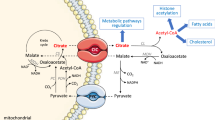
Abstract
Guanidinoacetic acid (GAA) is a natural precursor of creatine, and a possible substrate for the creatine kinase (CK) enzyme system, serving as a creatine mimetic. Its direct role in cellular bioenergetics has been confirmed in several studies, however GAA utilization by CK seems to be a second-rate as compared to creatine, and compartment-dependent. Here we discuss various factors that might affect GAA use in high-energy phosphoryl transfer in the cytosol and mitochondria.

Similar content being viewed by others
References
Amayreh W, Meyer U, Das AM (2014) Treatment of arginase deficiency revisited: guanidinoacetate as a therapeutic target and biomarker for therapeutic monitoring. Dev Med Child Neurol 56:1021–1024
Boehm EA, Radda GK, Tomlin H, Clark JF (1996) The utilisation of creatine and its analogues by cytosolic and mitochondrial creatine kinase. Biochim Biophys Acta 1274:119–128
Braissant O, Béard E, Torrent C, Henry H (2010) Dissociation of AGAT, GAMT and SLC6A8 in CNS: relevance to creatine deficiency syndromes. Neurobiol Dis 37:423–433
Ellington WR (2001) Evolution and physiological roles of phosphagen systems. Annu Rev Physiol 63:289–325
Hanna-El-Daher L, Béard E, Henry H, Tenenbaum L, Braissant O (2015) Mild guanidinoacetate increase under partial guanidinoacetate methyltransferase deficiency strongly affects brain cell development. Neurobiol Dis 79:14–27
James E, Morrison JF (1966) The reaction of phosphagens with ATP:creatine phosphotransferase. Biochim Biophys Acta 128:327–336
Kan HE, Renema WK, Isbrandt D, Heerschap A (2004) Phosphorylated guanidinoacetate partly compensates for the lack of phosphocreatine in skeletal muscle of mice lacking guanidinoacetate methyltransferase. Physiol 560:219–229
Lim K, Pullalarevu S, Surabian KT, Howard A, Suzuki T, Moult J, Herzberg O (2010) Structural basis for the mechanism and substrate specificity of glycocyamine kinase, a phosphagen kinase family member. Biochemistry 49:2031–2041
Lygate CA, Aksentijevic D, Dawson D, ten Hove M, Phillips D, de Bono JP, Medway DJ, Sebag-Montefiore L, Hunyor I, Channon KM, Clarke K, Zervou S, Watkins H, Balaban RS, Neubauer S (2013) Living without creatine: unchanged exercise capacity and response to chronic myocardial infarction in creatine-deficient mice. Circ Res 112:945–955
Nasrallah F, Feki M, Kaabachi N (2010) Creatine and creatine deficiency syndromes: biochemical and clinical aspects. Pediatr Neurol 42:163–171
Ostojic SM, Niess B, Stojanovic M, Obrenovic M (2013) Creatine metabolism and safety profiles after six-week oral guanidinoacetic acid administration in healthy humans. Int J Med Sci 10:141–147
Rowley GL, Greenleaf AL, Kenyon GL (1971) On the specificity of creatine kinase. New glycocyamines and glycocyamine analogs related to creatine. J Am Chem Soc 93:5542–5551
Schlattner U, Tokarska-Schlattner M, Wallimann T (2006) Mitochondrial creatine kinase in human health and disease. Biochim Biophys Acta 1762:164–180
Schmidt A, Marescau B, Boehm EA, Renema WK, Peco R, Das A, Steinfeld R, Chan S, Wallis J, Davidoff M, Ullrich K, Waldschütz R, Heerschap A, De Deyn PP, Neubauer S, Isbrandt D (2004) Severely altered guanidino compound levels, disturbed body weight homeostasis and impaired fertility in a mouse model of guanidinoacetate N-methyltransferase (GAMT) deficiency. Hum Mol Genet 13:905–921
Schulze A (2013) Creatine deficiency syndromes. Handb Clin Neurol 113:1837–1843
Schulze A, Bachert P, Schlemmer H, Harting I, Polster T, Salomons GS, Verhoeven NM, Jakobs C, Fowler B, Hoffmann GF, Mayatepek E (2003) Lack of creatine in muscle and brain in an adult with GAMT deficiency. Ann Neurol 53:248–251
Speer O, Neukomm LJ, Murphy RM, Zanolla E, Schlattner U, Henry H, Snow RJ, Wallimann T (2004) Creatine transporters: a reappraisal. Mol Cell Biochem 256–257:407–424
Tachikawa M, Kasai Y, Yokoyama R, Fujinawa J, Ganapathy V, Terasaki T, Hosoya K (2009) The blood-brain barrier transport and cerebral distribution of guanidinoacetate in rats: involvement of creatine and taurine transporters. J Neurochem 111:499–509
Wallimann T, Tokarska-Schlattner M, Schlattner U (2011) The creatine kinase system and pleiotropic effects of creatine. Amino Acids 40:1271–1296
Wyss M, Kaddurah-Daouk R (2000) Creatine and creatinine metabolism. Physiol Rev 80:1107–1213
Acknowledgments
This work was supported by the Serbian Ministry of Science (Grant No. 175037).
Conflict of interest
The author declares no conflicts of interest related to this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ostojic, S.M. Cellular bioenergetics of guanidinoacetic acid: the role of mitochondria. J Bioenerg Biomembr 47, 369–372 (2015). https://doi.org/10.1007/s10863-015-9619-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-015-9619-7