Abstract
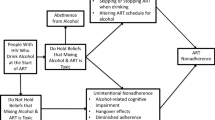
Erroneous beliefs that it is toxic to drink alcohol while taking antiretroviral therapies (ART) used for treating HIV infection, known as alcohol interactive toxicity beliefs, may at least in part account for ART nonadherence among alcohol drinkers. This study was conducted to test a conceptual framework to explain the effects of interactive toxicity beliefs on ART adherence. Computerized surveys were administered to 124 participants receiving HIV care in the southeastern US. Serial mediation model with perceived sensitivity to medicines predicting HIV viral load through three mediating variables: alcohol-ART interactive toxicity beliefs, alcohol-ART avoidance behaviors, and ART adherence. HIV viral load extracted from medical records. Perceived sensitivity to medicines predicted HIV viral load; greater perceptions of medication sensitivity predicted lower HIV viral loads. In addition, there was a significant indirect effect of the serial chain of interactive toxicity beliefs → avoidance behaviors → ART adherence, indicating partial mediation of the relationship between perceived sensitivity to medicines and higher HIV viral load. Perceived sensitivity to medicines provides a conceptual basis for the effects of alcohol-medication interactive toxicity beliefs on ART adherence. Interactive toxicity beliefs are modifiable and can be altered to prevent intentional ART nonadherence.

Similar content being viewed by others
References
Ajzen, I., & Fishbein, M. (1980). Understanding attitudes and predicting social behavior. Englewood Cliffs, NJ: Prentice-Hall.
Altice, F., Mostashari, F., & Friedland, G. (2001). Trust and the acceptance of and adherence to antiretroviral therapy. Journal of Acquired Immune Deficiency Syndrome, 28, 47–58. https://doi.org/10.1097/00126334-200109010-00008
Bangsberg, D. R., Hecht, F. M., Charlebois, E. D., Chesney, M., & Moss, A. (2001). Comparing objective measures of adherence to HIV antiretroviral therapy: Electronic medication monitors and unannounced pill counts. AIDS and Behavior, 5, 275–281. https://doi.org/10.1023/a:1011396711486
Carrieri, M. P., Villes, V., Raffi, F., Protopopescu, C., Preau, M., Salmon, D., et al. (2007). Self-reported side-effects of anti-retroviral treatment among IDUs: A 7-year longitudinal study. International Journal of Drug Policy, 18, 288–295. https://doi.org/10.1016/j.drugpo.2007.01.014
CDC. (2016). Lifetime risk of HIV diagnosis. Retrieved 2016, from http://www.cdc.gov/nchhstp/newsroom/2016/croi-press-release-risk.html
Faasse, K., Grey, A., Horne, R., & Petrie, K. J. (2015). High perceived sensitivity to medicines is associated with higher medical care utilisation, increased symptom reporting and greater information-seeking about medication. Pharmacoepidemiol Drug Safety, 24, 592–599. https://doi.org/10.1002/pds.3751
Fatch, R., Emenyonu, N. I., Muyindike, W., Kekibiina, A., Woolf-King, S., & Hahn, J. A. (2017). Alcohol interactive toxicity beliefs and ART nonadherence among HIV-infected current drinkers in Mbarara, Uganda. AIDS and Behavior, 21, 1812–1824. https://doi.org/10.1007/s10461-016-1429-3
Finitsis, D. J., Pellowski, J. A., Huedo-Medina, T. B., Fox, M. C., & Kalichman, S. C. (2016). Visual analogue scale (VAS) measurement of antiretroviral adherence in people living with HIV (PLWH): A meta-analysis. Journal of Behavioral Medicine, 39, 1043–1055. https://doi.org/10.1007/s10865-016-9770-6
Fritz, K., Morojele, N., & Kalichman, S. (2010). Alcohol: The forgotten drug in HIV/AIDS. Lancet, 376, 398–400. https://doi.org/10.1016/s0140-6736(10)60884-7
Giordano, T. P., Guzman, D., Clark, R., Charlebois, E. D., & Bangsberg, D. (2004). Measuring adherence to antiretroviral therapy in a diverse population using a visual analogue scale. HIV Clinical Trials, 5, 74–79. https://doi.org/10.1310/jfxh-g3x2-eym6-d6ug
Gupta, K., & Horne, R. (2001). The influence of health beliefs on the presentation and consultation outcome in patients with chemical sensitivities. Journal of Psychosomatic Research, 50, 131–137. https://doi.org/10.1016/s0022-3999(00)00218-x
Hayes, A. F. (2013). Model templates for PROCESS for SPSS and SAS. New York: Guilford Press.
Hayes, A. F., & Mathes, J. (2009). Computational procedures for probing interactions in OLS and logistic regression: SPSS and SAS implementations. Behavior Research Methods, 41, 929–936.
Hendershot, C. S., Stoner, S. A., Pantalone, D. W., & Simoni, J. M. (2009). Alcohol use and antiretroviral adherence: Review and meta-analysis. Journal of Acquired Immune Deficiency Syndromes, 52, 180–202. https://doi.org/10.1097/qai.0b013e3181b18b6e
Horne, R., Chapman, S. C., Parham, R., Freemantle, N., Forbes, A., & Cooper, V. (2013a). Understanding patients’ adherence-related beliefs about medicines prescribed for long-term conditions: A meta-analytic review of the Necessity-Concerns Framework. PLoS ONE, 8, e80633. https://doi.org/10.1371/journal.pone.0080633
Horne, R., Cooper, V., Gellaitry, G., Date, H. L., & Fisher, M. (2007). Patients’ perceptions of highly active antiretroviral therapy in relation to treatment uptake and adherence: The utility of the necessity-concerns framework. Journal of Acquired Immune Deficiency Syndromes, 45, 334–341. https://doi.org/10.1097/qai.0b013e31806910e3
Horne, R., Faasse, K., Cooper, V., Diefenbach, M. A., Leventhal, H., Leventhal, E., et al. (2013b). The perceived sensitivity to medicines (PSM) scale: An evaluation of validity and reliability. British Journal of Health Psychology, 18, 18–30. https://doi.org/10.1111/j.2044-8287.2012.02071.x
Horne, R., Weinman, J., & Hankins, M. (1999). The beliefs about medicines questionnaire: The development and evaluation of a new method for assessing the cognitive representation of medication. Psychology and Health, 14, 1–24. https://doi.org/10.1080/08870449908407311
International Advisory Panel on, HIV Care Continuum Optimization. (2015). IAPAC guidelines for optimizing the HIV care continuum for adults and adolescents. Journal of the International Association of Providers of AIDS Care, 14, S3–S34. https://doi.org/10.1177/2325957415613442
Jalbert, J. J., Quilliam, B. J., & Lapane, K. L. (2008). A profile of concurrent alcohol and alcohol-interactive prescription drug use in the US population. Journal of General Internal Medicine, 23, 1318–1323. https://doi.org/10.1007/s11606-008-0639-4
Judd, C. M., & Kenny, D. A. (1981). Process analysis: Estimating mediation in treatment evaluations. Evaluation Review, 5, 602–619. https://doi.org/10.1177/0193841x8100500502
Kalichman, S. C., Amaral, C. M., Swetzes, C., Jones, M., Macy, R., Kalichman, M. O., et al. (2009a). A simple single-item rating scale to measure medication adherence: Further evidence for convergent validity. Journal of the International Association of Physicians in AIDS Care, 8, 367–374. https://doi.org/10.1177/1545109709352884
Kalichman, S. C., Amaral, C. M., White, D., Swetsze, C., Kalichman, M. O., Cherry, C., et al. (2012). Alcohol and adherence to antiretroviral medications: Interactive toxicity beliefs among people living with HIV. Journal of the Association of Nurses in AIDS Care, 23, 511–520. https://doi.org/10.1016/j.jana.2011.11.005
Kalichman, S. C., Amaral, C. M., White, D., Swetsze, C., Pope, H., Kalichman, M. O., et al. (2009b). Prevalence and clinical implications of interactive toxicity beliefs regarding mixing alcohol and antiretroviral therapies among people living with HIV/AIDS. AIDS Patient Care STDS, 23, 449–454. https://doi.org/10.1089/apc.2008.0184
Kalichman, S. C., Grebler, T., Amaral, C. M., McNerey, M., White, D., Kalichman, M. O., et al. (2013). Intentional nonadherence to medications among HIV positive alcohol drinkers: Prospective study of interactive toxicity beliefs. Journal of General Internal Medicine, 28, 399–405.
Kalichman, S. C., Kalichman, M. O., Cherry, C., Hoyt, G., Washington, C., Grebler, T., et al. (2015). Intentional medication nonadherence because of interactive toxicity beliefs among HIV-positive active drug users. Journal of Acquired Immune Deficiency Syndromes, 70, 503–509.
Maisto, S. A., Conigliaro, J., McNeil, M., Kraemer, K., & Kelley, M. E. (2000). An empirical investigation of the factor structure of the AUDIT. Psychological Assessment, 12, 346–353. https://doi.org/10.1037//1040-3590.12.3.346
McCance-Katz, E. F., Gruber, V. A., Beatty, G., Lum, P. J., & Rainey, P. M. (2013). Interactions between alcohol and the antiretroviral medications ritonavir or efavirenz. Journal of Addiction Medicine, 7, 264–270. https://doi.org/10.1097/adm.0b013e318293655a
Nestoriuc, Y., Orav, E. J., Liang, M. H., Horne, R., & Barsky, A. J. (2010). Prediction of nonspecific side effects in rheumatoid arthritis patients by beliefs about medicines. Arthritis Care Research, 62, 791–799. https://doi.org/10.1002/acr.20160
Pellowski, J. A., Kalichman, S. C., Kalichman, M. O., & Cherry, C. (2016). Alcohol-antiretroviral therapy interactive toxicity beliefs and daily medication adherence and alcohol use among people living with HIV. AIDS Care, 28, 963–970. https://doi.org/10.1080/09540121.2016.1154134
Sankar, A., Wunderlich, T., Neufeld, S., & Luborsky, M. (2007). Sero-positive African Americans’ beliefs about alcohol and their Impact on anti-retroviral adherence. AIDS and Behavior, 11, 195–203. https://doi.org/10.1007/s10461-006-9144-0
Saunders, J. B., Aasland, O. G., Babor, T. F., DeLaFuente, J. R., & Grant, M. (1993). Development of the alcohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption II. Addictions, 88, 791–804. https://doi.org/10.1111/j.1360-0443.1993.tb02093.x
Simoni, J., Kurth, A. E., Pearson, C., Pantalone, D. W., Merrill, J., & Frick, P. (2006). Self-report measures of antiretroviral therapy adherence: A review with recommendations for HIV research and clinical management. AIDS and Behavior, 10, 227–331. https://doi.org/10.1007/s10461-006-9078-6
US Census. (2017). Quick facts: Georgia. Available from the US Census https://www.census.gov/quickfacts/GA. Accessed September 1, 2017.
Funding
This project was supported by National Institute of Alcohol Abuse and Alcoholism Grant R01-AA023727.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Seth C. Kalichman, Rob Horne, Harold Katner, and Dominica Hernandez declare that they have no conflicts of interest.
Human and animal rights and Informed consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research ethics committee (IRB) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all participants included in this study.
Rights and permissions
About this article
Cite this article
Kalichman, S.C., Horne, R., Katner, H. et al. Perceived sensitivity to medicines, alcohol interactive toxicity beliefs, and medication adherence among people living with HIV who drink alcohol. J Behav Med 42, 392–400 (2019). https://doi.org/10.1007/s10865-018-9987-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10865-018-9987-7