Abstract
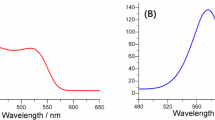
Detection of hydrogen peroxide is of significant importance for biological assays, and fluorescence methods are intensively reported for this purpose. Due to the highly oxidative property of this species, usually fluorescence quenching is obtained during the interactions and decreased signals. Unlike carbon quantum dots (CQD)-based probes, it has been reported so far, adding H2O2 to the probe increases the fluorescence intensity (with excitation/emission maxima at 450/530 nm). The probe has a linear response range in the 3.00 μM to 300 mM with a detection limit of 1.00 µM (at S/N = 3). It was successfully applied to the determination of H2O2 in spiked to serum samples.









Similar content being viewed by others
References
C. Samanta, Direct synthesis of hydrogen peroxide from hydrogen and oxygen: an overview of recent developments in the process. Appl. Catal. A 350(2), 133–149 (2008)
Y. Lu, Y. Jiang, W. Wei, H. Wu, M. Liu, L. Niu, W. Chen, Novel blue light emitting graphene oxide nanosheets fabricated by surface functionalization. J. Mater. Chem. 22(7), 2929–2934 (2012)
Y. Tian, G. Wang, J. Long, J. Cui, W. Jin, D. Zeng, Ultra-deep oxidative desulfurization of fuel with H2O2 catalyzed by phosphomolybdic acid supported on silica. Chin. J. Catal. 37(12), 2098–2105 (2016)
F. Pagliari, C. Mandoli, G. Forte, E. Magnani, S. Pagliari, G. Nardone, S. Licoccia, M. Minieri, P. Di Nardo, E. Traversa, Cerium oxide nanoparticles protect cardiac progenitor cells from oxidative stress. ACS Nano 6(5), 3767–3775 (2012)
D. Trachootham, J. Alexandre, P. Huang, Targeting cancer cells by ROS-mediated mechanisms: a radical therapeutic approach? Nat. Rev. Drug Discov. 8(7), 579 (2009)
S.G. Rhee, H2O2, a necessary evil for cell signaling. Science 312(5782), 1882–1883 (2006)
J. Liu, X. Bo, Z. Zhao, L. Guo, Highly exposed Pt nanoparticles supported on porous graphene for electrochemical detection of hydrogen peroxide in living cells. Biosens. Bioelectron. 74, 71–77 (2015)
Z. Wang, F. Liu, X. Teng, C. Zhao, C. Lu, Detection of hydrogen peroxide in rainwater based on Mg-Al-carbonate layered double hydroxides-catalyzed luminol chemiluminescence. Analyst 136(23), 4986–4990 (2011)
G. Shan, S. Zheng, S. Chen, Y. Chen, Y. Liu, Detection of label-free H2O2 based on sensitive Au nanorods as sensor. Colloids Surf. B 102, 327–330 (2013)
Y. Sang, L. Zhang, Y.F. Li, L.Q. Chen, J.L. Xu, C.Z. Huang, A visual detection of hydrogen peroxide on the basis of Fenton reaction with gold nanoparticles. Anal. Chim. Acta 659(1–2), 224–228 (2010)
M. Baghayeri, A. Amiri, Z. Alizadeh, H. Veisi, E. Hasheminejad, Non-enzymatic voltammetric glucose sensor made of ternary NiO/Fe3O4-SH/para-amino hippuric acid nanocomposite. J. Electroanal. Chem. 810, 69–77 (2018)
M. Baghayeri, H. Veisi, M. Ghanei-Motlagh, Amperometric glucose biosensor based on immobilization of glucose oxidase on a magnetic glassy carbon electrode modified with a novel magnetic nanocomposite. Sens. Actuators B 249, 321–330 (2017)
O. Tantawi, A. Baalbaki, R. El Asmar, A. Ghauch, A rapid and economical method for the quantification of hydrogen peroxide (H2O2) using a modified HPLC apparatus. Sci. Total Environ. 654, 107–117 (2019)
S. Choi, R.M. Dickson, J. Yu, Developing luminescent silver nanodots for biological applications. Chem. Soc. Rev. 41(5), 1867–1891 (2012)
X. Michalet, F. Pinaud, L. Bentolila, J. Tsay, S. Doose, J. Li, G. Sundaresan, A. Wu, S. Gambhir, S. Weiss, Quantum dots for live cells, in vivo imaging, and diagnostics. Science 307(5709), 538–544 (2005)
L. Shang, S. Dong, G.U. Nienhaus, Ultra-small fluorescent metal nanoclusters: synthesis and biological applications. Nano Today 6(4), 401–418 (2011)
R. Hardman, A toxicologic review of quantum dots: toxicity depends on physicochemical and environmental factors. Environ. Health Perspect. 114(2), 165–172 (2005)
A.M. Derfus, W.C. Chan, S.N. Bhatia, Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 4(1), 11–18 (2004)
S. Tedesco, H. Doyle, J. Blasco, G. Redmond, D. Sheehan, Oxidative stress and toxicity of gold nanoparticles in Mytilus edulis. Aquat. Toxicol. 100(2), 178–186 (2010)
P. Jing, D. Han, D. Li, D. Zhou, D. Shen, G. Xiao, B. Zou, S. Qu, Surface related intrinsic luminescence from carbon nanodots: solvent dependent piezochromism. Nanoscale Horiz. 4(1), 175–181 (2019)
A. Barati, M. Shamsipur, H. Abdollahi, Hemoglobin detection using carbon dots as a fluorescence probe. Biosens. Bioelectron. 71, 470–475 (2015)
T.T. Bui, S.-Y. Park, A carbon dot–hemoglobin complex-based biosensor for cholesterol detection. Green Chem. 18(15), 4245–4253 (2016)
S. Dolai, S.K. Bhunia, R. Jelinek, Carbon-dot-aerogel sensor for aromatic volatile organic compounds. Sens. Actuators B 241, 607–613 (2017)
C. Ding, A. Zhu, Y. Tian, Functional surface engineering of C-dots for fluorescent biosensing and in vivo bioimaging. Acc. Chem. Res. 47(1), 20–30 (2013)
S.K. Bhunia, S. Dolai, H. Sun, R. Jelinek, “On/off/on” hydrogen-peroxide sensor with hemoglobin-functionalized carbon dots. Sens. Actuators B 270, 223–230 (2018)
L.S. Walekar, P. Hu, F. Liao, X. Guo, M. Long, Turn-on fluorometric and colorimetric probe for hydrogen peroxide based on the in-situ formation of silver ions from a composite made from N-doped carbon quantum dots and silver nanoparticles. Microchim. Acta 185(1), 31 (2018)
J. Wei, L. Qiang, J. Ren, X. Ren, F. Tang, X. Meng, Fluorescence turn-off detection of hydrogen peroxide and glucose directly using carbon nanodots as probes. Anal. Methods 6(6), 1922–1927 (2014)
D. Jiang, X. Du, Q. Liu, L. Zhou, J. Qian, K. Wang, One-step thermal-treatment route to fabricate well-dispersed ZnO nanocrystals on nitrogen-doped graphene for enhanced electrochemiluminescence and ultrasensitive detection of pentachlorophenol. ACS Appl. Mater. Interfaces 7(5), 3093–3100 (2015)
M. Li, F. Xu, H. Li, Y. Wang, Nitrogen-doped porous carbon materials: promising catalysts or catalyst supports for heterogeneous hydrogenation and oxidation. Catal. Sci. Technol. 6(11), 3670–3693 (2016)
C. Karami, M.A. Taher, M. Shahlaei, A simple method for determination of mercury (II) ions by PNBS-doped carbon dots as a fluorescent probe. J. Mater. Sci. Mater. Electron. (2020). https://doi.org/10.1007/s10854-020-03157-5
V.K. Singh, P.K. Yadav, S. Chandra, D. Bano, M. Talat, S.H. Hasan, Peroxidase mimetic activity of fluorescent NS-carbon quantum dots and their application in colorimetric detection of H2O2 and glutathione in human blood serum. J. Mater. Chem. B 6(32), 5256–5268 (2018)
H.Y. Shin, B.-G. Kim, S. Cho, J. Lee, H.B. Na, M.I. Kim, Visual determination of hydrogen peroxide and glucose by exploiting the peroxidase-like activity of magnetic nanoparticles functionalized with a poly (ethylene glycol) derivative. Microchim. Acta 184(7), 2115–2122 (2017)
Y. Zhang, X. Yang, Z. Gao, In situ polymerization of aniline on carbon quantum dots: a new platform for ultrasensitive detection of glucose and hydrogen peroxide. RSC Adv. 5(28), 21675–21680 (2015)
S. Chen, X. Hai, X.-W. Chen, J.-H. Wang, In situ growth of silver nanoparticles on graphene quantum dots for ultrasensitive colorimetric detection of H2O2 and glucose. Anal. Chem. 86(13), 6689–6694 (2014)
G. Su, Y. Wei, M. Guo, Direct colorimetric detection of hydrogen peroxide using 4-nitrophenyl boronic acid or its pinacol ester. Am. J. Anal. Chem. 2(08), 879 (2011)
S. Liu, J. Tian, L. Wang, H. Li, Y. Zhang, X. Sun, Stable aqueous dispersion of graphene nanosheets: noncovalent functionalization by a polymeric reducing agent and their subsequent decoration with Ag nanoparticles for enzymeless hydrogen peroxide detection. Macromolecules 43(23), 10078–10083 (2010)
H. Yang, F. Li, C. Zou, Q. Huang, D. Chen, Sulfur-doped carbon quantum dots and derived 3D carbon nanoflowers are effective visible to near infrared fluorescent probes for hydrogen peroxide. Microchim. Acta 184(7), 2055–2062 (2017)
Acknowledgements
The authors gratefully acknowledge the Research Council of Kermanshah University of Medical Sciences (Grant Number: 980547) for financial support.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Star, B.G., Shahlaei, M. & Karami, C. A novel fluorescent turn-on probe for hydrogen peroxide based on carbon dots. J Mater Sci: Mater Electron 32, 5615–5623 (2021). https://doi.org/10.1007/s10854-021-05283-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-021-05283-0