Abstract
Introduction
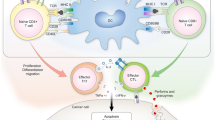
Our previous studies have revealed that nicotine-treated immature dendritic cells (imDCs) have anti-tumor effects in murine lymphoma models. The present study is to explore the preventive and therapeutic anti-tumor effects of nicotine-treated imDCs in murine lung and liver cancer.
Materials and Methods
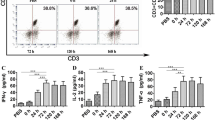
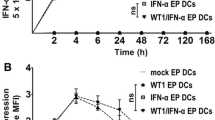
To address this objection, bone marrow-derived imDCs were firstly stimulated by nicotine in vitro and the expressions of CD80, CD86, CD40, CD11b, MHC class I and II were determined by flow cytometry. Then, DCs-dependent tumor-lysate-specific T cell proliferation, IL-12(p40+p70) secretion were determined by BrdU cell proliferation assay and enzyme-linked immunosorbent assay, respectively. The anti-tumor effects of such imDCs were further explored by intraperitoneal transfer against tumor challenge or implantation. By using kinase inhibitors, the mechanism of nicotine upregulating CD80 was finally explored by flow cytometry.
Results
The results showed that: firstly, nicotine could upregulate the expressions of CD80, CD86, CD40,CD11b, MHC class I and II molecules in imDCs. Secondly, nicotine could promote imDCs-dependent T cell priming and IL-12 secretion. Most importantly, systemic transfer of ex vivo nicotine-stimulated imDCs, which enhanced CD80 expression through PI3K activation, could reveal preventive and effectively therapeutic effects on tumor development.
Conclusions
Ex vivo nicotine stimulation can significantly improve imDCs efficacy for adaptive therapy of cancer. Nicotine-treated imDCs might be considered as a potential candidate for therapeutic tumor immunotherapy for lung and liver cancer.





Similar content being viewed by others
Abbreviations
- imDCs:
-
immature dendritic cells
- DCs:
-
dendritic cells
- nAChR:
-
nicotinic acetylcholine receptor
- LLC:
-
Lewis lung cancer
- MAPK:
-
mitogen-activated protein kinase
- PI3K:
-
phosphatidylinositol-3-kinase
- Ni:
-
nicotine
- BTX:
-
α-bungarotoxin
- TC:
-
tubocurarine chloride
- FACS:
-
flow cytometry
- MLRs:
-
mixed lymphocyte reactions
References
Reise Sousa C. Dendritic cells in a mature age. Nat Rev Immunol. 2006;6:476–83.
Banchereau J, Palucka AK. Dendritic cells as therapeutic vaccines against cancer. Nat Rev Immunol. 2005;5:296–306.
Wessler IK, Kirkpatrick CJ. The Non-neuronal cholinergic system: an emerging drug target in the airways. Pulm Pharmacol Ther. 2001;14:423–34.
Avila J, Diaz-Nido J. Tangling with hypothermia. Nat Med. 2004;10:460–1.
Mandavilli A. Nicotine fix. Nat Med. 2004;10:660–1.
Libert C. Inflammation-a nervous connection. Nature. 2003;421:328–9.
Guinet E, Yoshida K, Nouri-Shirazi M. Nicotinic environment affects the differentiation and functional maturation of monocytes derived dendritic cells (DCs). Immunol Lett. 2004;95:45–55.
Vassallo R, Tamada K, Lau JS, Kroening PR, Chen L. Cigarette smoke extract suppresses human dendritic cell function leading to preferential induction of Th-2 priming. J Immunol. 2005;175:2684–91.
Aicher A, Heeschen C, Mohanpt M, Cooke JP, Zeiher AM, Dimmeler S. Nicotine strongly activates dendritic cell-mediated adaptive immunity-potential role for progression of atherosclerotic lesions. Circulation. 2003;107:604–11.
Gao FG, Wan DF, Gu JR. Ex vivo nicotine stimulation augments the efficacy of therapeutic bone marrow-derived dendritic cell vaccination. Clin Cancer Res. 2007;13(12):3706–12.
Marrero MB, Bencherif M. Convergence of alpha 7 nicotinic acetylcholine receptor-activated pathways for anti-apoptosis and anti-inflammation: central role for JAK2 activation of STAT3 and NF-κB. Brain Res. 2009;1256:1–7.
Zhang MH, Tang H, Guo ZH, et al. Splenic stroma drives mature dendritic cells to differentiate into regulatory dendritic cells. Nat Immunol. 2004;5:1124–33.
Matsunaga K, Klein TW, Friedman H, Yamamoto Y. Involvement of nicotinic acetylcholine receptors in suppression of antimicrobial activity and cytokine responses of alveolar macrophages to Legionella pneumophila infection by nicotine. J Immunol. 2001;167:6518–24.
Gao FG, Khammanivong V, Liu WJ, Leggatt GR, Frazer IH, Fernando GJP. Antigen specific CD4+ T cell help is required to activate a memory CD8+ T cell to a fully functional tumor killer cell. Cancer Res. 2002;62:6438–41.
Nouri-Shirazi M, Guinet E. Evidence for the immunosuppressive role of nicotine on human dendritic cell functions. Immunology. 2003;109:365–73.
Wang H, Yu M, Ochani M, et al. Nicotinic acetylcholine receptor α7 subunits is an essential regulator of inflammation. Nature. 2003;421:384–8.
Nestle FO, Alijagic S, Gilliet M, et al. Vaccination of melanoma patients with peptide or tumor lysate pulsed dendritic cells. Nat Med. 1998;4:328–32.
Thurner B, Haendle I, Roder C, et al. Vaccination with mage-3A1 peptide-pulsed mature, monocyte-derived dendritic cells expands specific cytotoxic T cells and induces regression of some metastases in advanced stage IV melanoma. J Exp Med. 1999;190:1669–78.
Pulendran B, Dillon S, Joseph C, Curiel T, Banchereau J, Mohanadzadeh M. Dendritic cells generated in the presence of GM-CSF plus IL-15 prime potent CD8+ Tc1 responses in vivo. Eur J Immunol. 2004;34:66–73.
Santini SM, Lapenta C, Logozzi M, et al. TypeIinterferon as a powerful adjuvant for monocyte-derived dendritic cell development and activity in vitro and in Hu-PBL-SCID mice. J Exp Med. 2000;191:1777–88.
Pardoll D, Allison J. Cancer immunotherapy: breaking the barriers to harvest the crop. Nat Med. 2004;10:887–92.
Paleari L, Catassi A, Ciarlo M, et al. Role of alpha7-nicotinic acetylcholine receptor in human non-small cell lung cancer proliferation. Cell Prolif. 2008;41(6):936–59.
Chen RJ, Ho YS, Guo HR, et al. Rapid activation of Stat3 and ERK1/2 by nicotine modulates cell proliferation in human bladder cancer cells. Toxicol Sci. 2008;104(2):283–93.
Sun X, Ritzenthaler JD, Zhong X, et al. Nicotine stimulates PPARbeta/delta expression in human lung carcinoma cells through activation of PI3K/mTOR and suppression of AP-2alpha. Cancer Res. 2009;69(16):6445–53.
Zhang Q, Tang X, Zhang ZF, et al. Nicotine induces hypoxia-inducible factor-1alpha expression in human lung cancer cells via nicotinic acetylcholine receptor-mediated signaling pathways. Clin Cancer Res. 2007;13(16):4686–94.
Acknowledgments
The project was supported by the Natural Science Foundation of Fujian Province of China (No.2008J0112) and the grant from the National Laboratory for Oncogenes and Related Genes of China (90-08-02). We thank Professor Cao XT (Second Military Medical University, Shanghai, China) and Chen YH (University of Pennsylvania, Philadelphia, USA) for kindly providing Hepa 1-6 cell lines and polishing manuscripts. Also, we thank Mingxia Yan, Jinhua Su, and Ming Yao for excellent animal care. JR Gu designed the research and polished the paper. FG Gao and HT Li performed research and analyzed data. ZJ Li contributed to laboratory support.
Disclosure Statement
The authors declare no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gao, F.G., Li, H.T., Li, Z.J. et al. Nicotine Stimulated Dendritic Cells Could Achieve Anti-Tumor Effects in Mouse Lung and Liver Cancer. J Clin Immunol 31, 80–88 (2011). https://doi.org/10.1007/s10875-010-9459-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-010-9459-5