Abstract
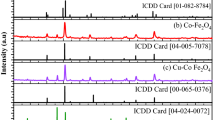
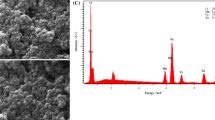
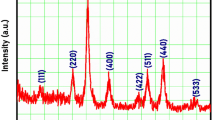
Lanthanum cerium ferrite nanoparticles has been synthesized for the first time via hydrothermal and co-precipitation method. The structural and morphological study of the nanoparticles have been examined using X-ray powder diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM) and energy dispersive X-ray spectroscopy (EDX). The electrochemical study of J1 and J2 electrodes have been examined using three electrode system in 6 M KOH electrolyte using cyclic voltammetry (CV), galvanostatic charging-discharging (GCD) and electrochemical impendence spectroscopy (EIS). The highest specific capacitance of 1195 F/g has been obtained at a scan rate of 10 mV/s from hydrothermal synthesis nanomaterial electrode (J2) and long cycling life 92.3% retention after 2000th cycles. Furthermore, the energy density and power density of the J2 electrode at a current density of 5 A/g was 59 Wh/kg and 9234 W/kg, respectively. Hence, the fabricated J2 electrode is a favorable candidate for super-capacitor applications.










Similar content being viewed by others
References
T. Zou et al., Improvement of the electrochemical performance of Li1.2Ni0.13Co0.13Mn0.54O2 cathode material by Al2O3 surface coating. J. Electroanal. Chem. 859, 113845 (2020)
J. Khan, U. Nasir, Voltage stabilization of hybrid micro-grid using super capacitors. J. Power Energy Eng. 3(06), 1 (2015)
G. Nabi et al., Cogent synergic effect of TiS2/g-C3N4 composite with enhanced electrochemical performance for supercapacitor. Ceram. Int. 46(17), 27601–27607 (2020)
A. Afzal et al., Polypyrrole/carbon nanotube supercapacitors: technological advances and challenges. J. Power Sources 352, 174–186 (2017)
B. Lobato et al., Capacitance and surface of carbons in supercapacitors. Carbon 122, 434–445 (2017)
N. Soin et al., Nanocrystalline ruthenium oxide dispersed few layered graphene (FLG) nanoflakes as supercapacitor electrodes. J. Mater. Chem. 22(30), 14944–14950 (2012)
Y. Wang, Y. Song, Y. Xia, Electrochemical capacitors: mechanism, materials, systems, characterization and applications. Chem. Soc. Rev. 45(21), 5925–5950 (2016)
S. Wang et al., Facile preparation of Ni–Mn layered double hydroxide nanosheets/carbon for supercapacitor. J. Mater. Sci.: Mater. Electron. 30(8), 7524–7533 (2019)
H. Hosseini, S. Shahrokhian, Vanadium dioxide-anchored porous carbon nanofibers as a Na+ intercalation pseudocapacitance material for development of flexible and super light electrochemical energy storage systems. Appl. Mater. Today 10, 72–85 (2018)
T. Brousse, D. Bélanger, J.W. Long, To be or not to be pseudocapacitive? J. Electrochem. Soc. 162(5), A5185–A5189 (2015)
S. Devaraj, N. Munichandraiah, High capacitance of electrodeposited MnO2 by the effect of a surface-active agent. Electrochem. Solid-State Lett. 8(7), A373–A377 (2005)
M. Toupin, T. Brousse, D. Bélanger, Influence of microstucture on the charge storage properties of chemically synthesized manganese dioxide. Chem. Mater. 14(9), 3946–3952 (2002)
J. Qi et al., Facile synthesis of N-doped activated carbon derived from cotton and CuCo2O4 nanoneedle arrays electrodes for all-solid-state asymmetric supercapacitor. J. Mater. Sci.: Mater. Electron. 30(10), 9877–9887 (2019)
J.-Y. Shieh et al., High-performance flexible supercapacitor based on porous array electrodes. Mater. Chem. Phys. 195, 114–122 (2017)
A.M. Obeidat, M.A. Gharaibeh, M. Obaidat, Solid-state supercapacitors with ionic liquid gel polymer electrolyte and polypyrrole electrodes for electrical energy storage. J. Energy Storage 13, 123–128 (2017)
L. Gonsalves, S. Mojumdar, V. Verenkar, Synthesis and characterization of Co0.8Zn0.2Fe2O4 nanoparticles. J. Therm. Anal. Calorim. 104(3), 869–873 (2011)
P. Rao, R. Godbole, S. Bhagwat, Copper doped nickel ferrite nano-crystalline thin films: a potential gas sensor towards reducing gases. Mater. Chem. Phys. 171, 260–266 (2016)
R. Sebastian et al., Structural and dielectric studies of Cr3+ doped ZnFe2O4 nanoparticles. Nano Stud. 8, 121–130 (2013)
C.-S. Hwang, N.-C. Wang, Preparation and characteristics of ferrite catalysts for reduction of CO2. Mater. Chem. Phys. 88(2–3), 258–263 (2004)
A. Manikandan et al., Optical and magnetic properties of Mg-doped ZnFe2O4 nanoparticles prepared by rapid microwave combustion method. Superlattices Microstruct. 64, 118–131 (2013)
M. Khandekar et al., Nanocrystalline Ce doped CoFe2O4 as an acetone gas sensor. Ceram. Int. 40(1), 447–452 (2014)
Y. Lin et al., Deposition of polyaniline on porous ZnFe2O4 as electrode for enhanced performance supercapacitor. J. Mater. Sci.: Mater. Electron. 29(19), 16369–16377 (2018)
A.B. Bodade et al., Conduction mechanism and gas sensing properties of CoFe2O4 nanocomposite thick films for H2S gas. Talanta 89, 183–188 (2012)
V. Kumbhar et al., Chemical synthesis of spinel cobalt ferrite (CoFe2O4) nano-flakes for supercapacitor application. Appl. Surf. Sci. 259, 39–43 (2012)
P. He et al., Reduced graphene oxide-CoFe2O4 composites for supercapacitor electrode. Russ. J. Electrochem. 49(4), 359–364 (2013)
Y.X. Zhang et al., One-pot controllable synthesis of flower-like CoFe2O4/FeOOH nanocomposites for high-performance supercapacitors. Mater. Lett. 123, 229–234 (2014)
P. Xiong, H. Huang, X. Wang, Design and synthesis of ternary cobalt ferrite/graphene/polyaniline hierarchical nanocomposites for high-performance supercapacitors. J. Power Sources 245, 937–946 (2014)
J. Huang et al., Effect of Al-doped β-Ni (OH) 2 nanosheets on electrochemical behaviors for high performance supercapacitor application. J. Power Sources 232, 370–375 (2013)
N. Ghassemi, S.S.H. Davarani, H.R. Moazami, Cathodic electrosynthesis of CuFe2O4/CuO composite nanostructures for high performance supercapacitor applications. J. Mater. Sci.: Mater. Electron. 29(15), 12573–12583 (2018)
E. Rios et al., Mixed valency spinel oxides of transition metals and electrocatalysis: case of the MnxCo3−xO4 system. Electrochim. Acta 44(8–9), 1491–1497 (1998)
Z. Du et al., Electrocatalytic performances of LaNi1−xMgxO3 perovskite oxides as bi-functional catalysts for lithium air batteries. J. Power Sources 265, 91–96 (2014)
M. De Koninck, S.-C. Poirier, B. Marsan, CuxCo3−xO4 used as bifunctional electrocatalyst physicochemical properties and electrochemical characterization for the oxygen evolution reaction. J. Electrochem. Soc. 153(11), A2103–A2110 (2006)
A.M. Abu-Dief et al., Effect of chromium substitution on the structural and magnetic properties of nanocrystalline zinc ferrite. Mater. Chem. Phys. 174, 164–171 (2016)
S.S.S. Afghahi et al., Single and double-layer composite microwave absorbers with hexaferrite BaZn0.6Zr0.3X0.3Fe10.8O19 (X= Ti, Ce, Sn) powders. Mater. Chem. Phys. 186, 584–591 (2017)
W. Shaheen, Effects of thermal treatment and doping with cobalt and manganese oxides on surface and catalytic properties of ferric oxide. Mater. Chem. Phys. 101(1), 182–190 (2007)
A.K. Gupta, M. Gupta, Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 26(18), 3995–4021 (2005)
N. Nogueira et al., X-ray diffraction and Mossbauer studies on superparamagnetic nickel ferrite (NiFe2O4) obtained by the proteic sol–gel method. Mater. Chem. Phys. 163, 402–406 (2015)
M. Islam et al., Electrical transport properties of CoZn ferrite–SiO2 composites prepared by co-precipitation technique. Mater. Chem. Phys. 109(2–3), 482–487 (2008)
Z. Yang, Z. Li, Y. Yang, Structural and magnetic properties of plate-like W-type barium ferrites synthesized with a combination method of molten salt and sol–gel. Mater. Chem. Phys. 144(3), 568–574 (2014)
U. Kurtan et al., Synthesis and magneto-electrical properties of MFe2O4 (Co, Zn) nanoparticles by oleylamine route. Ceram. Int. 42(12), 13350–13358 (2016)
J. Töpfer, A. Angermann, Nanocrystalline magnetite and Mn–Zn ferrite particles via the polyol process: synthesis and magnetic properties. Mater. Chem. Phys. 129(1–2), 337–342 (2011)
S. Bid, S. Pradhan, Preparation of zinc ferrite by high-energy ball-milling and microstructure characterization by Rietveld’s analysis. Mater. Chem. Phys. 82(1), 27–37 (2003)
J. Rehman et al., Structural, magnetic and dielectric properties of terbium doped NiCoX strontium hexagonal nano-ferrites synthesized via micro-emulsion route. Ceram. Int. 42(7), 9079–9085 (2016)
S. Azhagushanmugam, N. Suriyanarayanan, R. Jayaprakash, Magnetic properties of zinc-substituted cobalt ferric oxide nanoparticles: correlation with annealing temperature and particle size. Mater. Sci. Semicond. Process. 21, 33–37 (2014)
A. Schütz et al., High temperature (salt melt) corrosion tests with ceramic-coated steel. Mater. Chem. Phys. 159, 10–18 (2015)
K. Bindu et al., Microwave assisted growth of stannous ferrite microcubes as electrodes for potentiometric nonenzymatic H2O2 sensor and supercapacitor applications. Electrochim. Acta 217, 139–149 (2016)
M.M. Vadiyar et al., Reflux condensation mediated deposition of Co3O4 nanosheets and ZnFe2O4 nanoflakes electrodes for flexible asymmetric supercapacitor. Electrochim. Acta 222, 1604–1615 (2016)
L. Liu et al., Binary cobalt ferrite nanomesh arrays as the advanced binder-free electrode for applications in oxygen evolution reaction and supercapacitors. J. Power Sources 327, 599–609 (2016)
P. Xiong et al., Ternary manganese ferrite/graphene/polyaniline nanostructure with enhanced electrochemical capacitance performance. J. Power Sources 266, 384–392 (2014)
M. Sundararajan et al., Microwave combustion synthesis of zinc substituted nanocrystalline spinel cobalt ferrite: structural and magnetic studies. Mater. Sci. Semicond. Process. 40, 1–10 (2015)
G. Nabi et al., Role of cerium-doping in CoFe2O4 electrodes for high performance supercapacitors. J. Energy Storage 29, 101452 (2020)
B. Bhujun, M.T. Tan, A.S. Shanmugam, Evaluation of aluminium doped spinel ferrite electrodes for supercapacitors. Ceram. Int. 42(5), 6457–6466 (2016)
T. Gujar et al., Electrochemically deposited nanograin ruthenium oxide as a pseudocapacitive electrode. Int. J. Electrochem. Sci 2, 666–673 (2007)
D. Pawar et al., Synthesis of nanocrystalline nickel–zinc ferrite (Ni0.8Zn0.2Fe2O4) thin films by chemical bath deposition method. J. Alloys Compd. 509(8), 3587–3591 (2011)
J. Hao et al., Facile synthesis of 3D hierarchical flower-like Co3-xFexO4 ferrite on nickel foam as high-performance electrodes for supercapacitors. Electrochim. Acta 152, 13–18 (2015)
Z. Fan et al., Asymmetric supercapacitors based on graphene/MnO2 and activated carbon nanofiber electrodes with high power and energy density. Adv. Func. Mater. 21(12), 2366–2375 (2011)
B. Bhujun, M.T. Tan, A.S. Shanmugam, Study of mixed ternary transition metal ferrites as potential electrodes for supercapacitor applications. Results Phys. 7, 345–353 (2017)
A.E. Elkholy, F.E.-T. Heakal, N.K. Allam, Nanostructured spinel manganese cobalt ferrite for high-performance supercapacitors. RSC Adv. 7(82), 51888–51895 (2017)
P. Guo et al., Electrochemical properties of colloidal nanocrystal assemblies of manganese ferrite as the electrode materials for supercapacitors. J. Mater. Sci. 52(9), 5359–5365 (2017)
Acknowledgements
The author (Dr. Ghulam Nabi) acknowledge the Higher Education Commission (HEC) of Pakistan, for providing funding under NRPU Research Project Nos. 6502 and 6510.
Author information
Authors and Affiliations
Contributions
WR: Conceptualization, Methodology, Investigation and performed the whole experiments also wrote the manuscript. GN: Supervision, Resources. AS: Writing—review & editing, Data curation. NM: Writing—review & editing. NR: Discussed the characterizations and reviewed the manuscript. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raza, W., Nabi, G., Shahzad, A. et al. Electrochemical performance of lanthanum cerium ferrite nanoparticles for supercapacitor applications. J Mater Sci: Mater Electron 32, 7443–7454 (2021). https://doi.org/10.1007/s10854-021-05457-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-021-05457-w