Abstract
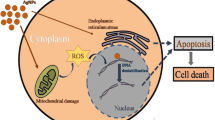
Nanoparticles research is currently an area of passionate scientific interest due to its wide variety of potential applications in therapeutic and biomedical interest. This paper presents cytotoxicity and genotoxicity of gold and silver nanoparticles synthesized by using Cassia auriculata leaf extract at room temperature on different cancer cell lines. The characterization was performed by UV-Vis spectroscopy, Fourier transform infrared (FTIR) spectroscopy, X-ray diffraction (XRD) and Transmission Electron Measurement (TEM). Cytotoxicity was analyzed against human carcinoma cells lines by MTT assay, while genotoxicity was monitored by agarose gel electrophoresis method. The UV-Vis spectroscopy reveals surface plasmon absorption maxima at 541 nm for gold and 425 nm for silver. The peaks in XRD pattern were in good agreement with the standard values of the face centered cubic form, with an average size of 21 nm in gold and 20 nm in silver. TEM reveals that the particles were spherical and polydisperse. This biological procedure for synthesis of AuNPs and AgNPs and selective inhibition of cancerous cells opens an alternative avenue to treat human cancer effectively. Least concentration of AgNPs was more toxic and AuNPs reveals dose dependent response.







Similar content being viewed by others
References
R. Siegel, D. Naishadham, and A. Jemal (2012). Cancer J. Clin. 62, 10.
World Health Organization (2012) Available at: http://www.who.int/mediacenter/factsheets/fs297/en/index/html. Accessed November 7.
A. Kotnis, R. Sarin, and R. Mulherkar (2005). J. Biosci. 30, 93.
A. Jemal, R. Siegel, E. Ward, T. Murray, J. Xu, and M. J. Thun (2007). Cancer J. Clin. 57, 43.
F. Brayand and B. Moller (2006). Nat. Rev. Cancer. 6, 63.
M. M. Gottesman, T. Fojo, and S. E. Bates (2002). Nat. Rev. Cancer. 2, 48.
A. L. Harris and D. Hochhauser (1992). Acta Oncol. 31, 205.
L. Zhang, F. X. Gu, J. M. Chan, A. Z. Wang, R. S. Langer, and O. C. Farokhzad (2008). Clin. Pharmacol. Ther. 83, 761.
V. Wagner, A. Dullaart, A. K. Bock, and A. Zweck (2006). Nat. Biotechnol. 24, 1211.
N. J. Farrer, L. Salassa, and P. J. Sadler (2009). Dalton. Trans. 48, 10690. [PubMed: 20023896].
Y. Unno, Y. Shino, F. Kondo, N. Igarashi, et al. (2005). Clin. Cancer. Res. 11, 4553.
S. A. Abraham, C. McKenzie, D. Masin, T. O. Harasym, L. D. Mayer, and M. B. Bally (2004). J. Clin. Cancer. Res. 10, 728.
J. C. Byrd, D. M. Lucas, A. P. Mone, J. B. Kitner, J. J. Drabick, and M. R. Grever (2000). J. Hematol. 101, 4547.
T. J. Webster (2006). Int. J. Nanomedicine. 1, 373.
D. Prabhu, C. Arulvasu, G. Babu, R. Manikandan, and P. Srinivasan (2013). Process Biochem. 48, 317.
M. Jeyaraj, G. Sathishkumar, G. Sivanandhan, D. MubarakAli, M. A. R. Rajesh, G. Kapildev, M. Manickavasagam, N. Thajuddin, K. Premkumar, and A. Ganapathi (2013). Colloids Surf. B Biointerfaces. 106, 86.
K. Satyavani, S. Gurudeeban, T. Ramanathan, and T. Balasubramanian (2011). J. Nanobiotechnol. 9, 43.
R. Govender, A. Phulukdaree, R. M. Gengan, K. Anand, and A. A. Chuturgoon (2013). J. Nanobiotechnol. 11, 5.
S. Lokina and V. Narayanan (2013). Chem. Sci. Trans. 2, 105. doi:10.7598/cst2013.22.
D. Raghunandan, B. Ravishankar, S. Ganachari, B. Mahesh, et al. (2011). Cancer Nanotechnol. 2, 57. doi:10.1007/s12645-011-0014-8.
S. Kaliyamurthi, G. Selvaraj, R. Thiruganasambandam, and B. Thangavel (2012). Avicenna J. Med. Biotechnol. 4, 35.
M. S. Ghassan, H. M. Wasnaa, R. M. Thorria, et al. (2013). Asian Pac. J. Trop. Biomed. 3, 58.
R. Geetha, T. Ashokkumar, S. Tamilselvan, K. Govindaraju, M. Sadiq, and G. Singaravelu (2013). Cancer Nanotechnol. 4, 91. doi:10.1007/s12645-013-0040-9.
M. Jannathul Firdhouse and P. Lalitha (2013). Cancer Nanotechnol. 4, 137. doi:10.1007/s12645-013-0045-4.
A. Nan, X. Bai, S. J. Son, S. B. Lee, and H. Ghandehari (2008). Nano Lett. 8, 2150.
S. H. Kim, H. S. Lee, D. S. Ryu, et al. (2011). Korean J. Microbiol. Biotechnol. 39, 77.
C. Carlson, S. M. Hussein, A. M. Schrand, et al. (2008). J. Phys. Chem. 112, 13608.
P. V. Asharani, G. L. K. Mun, M. P. Hande, and S. Valiyaveettil (2009). ACS Nano. 3, 279.
R. Foldbjerg, P. Olesen, M. Hougaard, D. A. Dang, H. J. Hoffmann, and H. Autrup (2009). Toxicol. Lett. 190, 156.
S. K. Kumar and J. Yadav (2009). Chem. Technol. Biotechnol. 84, 151.
S. ShivShankar, R. Akhilesh, A. Absar, and S. Murali (2004). J. Colloid Interface Sci. 275, 496.
M. V. Yezhelyev, X. Gao, Y. Xing, A. Al-Hajj, S. Nie, and R. M. O’Regan (2006). Lancet Oncol. 7, 657.
P. K. Jain, K. S. Lee, I. H. El-Sayed, and M. A. El-Sayed (2006). J. Phys. Chem. B 110, 7238.
S. L. Smitha, D. Philip, and K. G. Gopchandran (2009). Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 74, 735.
V. Usha and A. K. Bopaiah (2012). Int. J. Pharm. Bio. Sci. 3, 260.
S. A. Gaikwad, K. Asha, M. Kavita, N. R. Deshpande, and J. P. Salveka (2010). Int. J. Pharm. Tech. Res. 2, 1092.
L. G. Griffith and M. A. Swartz (2006). Nat. Rev. Mol. Cell Biol. 7, 211. doi:10.1038/nrm1858.
C. M. Goodman, C. D. McCusker, T. Yilmaz, and V. M. Rotello (2004). Bioconjug. Chem. 15, 897. doi:10.1021/bc049951i.
Y. S. Chen, Y. C. Hung, I. Liau, and G. S. Huang (2009). Nanoscale Res. Lett. 4, 858. doi:10.1007/s11671-009-9334-6.
Y. Pan, A. Leifert, D. Ruau, and S. Neuss (2009). Small. 5, 2067. doi:10.1002/smll.200900466.
M. I. Sriram, S. B. M. Kanth, K. Kalishwaralal, and S. Gurunathan (2010). Int. J. Nanomedicine. 5, 753.
M. A. Franco-Molina, E. Mendoza-Gamboa, C. A. Sierra-Rivera, et al. (2010). J. Exp. Clinical Cancer Res. 29, 148.
P. Sanpui, A. Chattopadhyay, and S. S. Ghosh (2011). ACS Appl. Mater. Interfaces. 3, 218.
P. Gopinath, S. K. Gogoi, A. Chattopadhyay, and S. S. Ghosh (2008). Nanotechnology. 19, Article ID 075104.
C. Zanette, M. Pelin, M. Crosera, et al. (2011). Toxicol. In Vitro. 25, 1053.
W. Liu, Y. Wu, C. Wang, et al. (2010). Nanotoxicology. 4, 319.
R. Foldbjerg, D. A. Dang, and H. Autrup (2011). Arch. Toxicol. 85, 743.
H. J. Yen, S. H. Hsu, and C. L. Tsai (2009). Small. 5, 1553.
S. Moaddab, H. Ahari, D. Shahbazzadeh, et al. (2011). Int. J. Nano Lett. 1, 11.
J. L. Martindale and N. J. Holbrook (2002). J. Cell. Physiol. 192, 1.
C. Carlson, S. M. Hussein, A. M. Schrand, et al. (2008). J. Phys. Chem. 112, 13608.
S. Hackenberg, A. Scherzed, and M. Kessler (2011). Toxicol. Lett. 201, 27.
R. P. Singh and P. Ramarao (2012). Toxicol. Lett. 213, 249.
M. Dizdaroglu (1991). Free. Radic. Biol. Med. 10, 225.
P. Yang and F. Gao (2002). Principle of Bioinorganic Chemistry. Science press, Beijing, pp. 322 (in Chinese).
J. Boonstra and J. A. Post (2004). Gene. 337, 1.
K. K. Panda, V. M. Achary, R. Krishnaveni, B. K. Padhi, S. N. Sarangi, S. N. Sahu, and B. B. Panda (2011). Toxicol. In Vitro. 25, 1097.
N. Lubick (2008). Environ. Sci. Technol. 42, 8617.
J. Cadet, T. Douki, and J. L. Ravanat (2010). Free. Radic. Biol. Med. 49, 9.
Acknowledgments
The financial support of University Grants Commission (F1-17.1/2010/MANF-MUS-KAR-6091) is highly appreciated. The authors are thankful to Dr. Prakasham Reddy Shetty, Indian Institute of Chemical Technology, Hyderabad for providing TEM facility.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Parveen, A., Rao, S. Cytotoxicity and Genotoxicity of Biosynthesized Gold and Silver Nanoparticles on Human Cancer Cell Lines. J Clust Sci 26, 775–788 (2015). https://doi.org/10.1007/s10876-014-0744-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-014-0744-y