Abstract
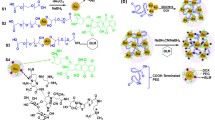
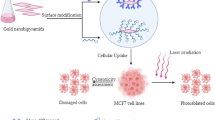
Gold nanoparticles (AuNPs) are potent anticancer agent that controls drug delivery to tumors. Here, we describe the identification of TAT-Cell Penetrating Peptide (TAT-CPP) conjugated AuNPs, as a novel delivery system to the cancerous regions. TAT-peptide was modified to BSA-AuNPs [Bovine Serum Albumin (BSA) coated AuNPs] electrostatically. The binding efficiency of TAT-AuNPs was tested using Dynamic Light Scattering, UV–Vis spectrophotometer and Zeta potential. The nano-complex (BSA-AuNPs; with and without TAT-CPP) was applied against Rhabdomyosarcoma and Murine fibroblast (L20B) cancer lines, in vitro. Cytotoxicity effect was evaluated by MTT assay at 0.125, 0.25, 0.5 and 1 mg/ml concentration for 24 and 48 h incubation time. Results demonstrated that TAT-(BSA-AuNPs) exhibits significant toxicity for both cancer cell lines. TAT-CPP has improved cancer cell reduction, where cytotoxicity more than 80% has been achieved. This study was conducted to achieve the simplicity and facility in cancer therapy, where the small-sized TAT-AuNPs acts as a simple therapeutic agent in the specific delivery and targeting the deep, irregular, and complicated cancer regions in the human body. Therefore, it could replace other cancer treatment techniques, even dispense the laser irradiation in the phototherml therapy.







Similar content being viewed by others
References
E. Blanco, A. Hsiao, A. P. Mann, M. G. Landry, F. Meric-Bernstam, and M. Ferrari (2011). Cancer Sci. 102, (7), 1247–1252.
S. M. H. AL-Jawad, A. A. Taha, M. M. F. Al-Halbosiy, and L. F. A. AL-Barram (2018). Photodiagnosis Photodyn. Ther. 21, 201–210.
V. V. Tuchin (2015). J. Biomed. Photonics Eng. 1, 2.
T. Gould, Q. Wang, and T. J. Pfefer (2014). Biomed. Opt. Exp. 5, 832–847.
G. Han and J. Xi (2016). Light-Med. Deep-Tissue Theranostics. 6, 2292–2294.
S. Jafari, S. Maleki Dizaj, and K. Adibkia (2015). Bioimpacts 5, (2), 103–111.
A. A. Sousa, J. T. Morgan, P. H. Brown, A. Adams, M. P. Jayasekara, G. Zhang, C. J. Ackerson, M. J. Kruhlak, and R. D. Leapman (2012). Small 8, (14), 2277–2286.
J. R. Nicol, D. Dixon, and J. A. Coulter (2015). Nanomedicine 10, (8), 1315–1326.
R. Sawant and V. Torchilin (2010). Mol. BioSyst. 6, 628–640.
S. Kalmodia, S. Vandhana, B. R. Tejaswini Rama, B. Jayashree, T. Sreenivasan Seethalakshmi, V. Umashankar, W. Yang, C. J. Barrow, S. Krishnakumar, and S. V. Elchuri (2016). Cancer Nanotechnol. 7, (1), 2–19.
C. Ciobanasu, J. P. Siebrasse, and U. Kubitscheck (2010). Biophys. J. 99, (1), 153–162.
L. Singh, R. Parboosing, H. G. Kruger, G. E. M. Maguire, and T. Govender (2016). Nanotechnology 7, 1–7.
T. Mironava, M. Hadjiargyrou, M. Simon, V. Jurukovski, and M. H. Rafailovich (2010). Nanotoxicology 4, (1), 120–137.
L. D. F. Vasconcelos Complexes of Cell-Penetrating Peptides with Oligonucleotides Structure, Binding and Translocation in Lipid Membranes (Stockholm University, Stockholm, 2017), pp. 1–79.
J. H. Grossman and S. E. McNeil (2012). Phys. Today. 65, (8), 38.
M. Umair, I. Javed, M. Rehman, A. Madni, A. Javeed, A. Ghafoor, and M. Ashraf (2016). J. Pharm. Pharm. Sci. 19, (2), 161–180.
S. Huo, S. Jin, X. Ma, X. Xue, K. Yang, A. Kumar, P. C. Wang, J. Zhang, Z. Hu, and X. J. Liang (2014). ACS Nano. 8, (6), 5852–5862.
I. Capek (2015). J. Nanomed. Res. 2, (1), 1–10.
E. Oh, J. B. Delehanty, K. E. Sapsford, K. Susumu, R. Goswami, J. B. Blanco-Canosa, P. E. Dawson, J. Granek, M. Shoff, Q. Zhang, P. L. Goering, A. Huston, and I. L. Medintz (2011). ACS Nano. 5, (8), 6434–6448.
Y. Jiang, S. Huo, T. Mizuhara, R. Das, Y. W. Lee, S. Hou, D. F. Moyano, B. Duncan, X. J. Liang, and V. M. Rotello (2015). ACS Nano. 9, (10), 9986–9993.
P. Wang, X. Wang, L. Wang, X. Hou, W. Liu, and C. Chen (2015). Sci. Technol. Adv. Mater. 16, (3), 1–15.
Hyejin Park, Hiroshi Tsutsumi, and Hisakazu Mihara (2013). Biomaterials 34, 4872–4879.
G. Guidotti, L. Brambilla, and D. Rossi (2017). Trends Pharmacol Sci. 38, (4), 406–424.
F. Madani, S. Lindberg, U. Langel, S. Futaki, and A. Gräslund (2011). J Biophys. 2011, 414729.
S. Trabulo, A. L. Cardoso, M. Mano, and M. C. P. Lima (2010). Pharmaceuticals 3, (4), 961–993.
J. Song, Y. Zhang, W. Zhang, J. Chen, X. Yang, P. Ma, B. Zhang, B. Liu, J. Ni, and R. Wang (2015). Peptides. 63, 143–149.
J. Thundimadathil (2012). J. Amino. Acids. 2012, 967347.
Z. Krpetić, S. Saleemi, I. A. Prior, V. See, R. Qureshi, and M. Brust (2011). ACS Nano. 5, (6), 5195–5201.
A. Mishra, G. H. Lai, N. W. Schmidt, V. Z. Sun, A. R. Rodriguez, R. Tong, L. Tang, J. Cheng, T. J. Deming, D. T. Kamei, and G. C. Wong (2011). Proc. Natl. Acad. Sci. USA. 108, (41), 16883–16888.
D. Zhang, J. Wang, and D. Xu (2016). J. Control Release. 229, 130–139.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Taha, A.A., AL-Jawad, S.M.H. & AL-Barram, L.F.A. Improvement of Cancer Therapy by TAT Peptide Conjugated Gold Nanoparticles. J Clust Sci 30, 403–414 (2019). https://doi.org/10.1007/s10876-019-01497-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-019-01497-9