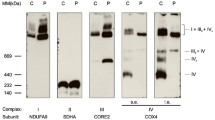
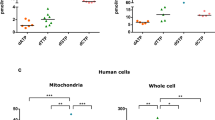
Abstract
Cytochrome c oxidase or complex IV, catalyzes the final step in mitochondrial electron transfer chain, and is regarded as one of the major regulation sites for oxidative phosphorylation. This enzyme is controlled by both nuclear and mitochondrial genomes. Among its 13 subunits, three are encoded by mitochondrial DNA and ten by nuclear DNA. In this work, an RNA interference approach was taken which led to the generation of mouse A9 cell derivatives with suppressed expression of nuclear-encoded subunit IV (COX IV) of this complex. The amounts of this subunit are decrease by 86% to 94% of normal level. A detail biosynthetic and functional analysis of several cell lines with suppressed COX IV expression revealed a loss of assembly of cytochrome c oxidase complex and, correspondingly, a reduction in cytochrome c oxidase-dependent respiration and total respiration. Furthermore, dysfunctional cytochrome c oxidase in the cells leads to a compromised mitochondrial membrane potential, a decreased ATP level, and failure to grow in galactose medium. Interestingly, suppression of COX IV expression also sensitizes the cells to apoptosis. These observations provide the evidence of the essential role of the COX IV subunit for a functional cytochrome c oxidase complex and also demonstrate a tight control of cytochrome c oxidase over oxidative phosphorylation. Finally, our results further shed some insights into the pathogenic mechanism of the diseases caused by dysfunctional cytochrome c oxidase complex.
Similar content being viewed by others
Bibliography
Capaldi RA (1990) Annu Rev Biochem 59:569–596
Barrientos A, Barros M H, Valnot I, Rotig A, Rustin P, Tzagoloff A (2002) Gene 286:53–63
Kadenbach B, Huttemann M, Arnold S, Lee I, Bender E (2000) Free Radic Biol Med 29:211–221
Attardi G, Schatz G (1988) Annu Rev Cell Biol 4:289–333
Villani G, Greco M, Papa S, Attardi G (1998) J Biol Chem 273:31829–31836
Shoubridge EA (2001) Am J Med Genet 106:46–52
Littlefield JW (1963) Proc Natl Acad Sci USA 50:568–573
Chomyn A (1996) Methods Enzymol 264:197–211
Nijtmans LG, Henderson NS, Holt IJ (2002) Methods 26:327–334
Schagger H (1996) Methods Enzymol 264:555–566
Bai Y, Attardi G (1998) Embo J 17:4848–4858
Hofhaus G, Shakeley RM, Attardi G (1996) Methods Enzymol 264:476–483
Wong A, Cortopassi, GA (2002) Biochem Biophys Res Commun 298:750–754
Hayashi J, Ohta S, Kikuchi A, Takemitsu M, Goto Y, Nonaka I (1991) Proc Natl Acad Sci USA 88:10614–10618
Schagger H (1995) Methods Enzymol 260:190–202
Bai Y, Hu P, Park JS, Deng JH, Song X, Chomyn A, Yagi T, Attardi G (2004) Ann N Y Acad Sci 1011:272–283
Chen LB (1988) Annu Rev Cell Biol 4:155–181
Robinson BH (1996) Methods Enzymol 264:454–464
Kroemer G (2003) Biochem Biophys Res Commun 304:433–435
Polster BM, Fiskum G. (2004) J Neurochem 90:1281–1289
Danielson SR, Wong A, Carelli V, Martinuzzi A, Schapira AH, Cortopassi GA (2002) J Biol Chem 277:5810–5815
Wang X (2001) Genes Dev 15:2922–2933
Bruno C, Martinuzzi A, Tang Y, Andreu AL, Pallotti F, Bonilla E, Shanske S, Fu J, Sue CM, Angelini C, DiMauro S, Manfredi G (1999) Am J Hum Genet 65:611–620
Clark KM, Taylor RW, Johnson MA, Chinnery PF, Chrzanowska-Lightowlers ZM, Andrews RM, Nelson IP, Wood NW, Lamont PJ, Hanna MG, Lightowlers RN, Turnbull DM (1999) Am J Hum Genet 64:1330–1339
Rahman S, Taanman JW, Cooper JM, Nelson I, Hargreaves I, Meunier B, Hanna MG, Garcia JJ, Capaldi RA, Lake BD, Leonard JV, Schapira AH (1999) Am J Hum Genet 65:1030–1039
Tiranti V, Corona P, Greco M, Taanman JW, Carrara F, Lamantea E, Nijtmans L, Uziel G, Zeviani M (2000) Hum Mol Genet 9:2733–2742
D'Aurelio M, Pallotti F, Barrientos A, Gajewski CD, Kwong JQ, Bruno C, Beal MF, Manfredi G (2001) J Biol Chem 276:46925–46932
Ludwig B, Bender E, Arnold S, Huttemann M, Lee I, Kadenbach B (2001) Chembiochem 2:392–403
Lenaz G, D'Aurelio M, Merlo Pich M, Genova ML, Ventura B, Bovina C, Formiggini G, Parenti Castelli G (2000) Biochim Biophys Acta 1459:397–404
Ferguson M, Mockett RJ, Shen Y, Orr WC, Sohal RS (2005) Biochem J 390:501–511
Navarro A, Sanchez Del Pino MJ, Gomez C, Peralta JL, Boveris A (2002) Am J Physiol Regul Integr Comp Physiol 282:R985–992
Sullivan PG, Brown MR (2005) Prog Neuropsychopharmacol Biol Psychiatry 29:407–410
Nicholls DG (2004) Aging Cell 3:35–40
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, Y., Park, JS., Deng, JH. et al. Cytochrome c oxidase subunit IV is essential for assembly and respiratory function of the enzyme complex. J Bioenerg Biomembr 38, 283–291 (2006). https://doi.org/10.1007/s10863-006-9052-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-006-9052-z