, Z. Huang, C. P. Tsui, C. He, X. Han, and Y. Yang (2019). Compos. Part B Eng. 161, 37.
N. Palaniyandy, F. P. Nkosi, K. Raju, and K. I. Ozoemena (2019). J. Electroanal. Chem. 833, 79.
P. Wu, S. Dai, G. Chen, S. Zhao, Z. Xu, M. Fu, P. Chen, Q. Chen, X. Jin, Y. Qiu, S. Yang, and D. Ye (2020). Appl. Catal. B Environ. 268, 118418.
X. Jiang, P. Gray, M. Patel, J. Zheng, and J. J. Yin (2020). J. Mater. Chem. B 8, 1191.
F. W. Boyom-Tatchemo, F. Devred, G. Ndiffo-Yemeli, S. Laminsi, and E. M. Gaigneaux (2020). Appl. Catal. B Environ. 260, 118159.
M. S. Osgouei, M. Khatamian, and H. Kakili (2020). Mater. Chem. Phys. 239, 122108.
K. Byrappa and T. Adschiri (2007). Prog. Cryst. Growth Charact. Mater. 53, 117.
Y. Zhao, M. Hao, Y. Wang, Y. Sha, and L. Su (2016). J. Solid. State. Electrochem. 20, 81.
H. Liu, H. Zhang, H. Xu, T. Lou, Z. Sui, and Y. Zhang (2018). J. Electrochem. Soc. 165, A97.
H. U. Shah, F. Wang, M. S. Javed, N. Shaheen, M. Saleem, and Y. Li (2018). Ceram. Int. 44, 3580.
H. Ullah Shah, F. Wang, M. Sufyan Javed, N. Shaheen, S. Ali, M. Ashfaq Ahmad, and K. He (2018). Mater. Lett. 210, 148.
J. Yao, S. Yao, F. Gao, L. Duan, M. Niu, and J. Liu (2018). J. Colloid. Interface. Sci. 511, 434.
L. Wang, G. Duan, S. M. Chen, and X. Liu (2018). J. Alloys. Compd. 752, 123.
Y. Xiao, Y. Cao, Y. Gong, A. Zhang, J. Zhao, S. Fang, D. Jia, and F. Li (2014). J. Power. Sources. 246, 926.
A. K. Singh, D. Sarkar, G. G. Khan, and K. Mandal (2013). J. Mater. Chem. A. 1, 12759.
L. Halder, A. Maitra, A. K. Das, R. Bera, S. K. Karan, S. Paria, A. Bera, S. K. Si, and B. B. Khatua (2019). ACS Appl. Electron. Mater. 1, 189.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Godlaveeti, S.K., Jangiti, S., Somala, A.R. et al. Different Phase and Morphology Effect of Manganese Oxide on Electrochemical Performance for Supercapacitor Application. J Clust Sci 32, 703–710 (2021). https://doi.org/10.1007/s10876-020-01833-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-020-01833-4