Abstract
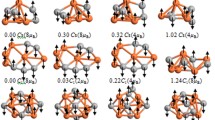
We report a quantum-chemistry investigation of the cationic, neutral, and anionic \(({\text {CeO}}_2)_{6}\) clusters to obtain an atom-level understanding of the effects induced by the release or addition of a single electron on the physical and chemical properties of small oxide clusters. Our ab initio calculations are based on density functional theory (DFT) within the hybrid Heyd–Scuseria–Ernzerhof (HSE06) and semilocal Perdew–Burke–Ernzerhof (PBE) functional. Compared with PBE, the HSE06 functional changes the relative stability of the neutral \(({\text {CeO}}_2)_{6}\) isomers, in particular, for structures with small total energy differences, e.g., about \(100 \, \text {meV/fu}\), which can be explained by the enhancement of the exchange interactions. The addition of an electron to the \(({\text {CeO}}_2)_{6}\) clusters change the oxidation state of a single \(\text {Ce}\) atom from + IV to + III, which drives a local distortion and the formation of a small polaron near to the \({\text {Ce}}^{\text{III}}\) cation. In contrast, the release of an electron induces the formation of a localized hole on one of the \(\text {O}\) atoms combined with local structural distortions. For the anionic and cationic clusters in the putative global minimum configurations, we found a strain energy induced by the distortion of 1.00 and 1.31 eV, respectively.





Similar content being viewed by others
Abbreviations
- DFT:
-
density functional theory
- PBE:
-
Perdew–Burke–Ernzerhof
- HSE06:
-
Heyd–Scuseria–Ernzerhof
- FHI-aims:
-
Fritz–Haber Institute ab initio molecular simulations
References
Q. Fu, H. Saltsburg and M. Flytzani-Stephanopoulos (2003). Active nonmetallic Au and Pt species on ceria-based water-gas shift catalysts. Science301, 935–938.
R. J. Gorte (2010). Ceria in catalysis: from automotive applications to the water-gas shift reaction. AlChE J. 56, 1126–1135.
A. F. Diwell, R. R. Rajaram, H. A. Shaw and T. J. Truex (1991). The role of ceria in three-way catalysts. Stud. Surf. Sci. Catal. 71, 139–152.
A. Trovarelli Catalysis by ceria and related materials, 1st ed (World Scientific Publishing Company, Singapore, 2002).
A. Beste and S. H. Overbury (2015). Pathways for ethanol dehydrogenation and dehydration catalyzed by ceria (111) and (100) surfaces. J. Phys. Chem. C2015, 2447–2455.
Z.-X. Zhou, L.-N. Wang, Z.-Y. Li, S.-G. He and T.-M. Ma (2016). Oxidation of \({\text{ SO }}_{2}\) to \({\text{ SO }}_{3}\) by cerium oxide cluster cations \({\text{ Ce }}_{2} {\text{ O }}_{4}{}^{+}\) and \({\text{ Ce }}_{3} {\text{ O }}_{6}{}^{+}\). J. Phys. Chem. C120, 3843–3848.
X.-L. Ding, X.-N. Wu, Y.-X. Zhao, J.-B. Ma and S.-G. He (2011). Double-oxygen-atom transfer in reactions of \({\text{ Ce }}_{m} {\text{ O }}_{2m} {}^{+}\) (m= 2–6) with \({\text{ C }}_{2} {\text{ H }}_{2}\). ChemPhysChem12, 2110–2117.
S. Hirabayashi and M. Ichihashi (2013). Oxidation of CO and NO on composition-selected cerium oxide cluster cations. J. Phys. Chem. A117, 9005–9010.
J. A. Felton, M. Ray, S. E. Waller, J. O. Kafader and C. C. Jarrold (2014). \({\text{ Ce }}_{x} {\text{ O }}_{y} - (\text{ x }= 2{-}3) + {\text{ D }}_2\text{ O }\) reactions: stoichiometric cluster formation from deuteroxide decomposition and anti-arrhenius behavior. J. Phys. Chem. A118, 9960–9969.
I. E. Wachs and K. Routray (2012). Catalysis science of bulk mixed oxides. ACS Catal. 2, 1235–1246.
M. V. Ganduglia-Pirovano, J. L. F. Da Silva and J. Sauer (2009). Density functional calculations of the structure of near-surface oxygen vacancies and electron localization \(\text{ CeO }_2\)(111). Phys. Rev. Lett. 102, 026101.
L. Sun, X. Huang, L. Wang and A. Janotti (2017). Disentangling the role of small polarons and oxygen vacancies in \({\text{ CeO }}_{2}\). Phys. Rev. B95, 245101.
J. L. F. Da Silva, M. V. Ganduglia-Pirovano, J. Sauer, V. Bayer and G. Kresse (2007). Hybrid functionals applied to rare-earth oxides: the example of ceria. Phys. Rev. B75, 045121.
M. J. Piotrowski, P. Tereshchuk and J. L. F. Da Silva (2014). Theoretical investigation of small transition-metal clusters supported on the \({\text{ CeO }}_{2}\)(111) surface. J. Phys. Chem. C188, 21438–21446.
P. Tereshchuk, R. L. H. Freire, C. G. Ungureanu, Y. Seminovski, A. Kiejna and J. L. F. Da Silva (2015). The role of charge transfer in the oxidation state change of Ce atoms in the \({\text{ TM }}_{13} - {\text{ CeO }}_{2}\)(111) systems (\(\text{ TM } = \text{ Pd }, \text{ Ag }, \text{ Pt }, \text{ Au }\)): A DFT+U investigation. Phys. Chem. Chem. Phys. 17, 13520–13530.
T. Nagata, J. W. J. Wu, M. Nakano, K. Ohshimo and F. Misaizu (2019). Geometrical structures of gas-phase cerium oxide cluster cations studied by ion mobility mass spectrometry. J. Phys. Chem. C123, 16641–16650.
S. T. Akin, S. G. Ard, B. E. Dye, H. F. Schaefer and M. A. Duncan (2016). Photodissociation of cerium oxide nanocluster cations. J. Phys. Chem. A120, 2313–2319.
A. M. Burow, T. Wende, M. Sierka, R. Włodarczyk, J. Sauer, P. Claes, L. Jiang, G. Meijer, P. Lievens and K. R. Asmis (2011). Structures and vibrational spectroscopy of partially reduced gas-phase cerium oxide clusters. Phys. Chem. Chem. Phys. 13, 19393–19400.
L. Zibordi-Besse, Y. Seminovski, I. Rosalino, D. Guedes-Sobrinho and J. L. F. D. Silva (2018). Physical and chemical properties of unsupported \(({\text{ MO }}_2)_{n}\) clusters for M = Ti, Zr, or \(\text{ Ce }\) and n = 1–15: a density functional theory study combined with the tree-growth scheme and euclidean similarity distance algorithm. J. Phys. Chem. C122, 27702–27712.
C. Chuan, C. Hui-Lung, W. Meng-Hsiung, J. Shin-Pon, J.-G. Chang and C.-S. Chang (2008). Structural properties of \(({\text{ CeO }}_2)_{n}\) (n= 1–5) nanoparticle: molecular mechanics and first principle studies. Chin. J. Catal. 29, 1117–1121.
S. F. Li, H. Lu, P. Li, Z. Yang and Z. X. Guo (2008). First-principles local density approximation (generalized gradient approximation) + U study of catalytic \({\text{ Ce }}_{n} {\text{ O }}_{m}\) clusters: U value differs from bulk. J. Chem. Phys. 128, 164718.
J. Heyd, G. E. Scuseria and M. Ernzerhof (2003). Hybrid functionals based on a screened coulomb potential. J. Chem. Phys. 118, 8207–8215.
J. P. Perdew, K. Burke and M. Ernzerhof (1996). Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868.
V. Blum, R. Gehrke, F. Hanke, P. Havu, V. Havu, X. Ren, K. Reuter and M. Scheffler (2009). Ab initio molecular simulations with numeric atom-centered orbitals. Comput. Phys. Commun. 180, 2175–2196.
V. Havu, V. Blum, P. Havu and M. Scheffler (2009). Efficient \(O(N)\) integration for all-electron electronic structure calculation using numeric basis functions. J. Comput. Phys. 228, 8367–8379.
M. Chen and D. A. Dixon (2013). Tree growth—hybrid genetic algorithm for predicting the structure of small \(({\text{ TiO }}_2)_{n}\), \(n = 2-13\). Nanoclusters. J. Chem. Theory Comput. 9, 3189–3200.
R. Gehrke and K. Reuter (2009). Assessing the efficiency of first-principles basin-hopping sampling. Phys. Rev. B79, 085412.
H. M. Cezar, G. G. Rondina and J. L. F. Da Silva (2017). Parallel tempering monte carlo combined with clustering euclidean metric analysis to study the thermodynamic stability of Lennard–Jones nanoclusters. J. Chem. Phys. 146, 064114.
J. Nocedal, J. Stephen and S. J. Wright Numerical optimization (Springer, Berlin, 2006).
J. L. F. Da Silva (2011). Effective coordination concept applied for phase change \((\text{ GeTe })_{m}~({\text{ Sb }}_2 {\text{ Te }}_3)_{n}\) compounds. J. Appl. Phys. 109, 023502.
Acknowledgements
The authors gratefully acknowledge support from FAPESP (São Paulo Research Foundation, Grant Number 2017/11631-2), Shell and the strategic importance of the support given by ANP (Brazils National Oil, Natural Gas and Biofuels Agency) through the R&D levy regulation. This study was financed in part by the National Counsel of Technological and Scientific Development (fellowships for Mailde S. Ozório and Augusto C. H. Da Silva). The authors acknowledge also the National Laboratory for Scientific Computing (LNCC/MCTI, Brazil) for providing HPC resources of the SDumont supercomputer, which have contributed to the research results reported within this paper. http://sdumont.lncc.br. We acknowledges also the Advanced Scientific Computational Laboratory (University of São Paulo) and the infrastructure provided to our computer cluster by the São Carlos Center of Informatics, University of São Paulo.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ozório, M.S., Da Silva, A.C.H. & Da Silva, J.L.F. A Hybrid Density Functional Theory Investigation of the \(({\text {CeO}}_2)_{6}\) Clusters in the Cationic, Neutral, and Anionic States. J Clust Sci 31, 1213–1220 (2020). https://doi.org/10.1007/s10876-019-01728-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-019-01728-z