Abstract
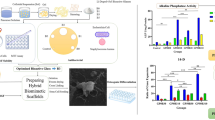
To prepare a bioactive bone substitute, which integrates biphasic calcium phosphate (BCP) and rhBMP-2/silk fibroin (SF) microsphere, and to evaluate its characteristics. Hydroxyapatite and β-tricalcium phosphate were integrated with a ratio of 60–40 %. RhBMP-2/SF (0.5 μg/1 mg) microsphere was prepared, and its rhBMP-2-release kinetics was assed. After joining pore-forming agent (Sodium chloride, NaCl), porous BCP/rhBMP-2/SF were manufactured, and its characteristics and bioactivity in vitro were evaluated. Mean diameter of rhBMP-2/SF microsphere was 398.7 ± 99.86 nm, with a loading rate of 4.53 ± 0.08 %. RhBMP-2 was released in a dual-phase pattern, of which fast-release (nearly half of protein released) focused on the initial 3 days, and slow-release sustained more than 28 days. With the increase in concentration of NaCl, greater was porosity and pore size, but smaller mechanical strength of BCP/rhBMP-2/SF. Material with 150 % (w/v) NaCl had an optimal performance, with a porosity of 78.83 %, pore size of 293.25 ± 42.77μm and mechanical strength of 31.03 MPa. Proliferation of human placenta-derived mesenchymal stem cells (hPMSCs) on leaching extract medium was similar to the normal medium (P = 0.89), which was better than that on control group (P = 0.03). Activity of alkaline phosphatase on BCP/rhBMP-2/SF surface was higher than on pure BCP at each time point except at 1 day (P < 0.05). RhBMP-2 has a burst release on early times and a sustaining release on later times. BCP/rhBMP-2/SF with 150 % (w/v) pore-forming agent has excellent porosity, pore size and mechanical strength. The biomaterial induces proliferation and differentiation hPMSCs effectively.











Similar content being viewed by others
References
LeGeros RZ, Lin S, Rohanizadeh R, Mijares D, LeGeros JP. Biphasic calcium phosphate bioceramics: preparation, properties and applications. J Mater Sci Mater Med. 2003;14(3):201–9.
Daculsi G, LeGeros RZ, Nery E, Lynch K, Kerebel B. Transformation of biphasic calcium phosphate ceramics in vivo: ultrastructural and physicochemical characterization. J Biomed Mater Res. 1989;23(8):883–94.
Houmard M, Fu Q, Genet M, Saiz E, Tomsia AP. On the structural, mechanical, and biodegradation properties of HA/β-TCP robocast scaffolds. J Biomed Mater Res B Appl Biomater. 2013;101(7):1233–42.
Ebrahimi M, Pripatnanont P, Suttapreyasri S, Monmaturapoj N. In vitro biocompatibility analysis of novel nano-biphasic calcium phosphate scaffolds in different composition ratios. J Biomed Mater Res B Appl Biomater. 2013;102(1):52–61.
Lomelino Rde O, Castro-Silva II, Linhares AB, Alves GG, Santos SR, Gameiro VS, et al. The association of human primary bone cells with biphasic calcium phosphate (β/TCP/HA 70:30) granules increases bone repair. J Mater Sci Mater Med. 2012;23(3):781–8.
Krijnen MR, Smit TH, Everts V, Wuisman PI. PLDLA mesh and 60/40 biphasic calcium phosphate in iliac crest regeneration in the goat. J Biomed Mater Res B Appl Biomater. 2009;89(1):9–17.
Bodde EW, Wolke JG, Kowalski RS, Jansen JA. Bone regeneration of porous beta-tricalcium phosphate (Conduit TCP) and of biphasic calcium phosphate ceramic (Biosel) in trabecular defects in sheep. J Biomed Mater Res A. 2007;82(3):711–22.
Burkus JK, Gornet MF, Schuler TC, Kleeman TJ, Zdeblick TA. Six-year outcomes of anterior lumbar interbody arthrodesis with use of interbody fusion cages and recombinant human bone morphogenetic protein-2. J Bone Joint Surg Am. 2009;91(5):1181–9.
Lim HP, Mercado-Pagan AE, Yun KD, Kang SS, Choi TH, Bishop J, et al. The effect of rhBMP-2 and PRP delivery by biodegradable β-tricalcium phosphate scaffolds on newbone formation in a non-through rabbit cranial defect model. J Mater Sci Mater Med. 2013;24(8):1895–903.
Ruhe PQ, Boerman OC, Russel FG, Mikos AG, Spauwen PH, Jansen JA. In vivo release of rhBMP-2 loaded porous calcium phosphate cement pretreated with albumin. J Mater Sci Mater Med. 2006;17:919–27.
Fu R, Selph S, McDonagh M, Peterson K, Tiwari A, Chou R, et al. Effectiveness and harms of recombinant human bone morphogenetic protein-2 in spine fusion: a systematic review and meta-analysis. Ann Intern Med. 2013;158(12):890–902.
Alt V, Chhabra A, Franke J, Cuche M, Schnettler R, Le Huec JC. An economic analysis of using rhBMP-2 for lumbar fusion in Germany, France and UK from a societal perspective. Eur Spine J. 2009;18(6):800–6.
Benglis D, Wang MY, Levi AD. A comprehensive review of the safety profile of bone morphogenetic protein in spine surgery. Neurosurgery. 2008;62(5 Suppl 2):ONS423–31 discussion ONS431.
Lan Levengood SK, Polak SJ, Poellmann MJ, Hoelzle DJ, Maki AJ, Clark SG, et al. The effect of BMP-2 on micro- and macroscale osteointegration of biphasic calcium phosphate scaffolds with multiscale porosity. Acta Biomater. 2010;6(8):3283–91.
Bessa PC, Balmayor ER, Azevedo HS, Nürnberger S, Casal M, van Griensven M, et al. Silk fibroin microparticles as carriers for delivery of human recombinant BMPs. Physical characterization and drug release. J Tissue Eng Regen Med. 2010;4(5):349–55.
Bessa PC, Balmayor ER, Hartinger J, Zanoni G, Dopler D, Meinl A, et al. Silk fibroin microparticles as carriers for delivery of human recombinant bone morphogenetic protein-2: in vitro and in vivo bioactivity. Tissue Eng Part C Methods. 2010;16(5):937–46.
Rong F, Qu SX. Preparation and characterization of porous calcium phosphate bioactive material for bone tissue engineering. J Chengdu Med Coll. 2008;3(4):259–62.
Berman HM, Battistuz T, Bhat TN, Bluhm WF, Bourne PE, Burkhardt K, et al. The protein data bank. Acta Crystallogr D Biol Crystallogr. 2002;58(Pt 6 No 1):899–907.
Zhang D, Tong A, Zhou L, Fang F, Guo G. Osteogenic differentiation of human placenta-derived mesenchymal stem cells (PMSCs) on electrospun nanofiber meshes. Cytotechnology. 2012;64(6):701–10.
Nery EB, LeGeros RZ, Lynch KL, Lee K. Tissue response to biphasic calcium phosphate ceramic with different ratios of HA/beta TCP in periodontal osseous defects. J Periodontol. 1992;63(9):729–35.
Yang C, Unursaikhan O, Lee JS, Jung UW, Kim CS, Choi SH. Osteoconductivity and biodegradation of synthetic bone substitutes with different tricalcium phosphate contents in rabbits. J Biomed Mater Res B Appl Biomater. 2013;102(1):80–8.
Houmard M, Fu Q, Genet M, Saiz E, Tomsia AP. On the structural, mechanical, and biodegradation properties of HA/b-TCP robocast scaffolds. J Biomed Mater Res, Part B. 2013;2013(101B):1233–42.
Schumacher M, Deisinger U, Detsch R, Ziegler G. Indirect rapid prototyping of biphasic calcium phosphate scaffolds as bone substitutes: influence of phase composition, macroporosity and pore geometry on mechanical properties. J Mater Sci Mater Med. 2010;21(12):3119–27.
Kim S, Jung UW, Lee YK, Choi SH. Effects of biphasic calcium phosphate bone substitute on circumferential bone defects around dental implants in dogs. Int J Oral Maxillofac Implants. 2011;26(2):265–73.
Oda S, Kinoshita A, Higuchi T, Shizuya T, Ishikawa I. Ectopic bone formation by biphasic calcium phosphate (BCP) combined with recombinant human bone morphogenetic protein-2 (rhBMP-2). J Med Dent Sci. 1997;44(3):53–62.
Alam I, Asahina I, Ohmamiuda K, Enomoto S. Comparative study of biphasic calcium phosphate ceramics impregnated with rhBMP-2 as bone substitutes. J Biomed Mater Res. 2001;54(1):129–38.
Matsumoto T, Okazaki M, Inoue M, Yamaguchi S, Kusunose T, Toyonaga T, et al. Hydroxyapatite particles as a controlled release carrier of protein. Biomaterials. 2004;25(17):3807–12.
Karageorgiou V, Tomkins M, Fajardo R, Meinel L, Snyder B, Wade K, et al. Porous silk fibroin-3D scaffolds for delivery of bone morphogenetic protein-2 in vitro and in vivo. J Biomed Mater Res A. 2006;78:324–34.
Diab T, Pritchard EM, Uhrig BA, Boerckel JD, Kaplan DL, Guldberg RE. A silk hydrogel-based delivery system of bone morphogenetic protein for the treatment of large bone defects. J Mech Behav Biomed Mater. 2012;11:123–31.
Ruhé PQ, Boerman OC, Russel FG, Spauwen PH, Mikos AG, Jansen JA. Controlled release of rhBMP-2 loaded poly (dl-lactic-co-glycolic acid)/calcium phosphate cement composites in vivo. J Control Release. 2005;106(1–2):162–71.
Kempen DH, Lu L, Hefferan TE, Creemers LB, Maran A, Classic KL, et al. Retention of in vitro and in vivo BMP-2 bioactivities in sustained delivery vehicles for bone tissue engineering. Biomaterials. 2008;29(22):3245–52.
Shi P, Abbah SA, Saran K, Zhang Y, Li J, Wong HK, et al. Silk fibroin-based complex particles with bioactive encrustation for bone morphogenetic protein 2 delivery. Biomacromolecules. 2013;14(12):4465–74.
Kim JW, Jung IH, Lee KI, Jung UW, Kim CS, Choi SH, et al. Volumetric bone regenerative efficacy of biphasic calcium phosphate-collagen composite block loaded with rhBMP-2 in vertical bone augmentation model of a rabbit calvarium. J Biomed Mater Res A. 2012;100(12):3304–13.
Acknowledgments
The study was financially supported by the National Natural Science Foundation of China (81071450, 81371930, 81171689 and 81301559), Natural Science Foundation of Jiang Su Province (BK2011264), Key Talented Man Project of Jiang Su Province (RC2011102), and Young Instructor Science Project of Soochow University (SDY2011A35).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, L., Gu, Y., Feng, Y. et al. Bioactivity of porous biphasic calcium phosphate enhanced by recombinant human bone morphogenetic protein 2/silk fibroin microsphere. J Mater Sci: Mater Med 25, 1709–1719 (2014). https://doi.org/10.1007/s10856-014-5194-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-014-5194-4