Abstract
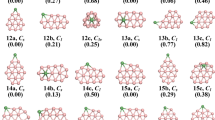
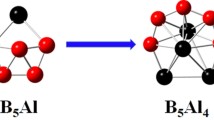
Doping transition metal atom is known as an effective approach to stabilize an atomic cluster and modify its structure and electronic properties. We herein report the effect of molybdenum doping on the structural evolution of medium-sized boron clusters. The lowest-energy structures of MoBn (n = 10, 12, 14, 16, 18, 20, 22, 24) clusters are globally searched using genetic algorithm combined with density functional theory calculations. We found that Mo doping has significantly affected the grow behaviors of Bn clusters, leading to a structural evolution from bowl-like to tubular and finally endohedral cage. The size-dependent binding energy, HOMO–LUMO gap, vertical ionization potential and vertical electron affinity show that MoB12, MoB22 and MoB24 clusters have relatively higher stability and enhanced chemical inertness. More interestingly, the endohedral MoB22 cage is identified as an elegant superatom, which satisfies 18-electron closed shell configuration well.



Similar content being viewed by others
References
J. Zhao, L. Ma, D. Tian, and R. Xie (2008). J. Comput. Theor. Nano. 5, 7.
H. J. Zhai, B. Kiran, J. Li, and L. S. Wang (2003). Nat. Mater. 2, 827.
J. Zhao, X. Huang, P. Jin, and Z. Chen (2015). Coord. Chem. Rev. 289–290, 315.
X. Huang, H. G. Xu, S. Lu, Y. Su, R. B. King, J. Zhao, and W. Zheng (2014). Nanoscale 6, 14617.
H. Hiura, T. Miyazaki, and T. Kanayama (2001). Phys. Rev. Lett. 86, 1733.
V. Kumar and Y. Kawazoe (2003). Phys. Rev. Lett. 91, 199901.
S. N. Khanna, B. K. Rao, and P. Jena (2002). Phys. Rev. Lett. 89, 016803.
P. Pyykkö (2006). J. Organomet. Chem. 691, 4336.
N. G. Szwacki, A. Sadrzadeh, and B. I. Yakobson (2008). Phys. Rev. Lett 98, 166804.
J. Zhao, L. Wang, F. Li, and Z. Chen (2010). J. Phys. Chem. A 114, 9969.
F. Li, P. Jin, D. Jiang, L. Wang, S. B. Zhang, J. Zhao, and Z. Chen (2012). J. Chem. Phys. 136, 074302.
P. Pochet, L. Genovese, S. De, S. Goedecker, D. Caliste, S. A. Ghasemi, K. Bao, and T. Deutsch (2011). Phys. Rev. B 83, 081403R.
J. Lv, Y. Wang, L. Zhang, H. Lin, J. Zhao, and Y. Ma (2015). Nanoscale 7, 10482.
L. S. Wang (2016). Int. Rev. Phys. Chem. 35, 69.
N. M. Tam, H. T. Pham, L. V. Duong, M. P. Phamho, and M. T. Nguyen (2015). Phys. Chem. Chem. Phys. 17, 3000.
L. Zhao, X. Qu, Y. Wang, J. Lv, L. Zhang, Z. Y. Hu, G. R. Gu, and Y. Ma (2017). J. Phys. Condens. Matter. 29, 265401.
H. R. Li, H. Liu, X. X. Tian, W. Y. Zan, Y. W. Mu, H. G. Lu, J. Li, Y. K. Wang, and S. D. Li (2017). Phys. Chem. Chem. Phys. 19, 27025.
C. Romanescu, T. R. Galeev, W. L. Li, A. I. Boldyrev, and L. S. Wang (2011). Angew. Chem. Int. Ed. 50, 9334.
W.-L. Li, C. Romanescu, T. R. Galeev, Z. A. Piazza, A. I. Boldyrev, and L.-S. Wang (2012). J. Am. Chem. Soc. 134, 165.
T. R. Galeev, C. Romanescu, W. L. Li, L. S. Wang, and A. I. Boldyrev (2012). Cheminform 43, 2101.
C. Romanescu, T. R. Galeev, A. P. Sergeeva, W. L. Li, L. S. Wang, and A. I. Boldyrev (2012). J. Organomet. Chem. 721–722, 148.
C. Romanescu, T. R. Galeev, W. L. Li, A. I. Boldyrev, and L. S. Wang (2013). J. Chem. Phys. 138, 6004.
I. A. Popov, T. Jian, G. V. Lopez, A. I. Boldyrev, and L. S. Wang (2015). Nat. Commun. 6, 8654.
T. Jian, W. L. Li, X. Chen, T. T. Chen, G. V. Lopez, J. Li, and L. S. Wang (2016). Chem. Sci. 7, 7020.
T. Jian, W. L. Li, I. A. Popov, G. V. Lopez, X. Chen, A. I. Boldyrev, J. Li, and L. S. Wang (2016). J. Chem. Phys. 144, 154310.
J. Zhao, R. Shi, L. Sai, X. Huang, and Y. Su (2016). Mol. Simul. 42, 1.
B. Delley (2000). J. Chem. Phys. 113, 7756.
J. P. Perdew, K. Burke, and M. Ernzerhof (1996). Phys. Rev. Lett. 77, 3865.
X. Huang, Y. Su, L. Sai, J. Zhao, and V. Kumar (2014). J. Cluster Sci. 26, 389.
X. Huang, H. G. Xu, S. Lu, Y. Su, R. B. King, J. Zhao, and W. Zheng (2014). Nanoscale 6, 14617.
X. Huang, S. J. Lu, X. Liang, Y. Su, L. Sai, Z. G. Zhang, J. Zhao, H. G. Xu, and W. Zheng (2015). J. Phys. Chem. C 119, 10987.
X. Wu, S. J. Lu, X. Liang, X. Huang, Y. Qin, M. Chen, J. Zhao, H. G. Xu, R. B. King, and W. Zheng (2017). J. Chem. Phys. 146, 044306.
X. Q. Liang, X. J. Deng, S. J. Lu, X. M. Huang, J. J. Zhao, H. G. Xu, W. J. Zheng, and X. C. Zeng (2017). J. Phys. Chem. C 121, 7037.
L. Sai, X. Wu, N. Gao, J. Zhao, and R. B. King (2017). Nanoscale 9, 13905.
C. Adamo and V. Barone (1999). J. Chem. Phys. 110, 6158.
G. W. Trucks, M. J. Frisch, and H. B. Schlegel Gaussian 09, Revision A.01 (Gaussian Inc., Wallingford, 2009).
R.-N. Zhao, Y. Yuan, and J.-G. Han (2014). J. Theor. Comput. Chem. 13, 1450036.
I. A. Popov, W. L. Li, Z. A. Piazza, A. I. Boldyrev, and L. S. Wang (2014). J. Phys. Chem. A 118, 8098.
C. Romanescu, D. J. Harding, A. Fielicke, and L. S. Wang (2012). J. Chem. Phys. 137, 014317.
D. C. Ghosh and R. Biswas (2002). Int J Mol Sci 3, 87.
W. L. Li, T. Jian, X. Chen, H. R. Li, T. T. Chen, X. M. Luo, S. D. Li, J. Li, and L. S. Wang (2017). Chem. Commun. (Camb) 53, 1587.
R. G. Pearson (2005). J. Chem. Sci. 117, 369.
A. P. Sergeeva, I. A. Popov, Z. A. Piazza, W. L. Li, C. Romanescu, L. S. Wang, and A. I. Boldyrev (2014). Cheminform 47, 1349.
K. B. Wiberg (1968). Tetrahedron 24, 1083.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (11574040), the Fundamental Research Funds for the Central Universities (DUT16-LAB01, DUT17LAB19), and the Supercomputing Center of Dalian University of Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Wu, X. & Zhao, J. Structural Evolution and Superatoms in Molybdenum Atom Stabilized Boron Clusters: MoBn (n = 10–24). J Clust Sci 29, 847–852 (2018). https://doi.org/10.1007/s10876-018-1369-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-018-1369-3