Abstract
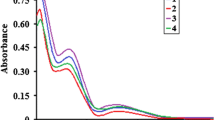
These complexes have been characterized by X-ray crystallography, spectroscopic, and cyclic voltammetry. In 1, the copper(II) ion has a distorted square-pyramidal geometry with three nitrogen atoms of the dpa ligand and two oxygen atoms of the nitrate anions. The crystal structure of 2 shows that the copper(II) ions are bridged by tp anion to form a dinuclear complex, in which each copper(II) ion exhibits a distorted square-pyramid with three nitrogen atoms of the dpa ligand, water molecule, and the oxygen atom of the bridging tp ligand. Cyclic voltammetric data indicate that 1 undergoes irreversible one-electron oxidation to the CuIII and reversible one-electron reduction to the CuI, while 2 gives one reversible oxidation and two reversible and irreversible reduced processes. The electronic spectra and redox potentials of the complexes are influenced significantly by the anionic ligands.
Graphical Abstract
The reaction of [Cu(dpa)Cl2] with K(NO3)2 and Na2tp yields mononuclear and dinuclear copper(II) complexes [Cu(dpa)(NO3)2] (1) and [Cu2(dpa)2(H2O)2(μ-tp)](tp)·6H2O (2) (dpa = di-(2-picolyl)amine, tp = terephthalate).





Similar content being viewed by others
References
Johnson DK, Murphy TB, Rose NJ, Goodwin WH, Pickart L (1982) Inorg Chim Acta 67:159
Pickart L, Goodwin WH, Burgua W, Murphy TB, Johnson DK (1983) Biochem Pharmacol 32:3868
Young MJ, Wahnon D, Hynes RC, Chin JJ (1995) Am Chem Soc 117:9441
Whitener GD, Hagadorn JR, Arnold JJ (1999) Chem Soc Dalton Trans 1249
Holland PL, Tolman WB (1999) Coord Chem Rev 190:855
Solomon EI, Sundaram UM, Machonkin TE (1996) Chem Rev 96:2563
Marsh WE, Hatfield WE, Hodgson DJ (1982) Inorg Chem 21:2679
Takizawa S, Somei H, Jayaprakash D, Sasai H (2003) Angew Chem Int Ed 42:5711
Sung N-D, Choi K-Y, Lee H-H, Lee K-C, Kim M-J (2005) Transit Met Chem 30:273
Choi K-Y, Ryu H, Sung N-D, Suh MJ (2003) Chem Crystallogr 33:947
Cortes R, Lezama L, Ruiz de Larramendi JI, Madariaga G, Luis Mesa J, Javier Zuniga F, Rojo T (1995) Inorg Chem 34:778
Shi X-S, Liu C-S, Li J-R, Guo Y, Zhou J-N, Bu X-H (2005) J Mol Struct 754:71
Sheldrick GM (1996) SADABS. University of Göttingen, Germany
Sheldrick GM (1990) Acta Crystallogr A46:467
Sheldrick GM (1997) SHELXL-97, Program for the refinement of crystal structures. University of Göttingen, Germany
Addison AW, Rao TN, Reedijik J, van Rijin J, Verschoor GC (1984) J Chem Soc Dalton Trans 1349
Cano J, De Munno G, Sanz JL, Ruiz R, Faus J, Lioret F, Julve M, Caneschi A (1997) J Chem Soc Dalton Trans 1915
Deacon GB, Phillips RJ (1980) Coord Chem Rev 33:227
Bakalbassis EG, Mrozinski J, Tsipis CA (1986) Inorg Chem 25:3684
Hathaway BJ (1991) Struct Bonding Berl 57:2801
Acknowledgment
This study was financially supported by Kongju National University in the 2007 Star Project Program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choi, KY., Kim, BR. & Ko, J. Synthesis, Properties, and Crystal Structures of Copper(II) Di-(2-picolyl)amine Complexes Containing Inorganic Salts. J Chem Crystallogr 37, 847–852 (2007). https://doi.org/10.1007/s10870-007-9258-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-007-9258-x